 |
 |
 |
| |
Boceprevir (NS3 Protease Inhibitor)
Combination Therapy in Non Responders:
Phase II Dose Finding Study
|
| |
| |
Reported by Jules Levin
43rd EASL
April 23-27, 2008
Milan, Italy (home of gelato and chocolates)
E. Schiff, F. Poordad, I. Jacobson, S. Flamm, B. Bacon, E. Lawitz, S. Gordon, J. McHutchison, R. Ghalib, T. Poynard, M. Sulkowski, C. Trepo, M. Rizzetto, S. Zeuzem, P. Marcellin , P. Mendez, C. Brass, J. Albrecht
Aims of the Study
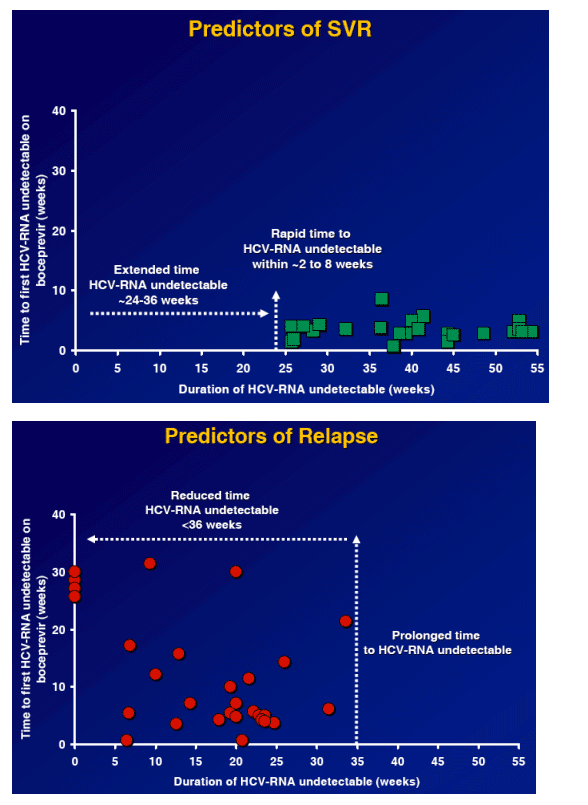
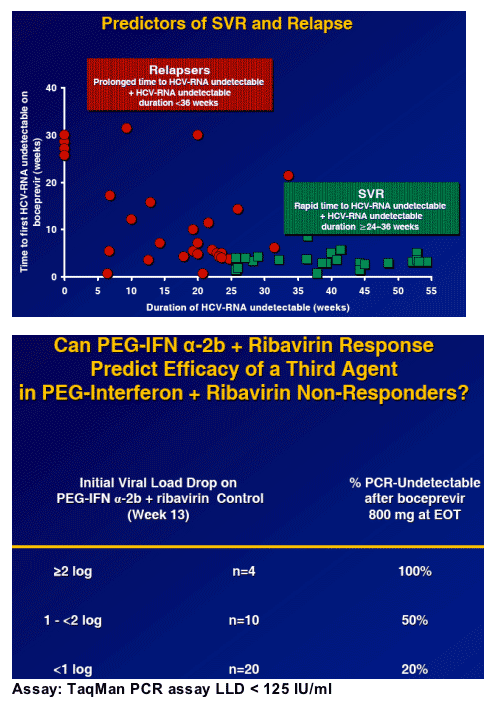
To evaluate the most effective boceprevir dose in non-responders
To evaluate whether ribavirin is required to enhance the efficacy of the regimen
To evaluate the most effective treatment duration in non-responders
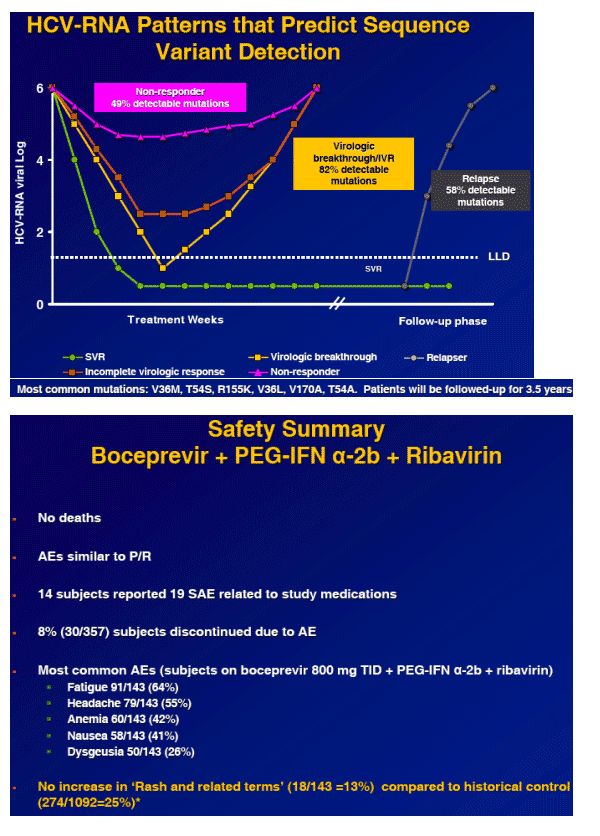
To evaluate the safety profile of boceprevir
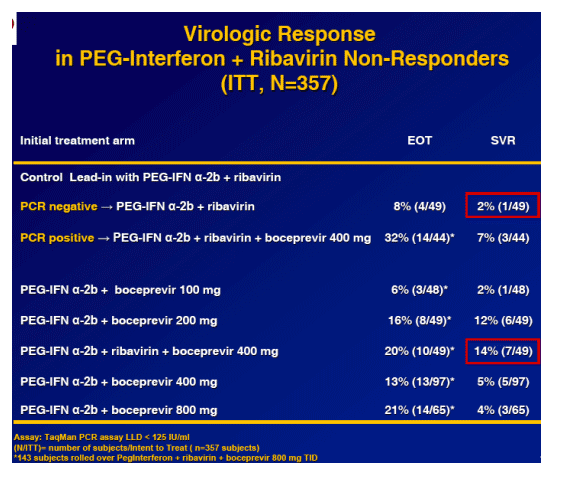
Patient Population
HCV-1 non-cirrhotic
Protocol defined criteria for treatment failures:
Never HCV-RNA (PCR) negative--
Previous treatment duration 12 weeks (Null-responders):
- <2 log10 drop in HCV-RNA vs. baseline at treatment week 12
- No Dose reductions and/or interruptions
Previous treatment duration >12 weeks (Non-responders):
- ≥80% of the assigned doses (PEG-Interferon + ribavirin)
- ≥80% of the treatment duration


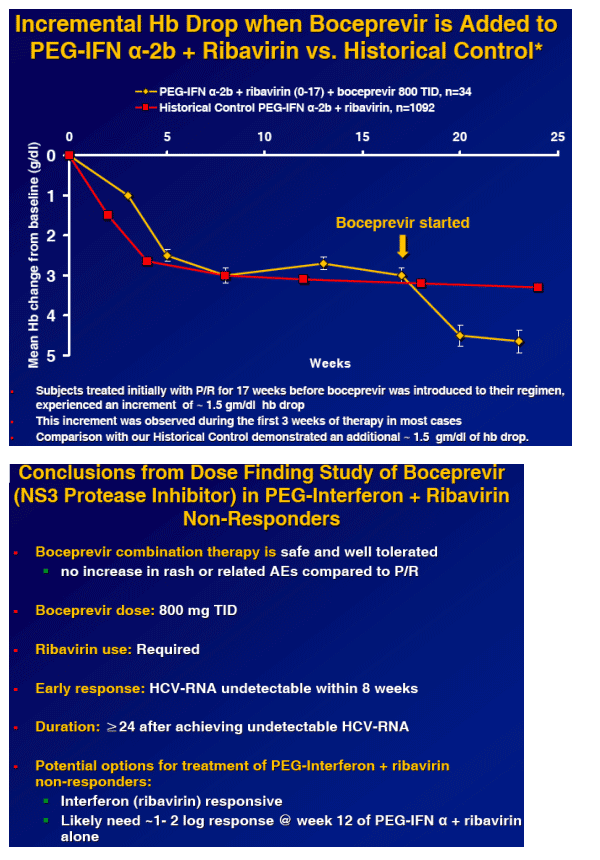
|
| |
|
 |
 |
|
|