 |
 |
 |
| |
Tenofovir Disoproxil Fumarate (TDF) for the
Treatment of HBeAg-Negative Chronic Hepatitis B: Week 72 TDF Data and Week 24 Adefovir Dipivoxil Switch Data (Study 102)
|
| |
| |
Reported by Jules Levin
43rd Annual Meeting of the European Association for the Study of the Liver
April 23-27, 2008
Milan, Italy
P Marcellin,1 I Jacobson,2 F Habersetzer,3 H Senturk,4 P Andreone,5 C Moyes,6 A Horban,7 G Teuber,8 J Sorbel,9 J Anderson,9 E Mondou,9 J Quinn,9 and F Rousseau9
1University of Paris, Clichy France; 2Weill Medical College of Cornell University, New York, NY, USA; 3Hopital Civil de Strasbourg, Strasbourg France; 4Istanbul University, Istanbul, Turkey; 5University of Bologna, Bologna, Italy; 6The
Hepatitis Foundation of New Zealand, Whakatane, New Zealand; 7Hospital of Infectious Diseases, Warsaw, Poland; 8Johann-Wolfgang Goethe University, Frankfurt, Germany; 9Gilead Sciences, Inc., Durham NC USA
Author Conclusions for HBeAg Negative Study (Week 72)
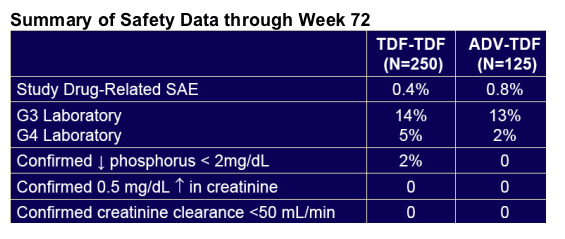
· Week 72 results confirm and reinforce the favourable safety and efficacy of TDF for treatment of CHB
· No resistance identified through 72 weeks of therapy
· A Week 24 predictive rule is not useful for TDF
· ADV patients can safely and effectively switch to TDF
· Study 102 is continuing through Year 8
INTRODUCTION
· Tenofovir DF (TDF) is a nucleotide analogue that inhibits viral replication by direct binding to HBV RT resulting in chain termination after being incorporated into HBV DNA
· TDF in combination therapy is approved for the treatment of HIV-1
· Recent favourable EMEA opinion for the treatment of CHB (TDF was approved in Europe during EASL)
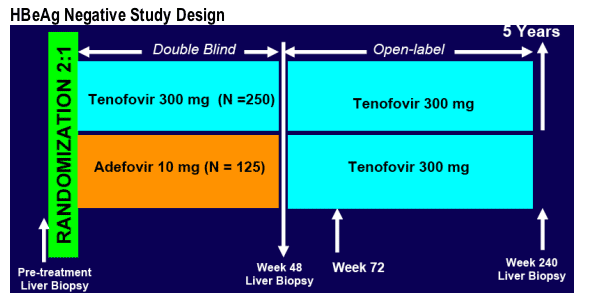
METHODS
Key eligibility criteria
· HBeAg- subjects
· Age 18-69 years
· Compensated liver disease
· Lamivudine experienced or naive
· HBV DNA > 105 c/mL
· ALT ≥ ULN and <10xULN
· Knodell Necroinflammatory score ≥ 3
· HIV, HDV, HCV negative
· 90 % of enrolled patients were still on study at week 72
Assessments
· HBV DNA and laboratory analyses every 4 weeks during Year 1 and every 8 weeks during Year 2
· HBsAg every 12 weeks during Year 1 and every 16 weeks during Year 2
· HBV DNA measured using Roche COBAS TaqMan assay (response <69 IU/mL or 400 c/mL)
· Resistance surveillance
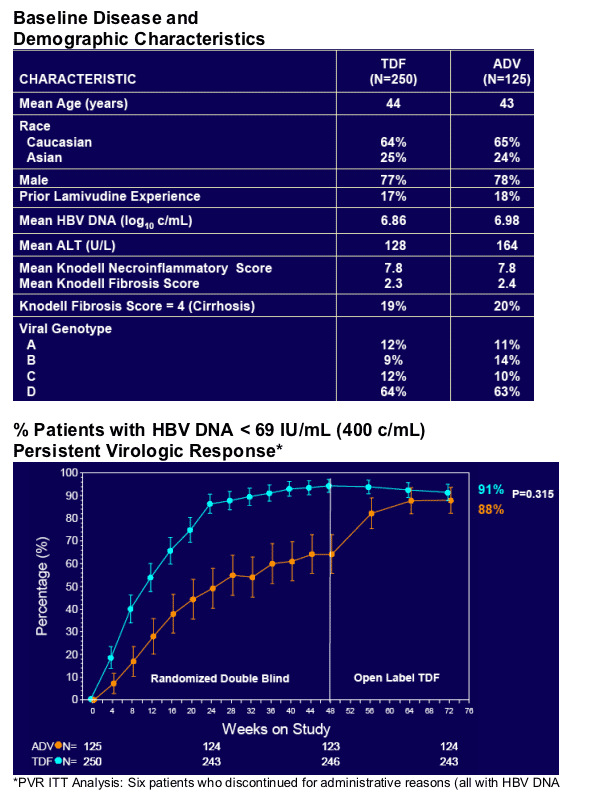
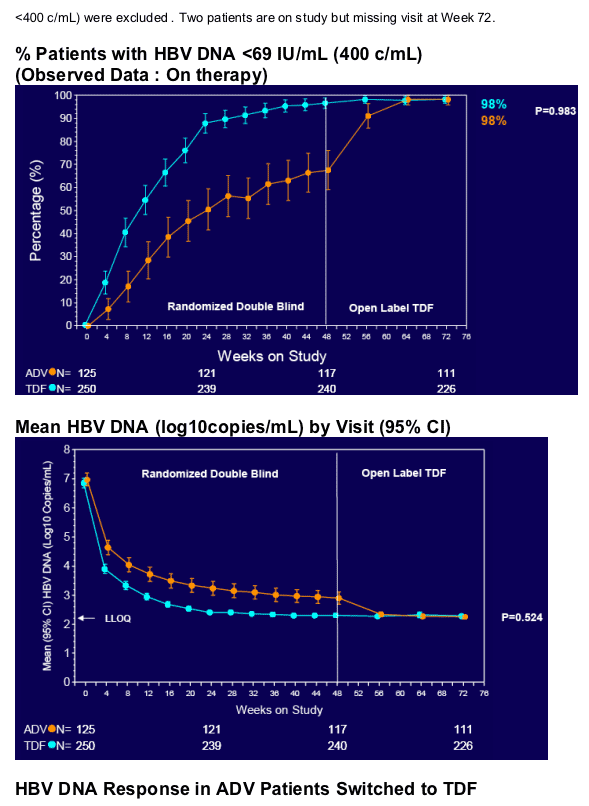
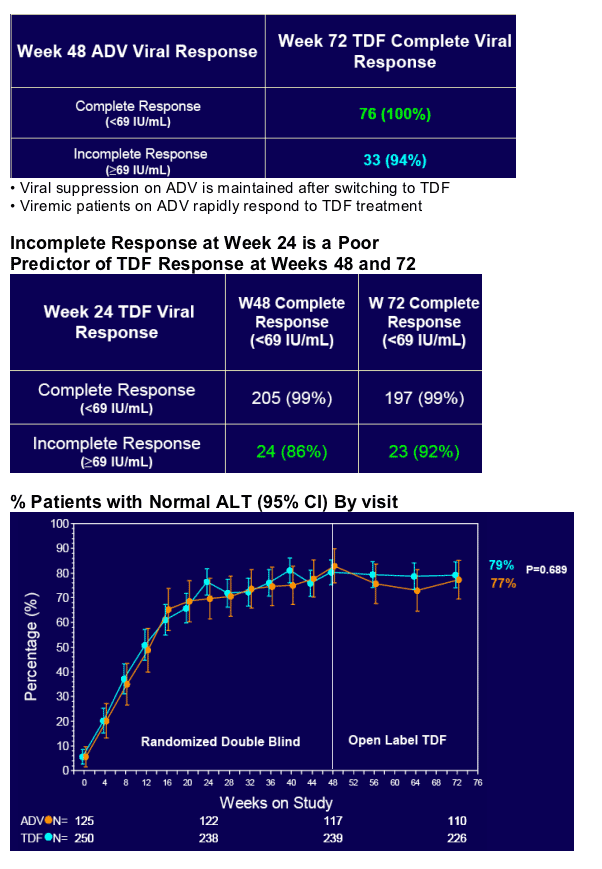
Resistance Surveillance
· Two patients (originally randomized to TDF) had a loss of viral response (≥400 copies/mL after being <400 copies/mL) between Weeks 48 and 72
· Non-adherence documented for both patients
· Neither patient developed mutations associated with TDF resistance
|
| |
|
 |
 |
|
|