 |
 |
 |
| |
Three Years of Continuous Treatment with Entecavir Results in High Proportions of Chinese Nucleoside-Naive Patients with Undetectable HBV DNA: Results from Studies ETV-023 and -050
|
| |
| |
Reported by Jules Levin
EASL Conference
April 23-27, 2008
Milan, Italy
G.B. Yao1, D.Z. Xu2, H. Ren3, X.Q. Zhou4, J.D. Jia5, Y.M. Wang6, C.W. Chen7, J. Liu8, D. Xu9, and C. Llamoso9
1Shanghai Jing An Qu Central Hospital, Shanghai, China; 2Beijing Di Tan Hospital, Beijing, China; 3No. 2 Hospital Affiliated to Chongqing Medical University, Chongqing, China; 4Ruijin Hospital, Shanghai, China; 5Beijing Friendship Hospital, Beijing, China;
6Southwest Hospital Affiliated to 3rd Military Medical University, Chongqing, China; 7Shanghai Liver Disease Center of Nangjing Military Command, Shanghai, China; 8Bristol-Myers Squibb Company Research and Development, Braine-l'Alleud, Belgium;
9Bristol-Myers Squibb Company Research and Development, Wallingford, CT, USA
AUTHOR CONCLUSIONS
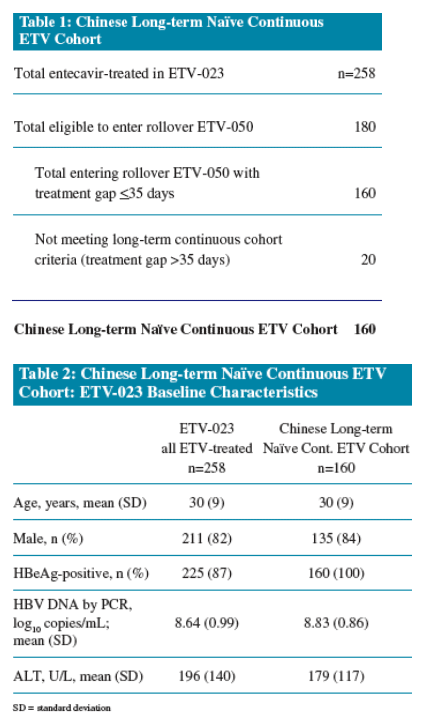
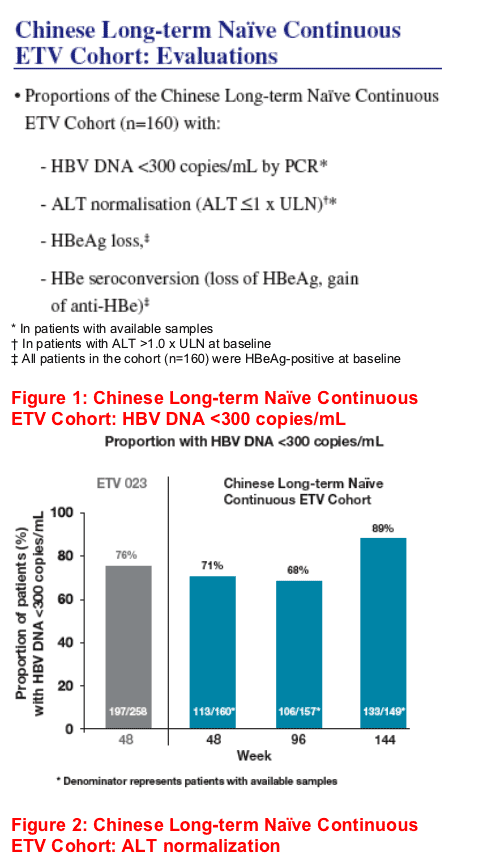
Eighty-nine percent of Chinese nucleoside-naive patients who received continuous treatment with entecavir through 3 years had HBV DNA <300 copies/mL at Week 144
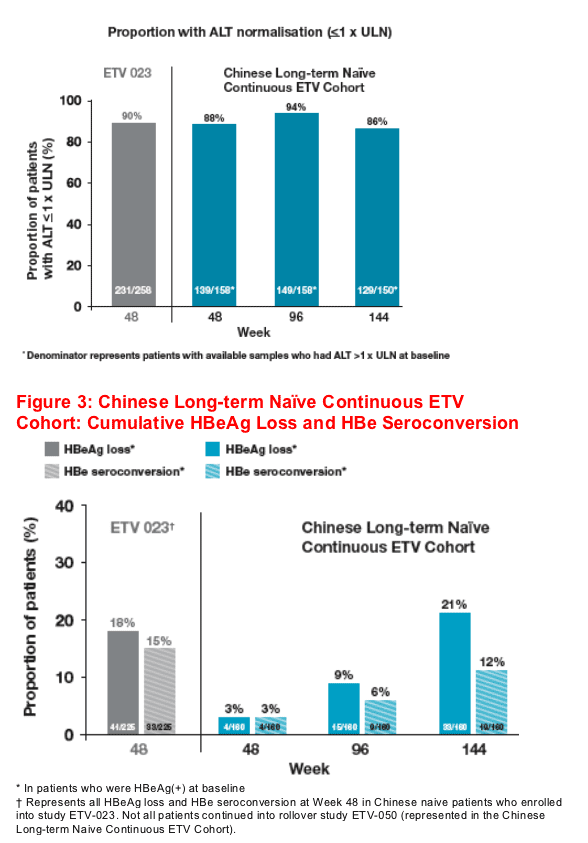
Continuous treatment during 3 years also resulted in maintenance of ALT normalisation: 86% at Week 144
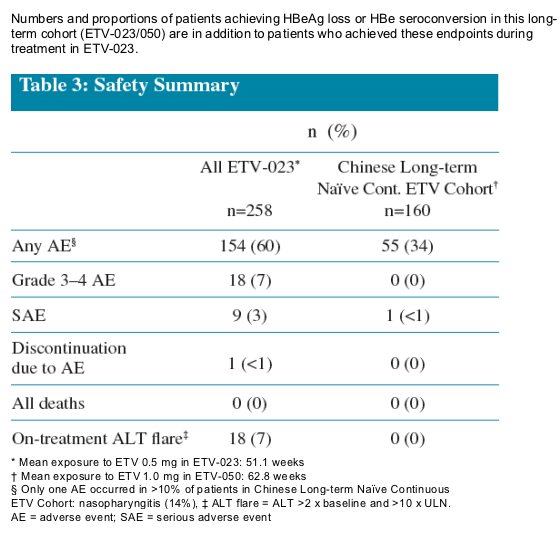
Continued treatment resulted in additional patients achieving HBeAg loss and HBe seroconversion
The results presented here are consistent with the results of long-term entecavir treatment of nucleoside-naive patients in global studies3
Safety profile for entecavir remained consistent with previously reported experience
Introduction
Entecavir (ETV) (0.5 mg, QD) was superior to lamivudine (100 mg QD) for virological and biochemical endpoints at Week 48 in nucleoside-naive, Chinese chronic hepatitis B patients in study ETV-0231
Patients treated with ETV for up to 96 weeks in study ETV-023 demonstrated continued benefit in terms of viral load reduction and alanine aminotransferase (ALT) normalisation2
Eligible patients based on virological and serological status could continue entecavir treatment in rollover study ETV-050 (entecavir 1.0 mg daily)
We present long-term efficacy and safety results in a cohort from study ETV-050 who received up to 3 years of ETV in studies ETV-023/-050
Chinese Long-term Naive Continuous
Entecavir (ETV) Cohort
The Chinese Long-term Naive ETV Cohort consists of patients initially treated with ETV 0.5 mg in ETV-023 who:
- Had a virological non-response after completing 52 weeks of blinded dosing, or
- Had not achieved consolidated response* after 96 weeks of blinded dosing, or
- Had a virological breakthrough during the second year of blinded dosing; AND
- Enrolled in ETV-050 with a <35-day gap in treatment between the last dose in ETV-023 and the first dose in ETV-050
* Consolidated response in ETV-023: HBV DNA < 0.7 MEq/mL by bDNA assay and HBeAg-negative for at least 24 weeks, and ALT < 1.25 x ULN, regardless of HBeAg status at baseline
References
1. Yao G, et al. Hepatol Int. 2007;1:365-372.
2. Yao G, et al. Hepatology 2006;44(suppl.1):559A-560A.
3. Han S, et al. Hepatology 2007;46(Suppl):654A. Abstract 938
|
| |
|
 |
 |
|
|