 |
 |
 |
| |
Tenofovir Disoproxil Fumarate (TDF) for the Treatment of HBeAg-Positive Chronic Hepatitis B: Week 72 TDF Data and Week 24
Adefovir Dipivoxil Switch Data (Study 103)
|
| |
| |
Reported by Jules Levin
43rd Annual Meeting of the European Association for the Study of the Liver
April 23-27, 2008
Milan, Italy
J Heathcote,1 J George,2 S Gordon,3 J-P Bronowicki,4 J Sperl,5 R Williams,6 P Martin,7 M Beniowski,8 J Sorbel,9 J Anderson,9 E Mondou,9 A Snow-Lampart9 and F Rousseau9
1University of Toronto, Toronto Canada; 2 Westmead Hospital, Westmean Australia; 3Henry Ford Hospital, Detroit, MI USA; 4Hospital de Brabois, Vandoeuvre les Nancy, France; 5Institute of Clinical & Experimental Medicine, Praha, Czech Republic; 6University College London Hospital, London UK; 7Mount Sinai School of Medicine, New York, NY USA; 8Oddzial Kliniczny Chorob Zakaznych, Chorzow Poland; 9Gilead Sciences, Inc., Durham, NC USA
Author Conclusions for HBeAg Positive Study (Week 72)
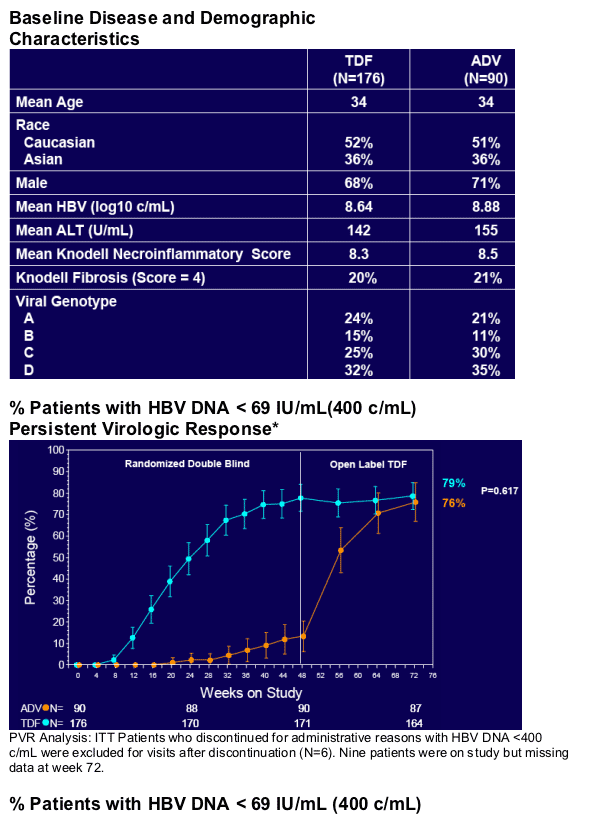
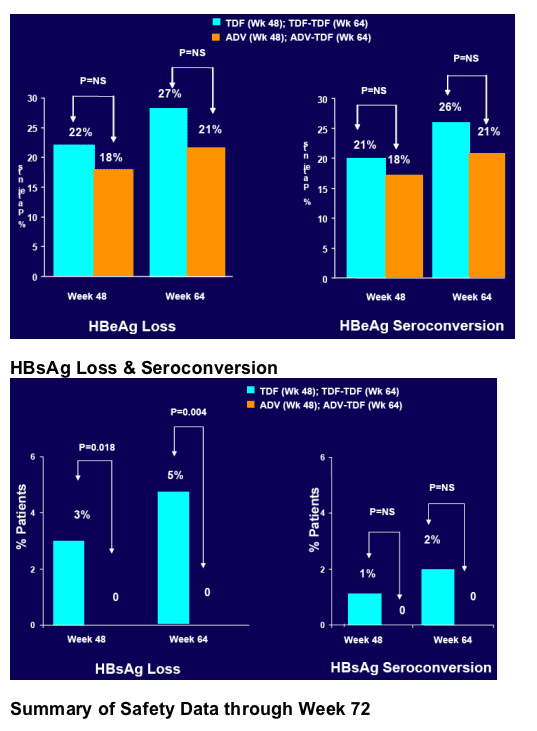
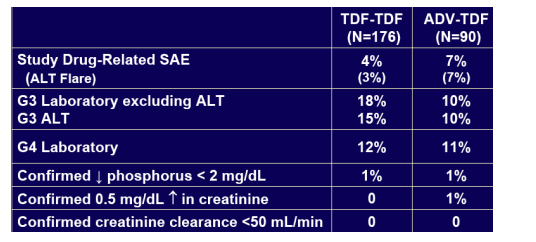
· Week 72 results (79% <400 c/ml) confirm and reinforce the continuing favourable safety and efficacy of TDF for treatment of CHB
· Progressive increase in HBsAg loss (5% by Week 64)
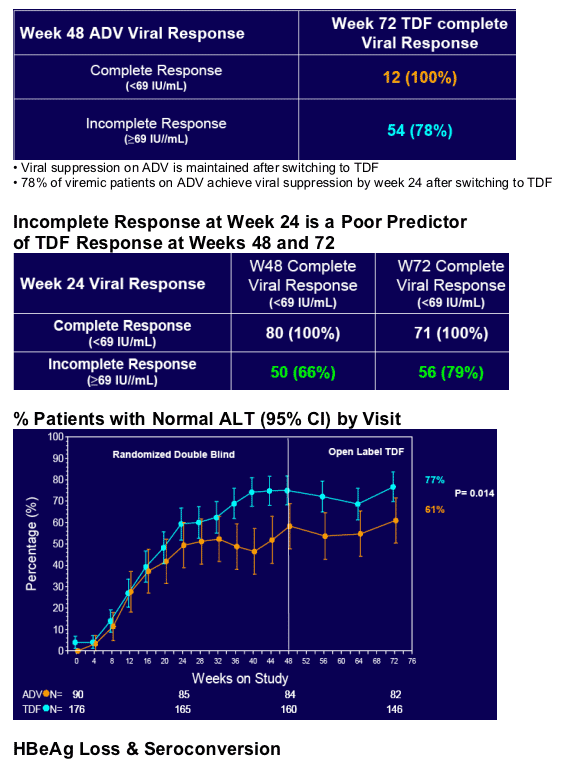
· A Week 24 HBV DNA predictive rule is not useful for TDF
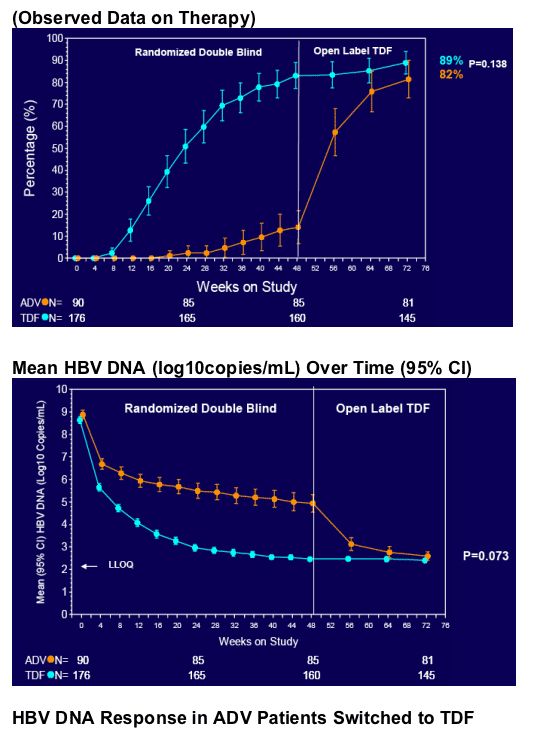
· ADV patients can safely and effectively switch to TDF
· Study 103 is continuing through Year 8
INTRODUCTION
· Tenofovir DF (TDF) is a nucleotide analogue that is an obligate HBV DNA chain terminator
· TDF in combination therapy is approved for HIV-1
· Recent favourable EMEA opinion for the treatment of CHB (EU Aprroved TDF during EASL meeting)
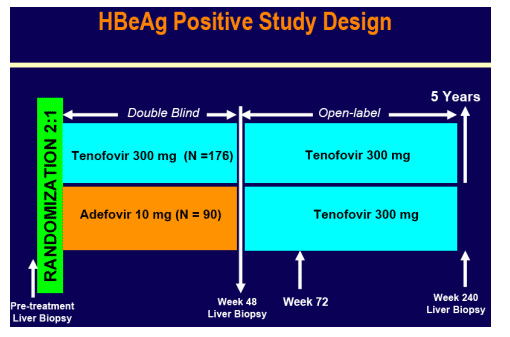
METHODS
Key eligibility criteria:
- HBeAg+
- Treatment naive
- 18-69 years of age
- Compensated liver disease
- HBV DNA >106 copies/mL
- ALT > 2x and < 10xULN
- Knodell Necroinflammatory score ≥ 3
- HIV, HDV, HCV negative
88% of patients still on study at week 72
Assessments
· HBV DNA and laboratory analyses every 4 weeks during Year 1 and every 8 weeks during Year 2
· HBeAg and HBsAg every 12 weeks during Year 1 and every 16 weeks during Year 2
· HBV DNA measured using Roche COBAS TaqMan assay (Virologic response 69 IU/mL or 400 c/mL)
· Resistance surveillance
Resistance Surveillance
· No mutations associated with TDF resistance detected at week 48
· No TDF-treated patient had a loss of viral response (≥400 copies/mL after being <400 copies/mL) between Weeks 48 and 72
|
| |
|
 |
 |
|
|