 |
 |
 |
| |
Final Results of the Phase IIIb IDEAL Study (Individualized Dosing Efficacy
vs. Flat Dosing to Assess OptimaL PegInterferon Therapy)
|
| |
| |
Reported by Jules Levin
European Association for the Study of the Liver (43rd EASL Conference)
Milan, Italy
April 26, 2008
MS Sulkowski, E Lawitz, ML Shiffman, AJ Muir, G Galler, J McCone, L
Nyberg, WM Lee, R Ghalib, E Schiff, J Galati, B Bacon, M Davis, P
Mukhopadhyay, Koury K, S Noviello, L Pedicone, J Albrecht, J
McHutchison on behalf of the IDEAL Study Team
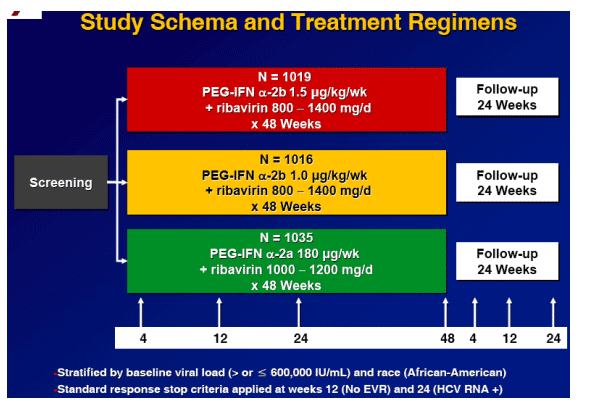
Study Rationale and Aim
Two co-primary comparisons
1 PEG-IFN _-2b: 1.5 _g/kg vs 1.0 _g/kg
-- Post-approval commitment to FDA
-- 1.0 dose approved as monotherapy, few data with ribavirin
2. PEG-IFN _-2b 1.5 _g/kg vs PEG-IFN _-2a 180 _g regimens
-- Cross study comparisons problematic
-- Scientific comparison of approved combination regimens needed to
guide treatment
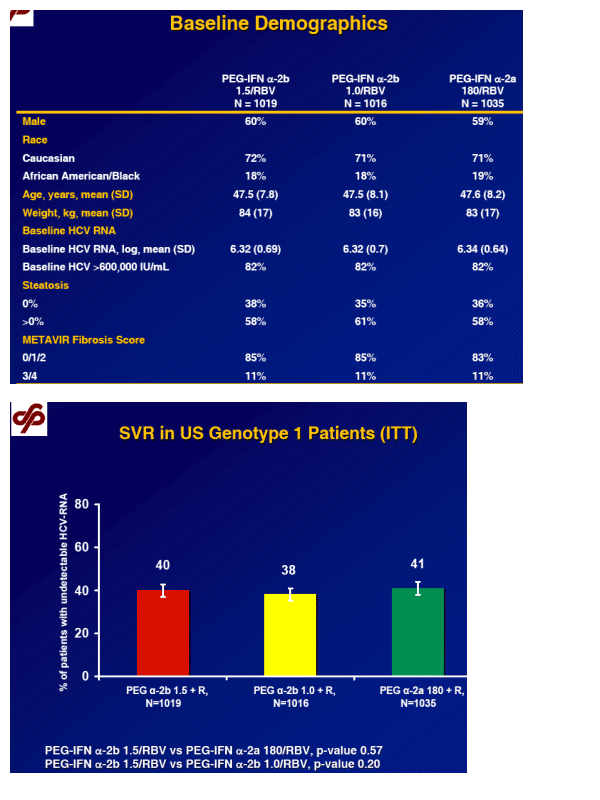
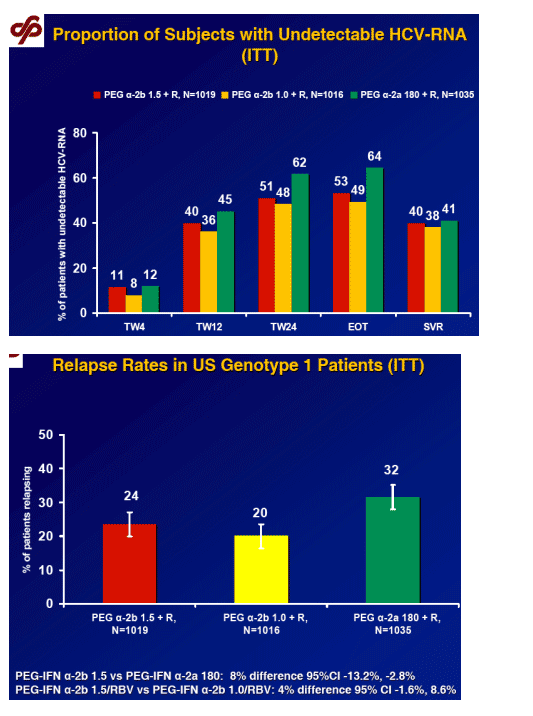
Primary endpoint
SVR, defined as HCV RNA < 29 IU/mL at the end of follow-up (week 24 or, if missing, week 12)
Manns M, et al. Lancet 2001; Fried M, et al. NEJM 2002
Study Design
Design: Randomized, open-label, parallel-group study
Population: Treatment-naive, genotype 1
Location: 118 US academic and community centers
Entry criteria:
-- Age 18 - 70 years old
-- Weight 40 - 125 kg
-- Compensated liver disease with liver biopsy with 3 years
Independent Data Analysis Committee:
-- 2 Co-lead principal investigators
-- 2 Independent expert clinicians
-- 1 External statistician who led the analyses



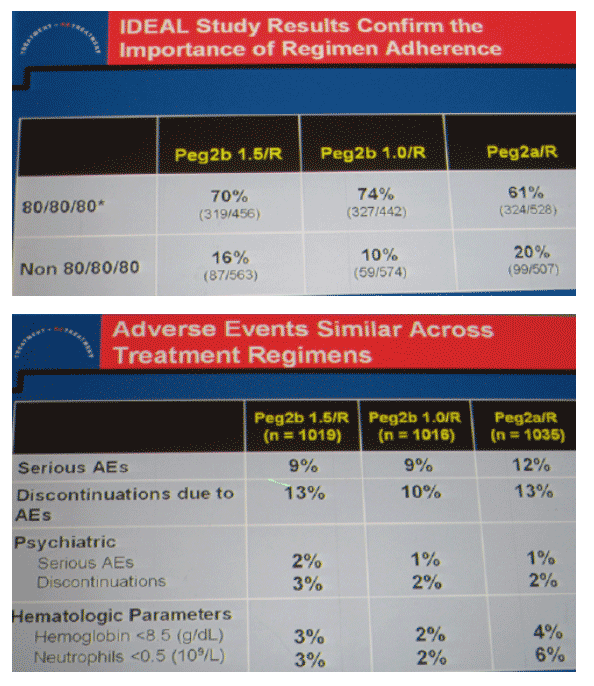
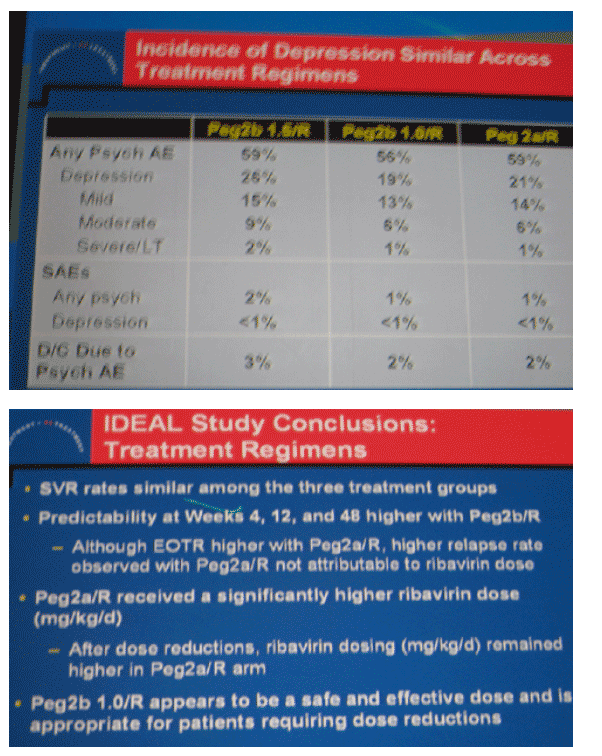

|
| |
|
 |
 |
|
|