 |
 |
 |
| |
The Antiviral Response To Tenofovir Disoproxil Fumarate (TDF) is Comparable in Lamivudine (LAM)-NaIve and LAM-Experienced Subjects
Treated for Chronic Hepatitis B (CHB)
|
| |
| |
Reported by Jules Levin
43rd Annual Meeting of the European Association for the Study of the Liver
April 23-27, 2008
Milan, Italy
M Manns,1 L Jeffers,2 G Dalekos,3 T Berg,4 C Trepo,5 S Roberts,6 M Prieto,7 M Rizzetto,8 J Sorbel,9 J Anderson,9 E Mondou, 9 J Quinn,9 and F Rousseau9
1Medical School of Hannover, Hannover, Germany; 2University of Miami School of Medicine Center for Liver Diseases, Miami FL; 3University of Thessaly Medical School Academic Liver Unit, Larissa Greece; 4Medizinische Klinik mit Schwerpunkt Hepatologie und Gastroenterologie, CharitŽ UniversitŠtsmedizin, Berlin; 5Hopital de Hotel Dieu, Service d' Hepatogastro enterologie, Lyon France; 6Alfred Hospital, Dept of Gastroenterology, Melbourne, Australia; 7Hospital La Fe, Servicio de Medicina Digestiva, Valencia Spain, 8 University of Torino, Dipartimento di Gastroenterologia, Torino Italy, 9Gilead Sciences, Inc., Durham NC USA
AUTHOR CONCLUSIONS
· Tenofovir DF demonstrated potent and consistent antiviral efficacy in both LAM Exp and LAM Naive patients
· No mutations associated with tenofovir DF resistance were observed in either LAM Exp or LAM Naive patients
· Tenofovir DF was well tolerated in both LAM Exp and LAM NaIve patients
INTRODUCTION
· Based on data from the Phase 3 studies, GS-174-0102 (HBeAg negative) and GS-174-0103 (HBeAg positive), tenofovir DF showed superior efficacy to adefovir dipivoxil
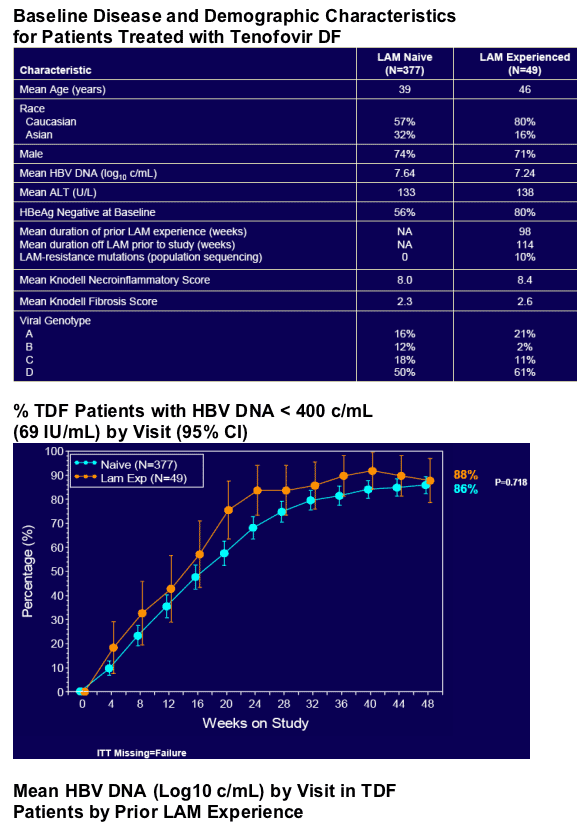
- At Week 48, 76% of HBeAg+ patients and 93% of HBeAg- patients receiving tenofovir DF had HBV DNA <400 c/mL (69 IU/mL)
· Both lamivudine naIve (<12 weeks treatment) and lamivudine-experienced patients were enrolled into these studies
OBJECTIVE
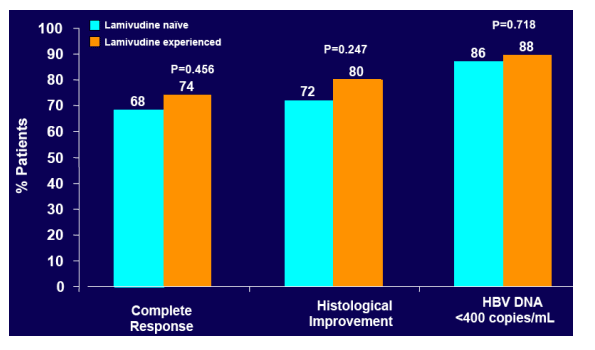
· To evaluate the Week 48 response to tenofovir DF in the subset of lamivudine-experienced (LAM Exp) compared to lamivudine-naIve (LAM NaIve) patients enrolled in these phase 3 studies
ENDPOINTS
· Proportion of patients with HBV DNA <400 c/mL (69 IU/mL)
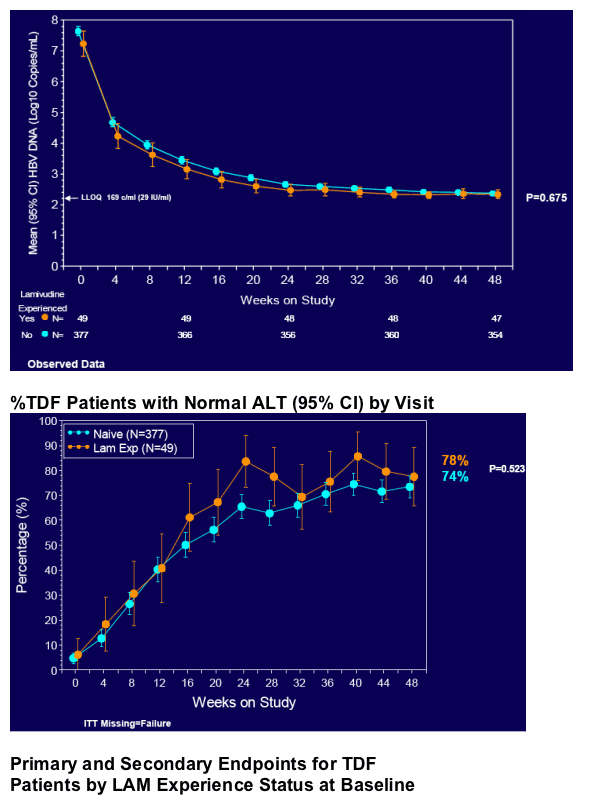
· Mean HBV DNA change from baseline
· Proportion of patients with normal ALT
· Safety and tolerability
METHODS
Key eligibility criteria for Studies 102 (HBeAg-) and 103 (HBeAg+)
· Age 18-69 years
· Compensated liver disease
· HBV DNA > 105 c/mL (HBeAg-) or HBV DNA > 106 c/mL (HBeAg+)
· ALT > ULN < 10xULN (HBeAg-) or ALT > 2xULN <10xULN (HBeAg+)
· Knodell Necroinflammatory score ≥ 3
· HIV, HDV, HCV negative
Assessments
· Liver biopsies pre-treatment and between Week 44 and 48
· HBV DNA and Laboratory analyses (serum chemistry, hematology, and urinalysis) every 4 weeks
· HBeAg and HBsAg every 12 weeks
· HBV DNA measured using the Roche COBAS TaqMan assay (LLOQ=169 c/mL or 29 IU/mL)
· Resistance surveillance performed at baseline for all patients and Week 48 for all viremic patients (≥ 400 c/mL or 69 IU/mL)

· Study 102 actively enrolled both LAM experienced and LAM-naIve patients
· Study 103 enrolled 8 LAM experienced patients despite LAM-naIve inclusion criteria
Resistance Surveillance
· No resistance to tenofovir DF was detected at Week 48 in any subject
· Across both pivotal studies 39/426 subjects had HBV DNA > 400 c/mL (69 IU/mL) at Week 48, which included 4 patients with LAM experience
· No LAM experienced or naIve patient had genotypic changes associated with resistance to tenofovir

|
| |
|
 |
 |
|
|