 |
 |
 |
| |
Long Term (3 Years) Efficacy, Safety and Resistance Analyses of Entecavir (ETV) Treatment in Japanese Nucleoside-naive Patients with Chronic Hepatitis B (CHB)
|
| |
| |
Reported by Jules Levin
43rd EASL April 23-27, 2008
Milan, Italy
S Mochida1, K Takaguchi2, O Yokosuka3, S Fujioka4, N Hayashi5, M Shindo6, K Chayama7, J Toyota8, H Ishikawa9, T Seriu9 and M Omata10
1Division of Gastroenterology and Hepatology, Department of Internal Medicine, Saitama Medical University, Saitama; 2Department of Internal Medicine, Kagawa Prefectural Central Hospital, Kagawa; 3Department of Medicine and Clinical Oncology, Graduate School of Medicine, Chiba University, Chiba; 4Department of Internal Medicine,
Okayama Saiseikai General Hospital, Okayama; 5Department of Gastroenterology and Hepatology, Osaka University Graduate School of Medicine, Osaka; 6Division of Liver Disease, Department of Internal Medicine, Akashi Municipal Hospital, Hyogo; 7Department of Medicine and Molecular Science, Graduate School of Biomedical Sciences,
Hiroshima University, Hiroshima; 8Department of Hepatology, Sapporo Kosei General Hospital, Hokkaido; 9Research and Development, Bristol-Myers Squibb Japan, Tokyo; 10Department of Gastroenterology, Graduate School of Medicine, University of Tokyo
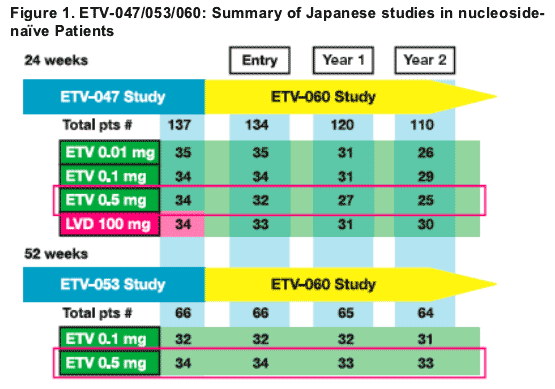
Introduction: ETV-047/053/060: Japanese nucleoside-naive studies
The goal of chronic hepatitis B (CHB) treatment is to achieve sustained suppression of HBV DNA and remission of liver disease
Entecavir 0.5 mg daily for 48 weeks resulted in high rates of HBV DNA suppression, ALT normalisation and histological improvement in study ETV-053 in Japan1
Entecavir 0.5 mg daily for 24 weeks demonstrated superior HBV DNA reduction compared to lamivudine (LVD) 100 mg daily in study ETV-047 in Japan2
Patients from studies ETV-053 or -047 could enroll in rollover study ETV-060
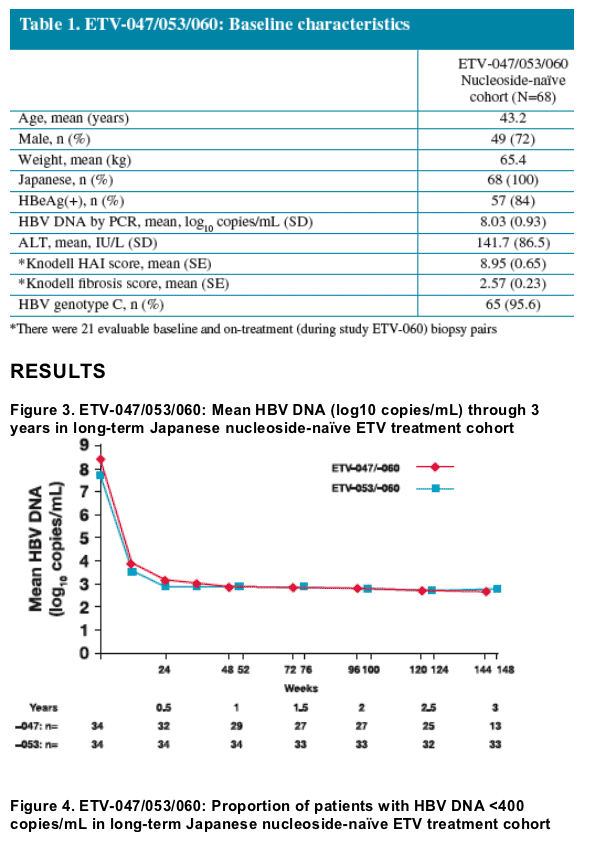
We present long-term efficacy, safety and resistance results of nucleoside-naive CHB patients who received up to 3 years of treatment with entecavir
AUTHOR CONCLUSIONS
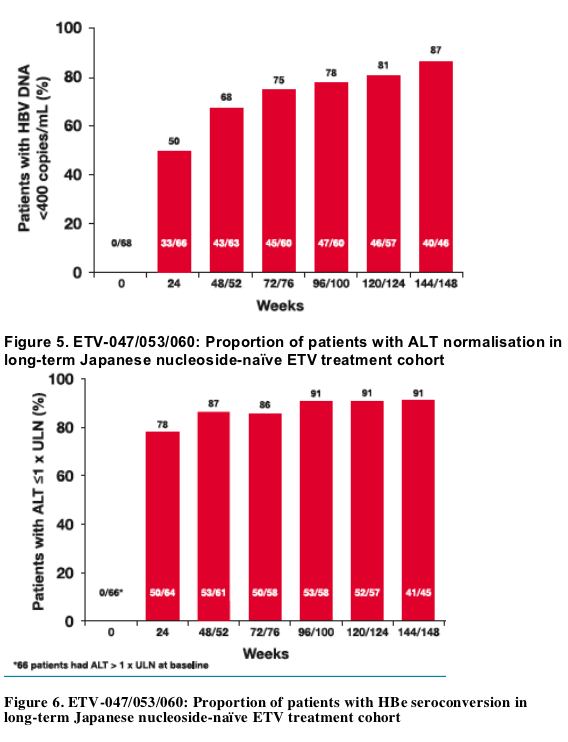
87% of nucleoside-naive patients treated continuously with entecavir 0.5 mg daily for 3 years achieved HBV DNA <400 copies/mL
Continuous treatment through 3 years also resulted in ALT normalisation in 91% of patients
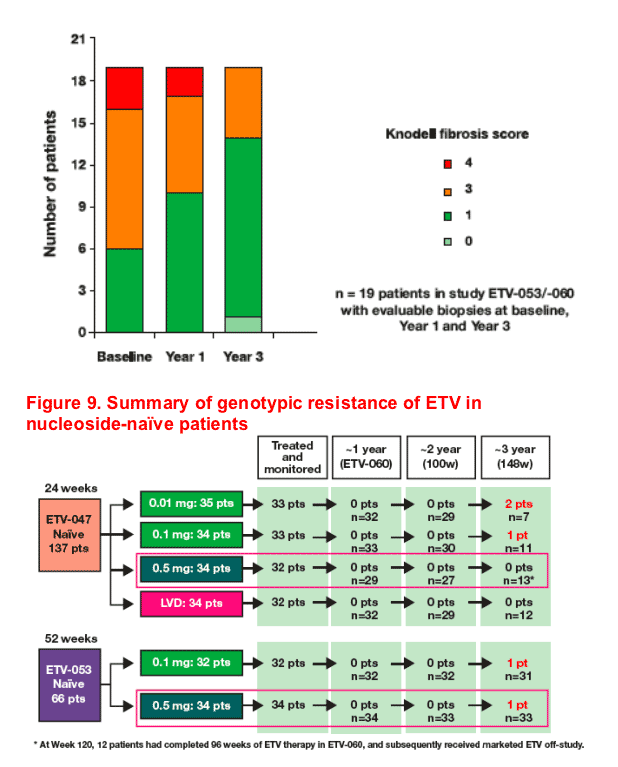
Repeated histological assessment over 3 years demonstrated improvement in necroinflammation in 100% of patients and improvement in fibrosis in 63% of patients
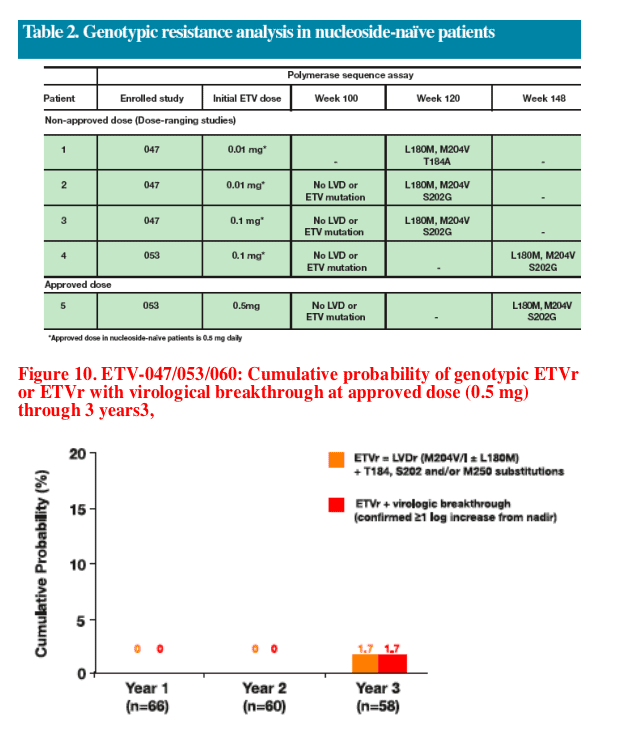
Genotypic analysis demonstrated that five patients developed amino acid substitutions T184 or S202 (ETV genotypic resistance) in addition to L180+M204 (LVD genotypic resistance) at Week 120 or 148
The cumulative probability of entecavir resistance at the approved 0.5 mg dose through 3 years was 1.7%
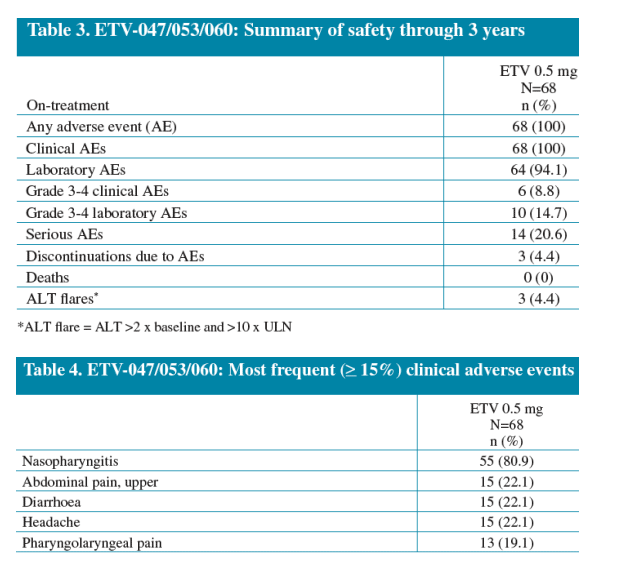
Entecavir was well tolerated through 3 years of treatment
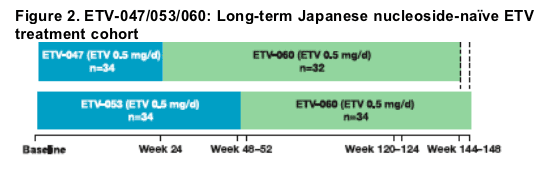
Sixty-eight patients from prior nucleoside-naive studies received entecavir 0.5 mg daily
Long-term cohort (N=66)
- ETV-047 to ETV-060: n=32
-- ETV-053 to ETV-060: n=34
ETV-047/053/060: Programme design
Eligibility criteria (studies ETV-047 and ETV-053)
- CHB infection with compensated liver disease
- HBV DNA >5 log10 copies/mL by PCR assay (>7.6 log10 copies/mL for ETV-047)
- <12 weeks prior treatment with anti-HBV nucleoside analogues
- ALT 1.25-10 x ULN
- HBeAg(+) or (-)
Study ETV-060
- Enrolment immediately after completion of ETV-047 or ETV-053 with no gap in dosing
- ETV 0.5 mg daily for a total treatment time of up to 148 weeks
Efficacy assessments through Week 148
- Mean change from baseline in HBV DNA by PCR
- Undetectable HBV DNA (<400 copies/mL) by PCR
- ALT <1.0 x ULN
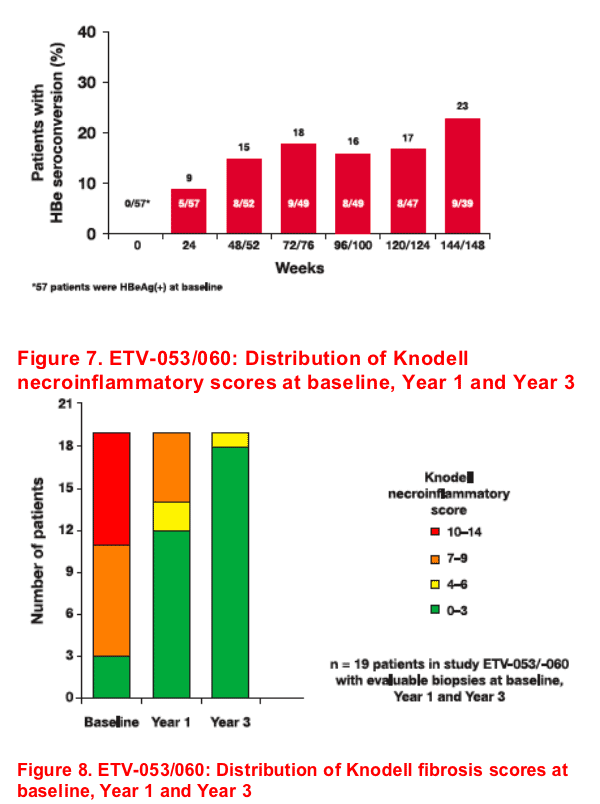
- HBe seroconversion
- Histological improvement by Knodell classification
- Resistance and virological breakthrough
Safety: Adverse events, discontinuations, deaths
References
1. Takaguchi K, et al. J Clin Virol. 2006;36(Suppl 2):S96.
2. Shindo M, et al. J Clin Virol. 2006;36(Suppl 2):S94.
3. Kobashi H, et al. Hepatology. 2007;46(Suppl 1):666A. Abstract 963.
4. Pawlotsky JM, et al. Gastroenterology. 2008;134:405-415.
|
| |
|
 |
 |
|
|