 |
 |
 |
| |
Tenofovir Disoproxil Fumarate (TDF) Versus Emtricitabine Plus TDF for Treatment of Chronic Hepatitis B (CHB) in Subjects with Persistent Viral Replication Receiving Adefovir Dipivoxil (ADV)
|
| |
| |
Reported by Jules Levin
43rd EASL
April 23-27, 2008
Milan, Italy
T Berg,1 B Moller,1 H Trinh,2 S Chan,3 P Marcellin,4 E Suarez,5 A Snow-Lampart,6 D Frederick,6 D Oldach,6 J Sorbel,6 K Borroto-Esoda,6 and F Rousseau6
1Berlin, Germany; 2San Jose, CA, USA; 3Flushing, NY, USA;
4Clichy, France; 5Sevilla, Spain; 6Gilead Sciences, Inc., Durham, NC, USA
AUTHOR CONCLUSIONS
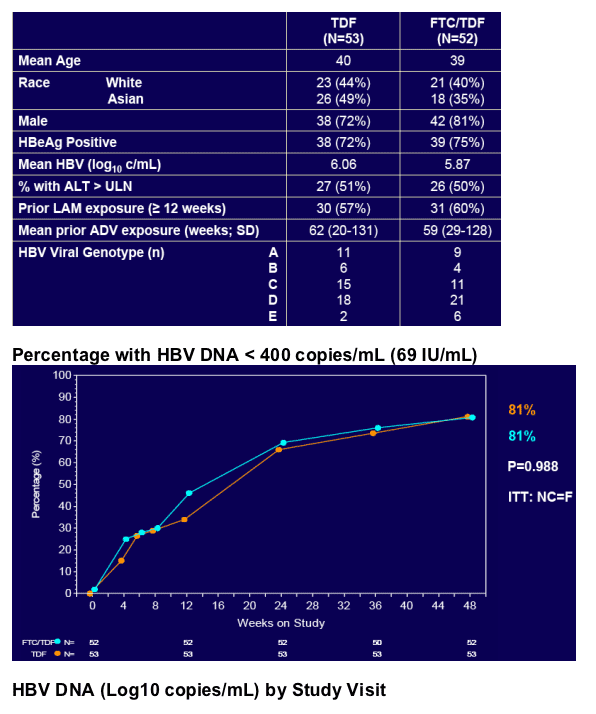
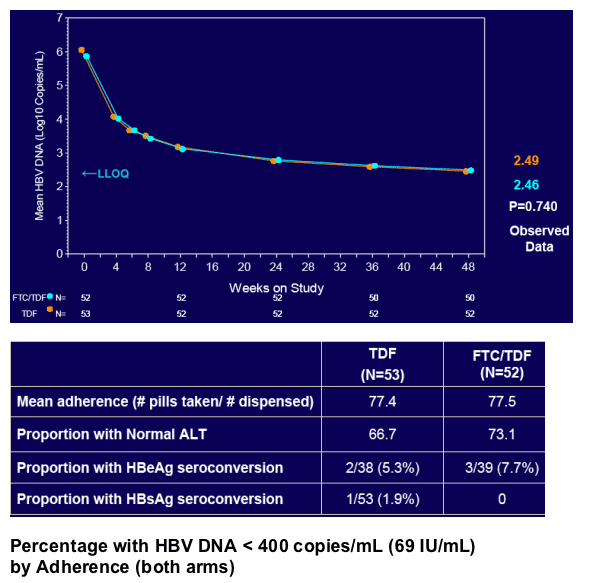
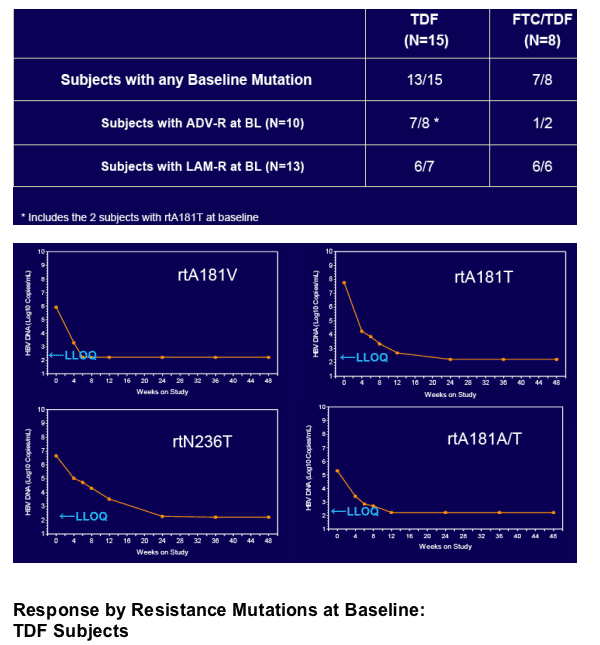
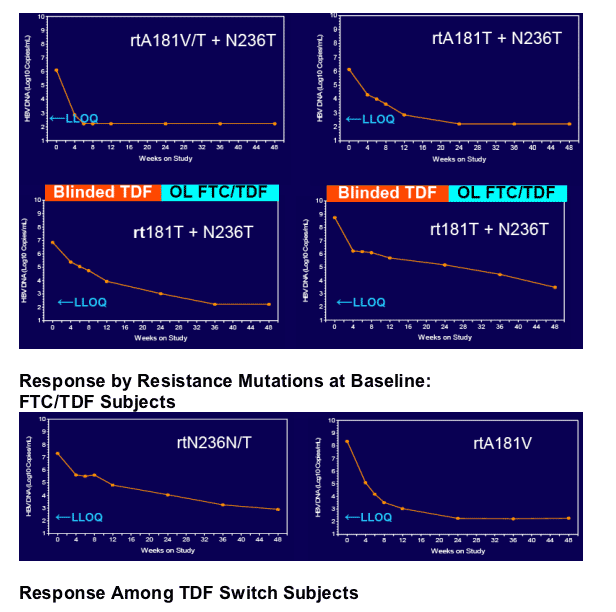
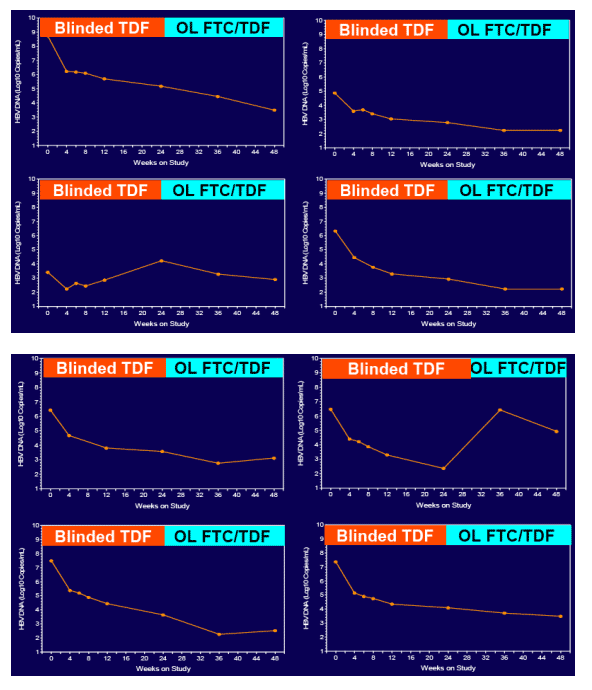
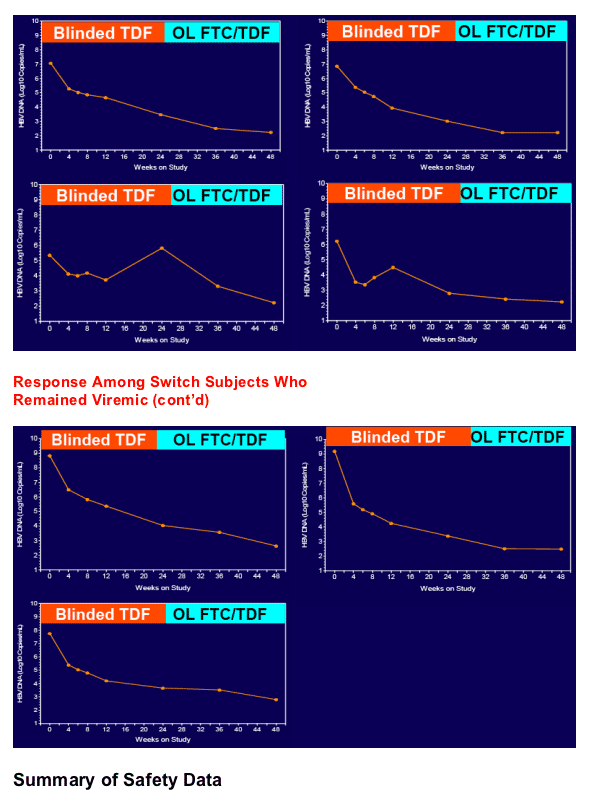
In patients with incomplete viral suppression on ADV (most with prior/current LAM use), TDF and FTC/TDF were well tolerated and achieved complete viral suppression in 81% of patients at week 48
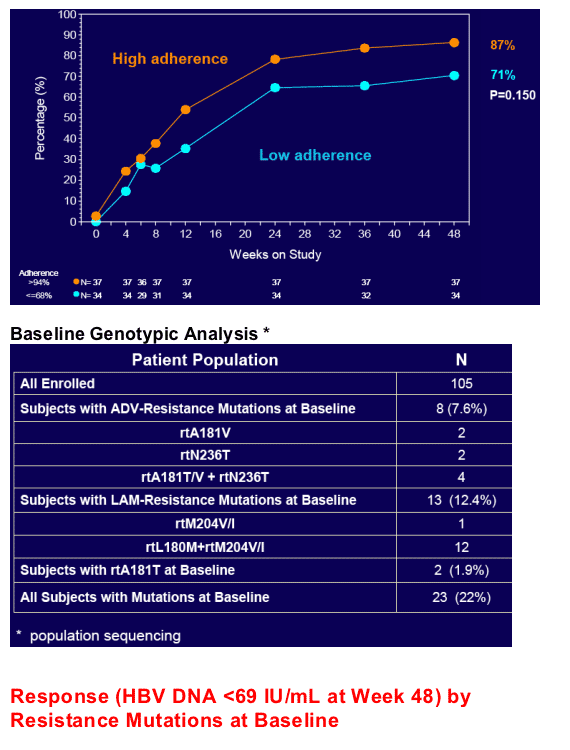
Virologic response was independent of pre-existing ADV- or LAM-associated mutations but dependent upon adherence to study medication (s)
Evaluation beyond 1 year is necessary to determine if FTC/TDF or TDF monotherapy is the preferred treatment strategy in this heavily pretreated population
INTRODUCTION
Virologic response to adefovir (ADV) is variable and a significant proportion of ADV treated patients showed persistent viremia on treatment.
Tenofovir disoproxil fumarate (TDF) has been shown to be superior to ADV in two large clinical trials of HBV-infected patients who were primarily naive to prior treatment.
In previous pilot studies, TDF also demonstrated significant efficacy in patients with persistent viremia on ADV therapy. 1,2
Emtricitabine (FTC) has activity against HBV and HIV. Fixed-dose combination of TDF and FTC is licensed for inclusion in HIV treatment regimens.
1 Van Bommel F et al. Hepatology 2006;44:318-325; 2 Tan et al. J Hepatol 2008;
Objectives
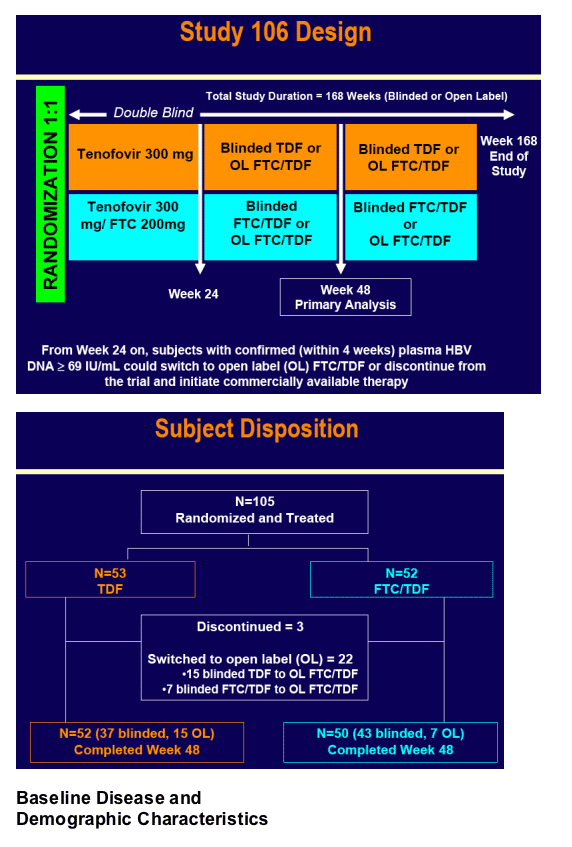
Primary objective: a comparison of treatment strategies
- Compare the antiviral activity of
· Monotherapy with TDF 300 mg QD [with option to switch to FTC 200 mg + TDF 300 mg QD combination therapy after 24 weeks for subjects with confirmed persistent HBV replication (HBV DNA > 69 IU/mL)],
versus
· Fixed-dose combination of FTC 200 mg + TDF 300 mg QD for 48 weeks in HBV-infected patients with persistent HBV replication on ADV 10 mg QD for at least 6 months
Secondary objective:
- To evaluate the safety and tolerability of TDF versus FTC/TDF
Methods
Key eligibility criteria
- 18-69 years of age
- HBeAg positive or negative
- Currently treated with ADV 10 mg QD (for ≥ 24 weeks but ≦ 96 weeks); concomitant and past treatment with lamivudine permitted
- HBV DNA ≥ 172 IU/mL (1000 copies/mL) (Roche Cobas TaqMan Assay, lower limit of quantification 29 IU/mL <169 copies/mL>)
- ALT levels < 10 _ the upper limit of normal (ULN)
- Compensated liver disease; no evidence of HCC
- No co-infection with HCV, HIV, or HDV
Assessments:
- HBV DNA evaluated on Weeks 1, 2, 3, 4, 6, 8 and 12; and every 12 weeks thereafter
- Serum chemistry, hematology, and urinalysis were evaluated every 4 weeks through Week 24 and every 12 weeks thereafter
- HBeAg and HBsAg testing every 12 weeks
- HBV DNA measured using Roche COBAS TaqMan assay (LLOQ=29 IU/mL;169 copies/mL)
- Baseline HBV polymerase sequencing for all subjects

|
| |
|
 |
 |
|
|