 |
 |
 |
| |
Three Years of Continuous Treatment in Chinese Patients who had Previously Failed Lamivudine: Results from Studies ETV-056 and ETV-050
|
| |
| |
Reported by Jules Levin
43rd EASL
April 23-27, 2008
Milan, Italy (land of gelato and chocolates)
GB Yao1, DZ Xu2, H Ren3, XQ Zhou4, JD Jia5, YM Wang6, CW Chen7, J Liu8, D Xu9 and C Llamoso9
1Shanghai Jing An Qu Central Hospital, Shanghai, China; 2Beijing Di Tan Hospital, Beijing, China; 3No. 2 Hospital Affiliated to Chongqing Medical University, Chongqing, China; 4Ruijin Hospital, Shanghai, China;
5Beijing Friendship Hospital, Beijing, China; 6Southwest Hospital Affiliated to 3rd Military Medical University, Chongqing, China; 7Shanghai Liver Disease Center of Nangjing Military Command, Shanghai, China;
8Bristol-Myers Squibb Company Research and Development, Braine-lŐAlleud, Belgium; 9Bristol-Myers Squibb Company Research and Development, Wallingford, CT, USA
AUTHOR CONCLUSIONS
LVD-refractory patients treated continuously with ETV 1 mg daily through 3 years achieved:
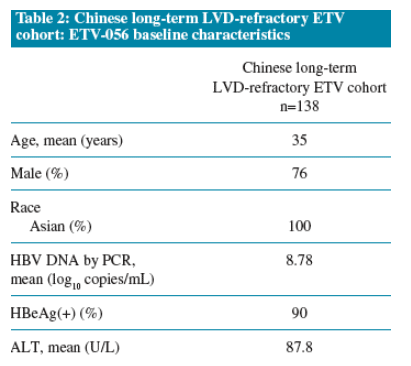
- HBV DNA suppression to less than 300 copies/mL, which increased from 30% at Week 48 to 55% at Week 144
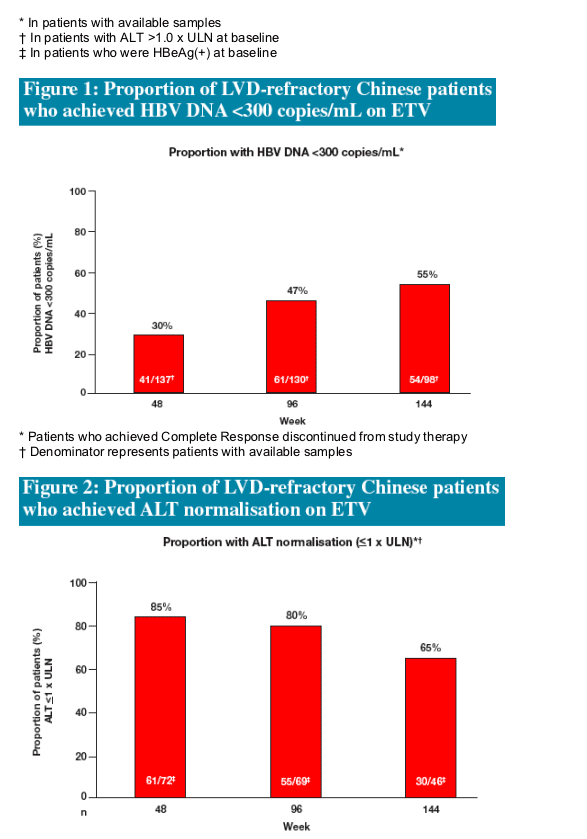
- ALT normalisation in 65% of patients
Long-term treatment with ETV was well tolerated, with a safety profile consistent with previously reported experience
Introduction
Lamivudine (LVD)-refractory Chinese patients with chronic hepatitis B (CHB) treated with entecavir (ETV) (1 mg daily) demonstrated marked HBV DNA reductions and normalisation of ALT in study ETV-0561
In ETV-056, patients were randomised to receive either ETV (1 mg daily) or placebo for 12 weeks, followed by 36 weeks of open-label ETV treatment1
All patients who completed the 36-week open-label phase of ETV-056 (treatment with ETV 1 mg daily) and who were assessed as likely to benefit from additional therapy were eligible to continue ETV treatment (1 mg daily) in rollover study ETV-050
We present efficacy and safety results for a cohort of LVD-refractory Chinese patients who received up to 3 years of treatment with ETV 1 mg daily in studies ETV-056 and -050
Chinese long-term LVD-refractory
ETV cohort
The Chinese long-term LVD-refractory ETV cohort consists of 138 patients initially treated in ETV-056 who:
- had previously failed LVD treatment and completed at least 36 weeks of ETV 1 mg daily in study ETV-056
-- enrolled in ETV-050 with a <35-day gap in treatment between the last dose in ETV-056 and the first dose in ETV-050 (None of the patients in this cohort had a treatment gap)

Patient management in ETV-050
Patients who achieved a Complete Response* during treatment stopped study drug and were followed off-treatment for 24 weeks
* Defined as HBV DNA <104 copies/mL by PCR and HBe seroconversion in patients who were HBeAg(+) at baseline; HBV DNA <104 copies/mL and ALT <1.25 x ULN in patients who were HBeAg(-) at baseline
By Week 144, 31 patients (22%) from this long-term LVD-refractory ETV cohort achieved a Complete Response and stopped ETV treatment
Chinese long-term LVD-refractory ETV
cohort: evaluations
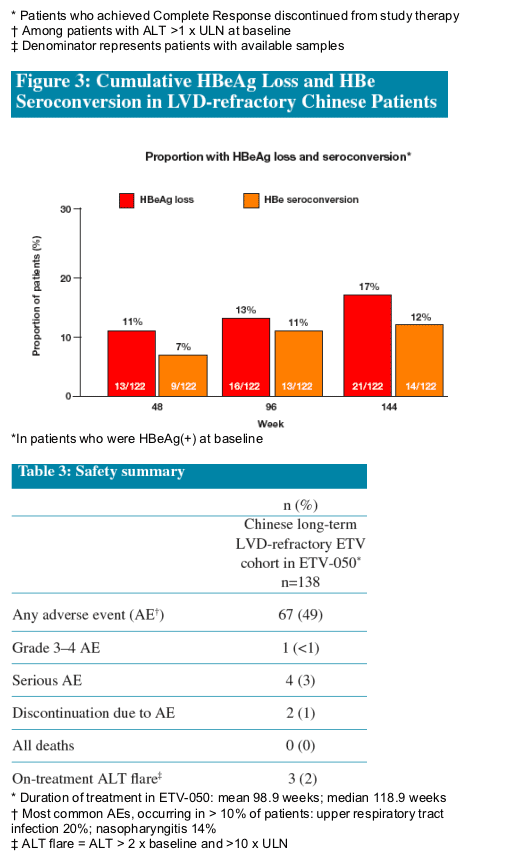
Proportion of the Chinese long-term LVD-refractory ETV cohort (n=138) with:
- HBV DNA <300 copies/mL*
- ALT normalisation (ALT <1 x ULN) *
- HBeAg lossŕ
- HBe seroconversion (loss of HBeAg, gain of anti-HBe)ŕ
Genotypic resistance testing was not performed in this study
References
1. Yao G, et al. Hepatol Int. 2007;3:373-381.
|
| |
|
 |
 |
|
|