 |
 |
 |
| |
Genotypic analysis of HCV NS5B variants selected from patients treated with VCH-759, and Viral Load Declines
|
| |
| |
Reported by Jules Levin
43rd EASL
April 23-27, 2008
Milan, Italy
Olivier Nicolas, Isabel Boivin, Corinne St-Denis, Jean Bedard
ViroChem Pharma Inc, 275 Armand-Frappier Blvd., Laval, QC, Canada H7V
AUTHOR CONCLUSIONS
VCH-759 dosing produced a rapid decline in viral load of up to 2 log10 or greater. In a subset of patients, clinical breakthrough associated with resistant mutant variants was observed and several of these variants were found to persist at day 24 (2 weeks post-treatment) of the study.
The most frequent mutations observed by "population sequencing" and by "clonal sequence analysis" were at the amino acid position 423 (M423T/V/I) followed by positions 419 (L419M) and 482 (I482L).
All recombinant replicon mutants investigated were found to have a reduced susceptibility to VCH-759 and a reduced replication capacity when compared to the parental wild-type replicon.
This study illustrates that VCH-759 should be used in a combination therapy to maintain viral suppression and prevent emergence of resistance.
ABSTRACT
Background: VCH-759 is a novel orally bioavailable non-nucleoside inhibitor of hepatitis C virus RNA-dependent RNA polymerase. In a phase Ib clinical study involving genotype 1a and 1b-infected subjects, VCH-759 has achieved a 2 log10 or greater decline in HCV RNA levels. During the 10-day dosing period, an initial rapid reduction of viral load was observed in all patients dosed with VCH-759. Some patients have experienced a sustained anti-viral response during treatment but viral breakthrough was noticed for others. The aim of this study was to genotypically and phenotypically characterize the selection of resistant variants and to investigate a potential correlation with the viral kinetics observed.
Methods: Selected samples collected at day 1 (pre-dosing), day 11 (day after last dose), and days 17 and 24 (short term follow-up) were used for analysis. The entire NS5B gene was amplified by RT-PCR and directly sequenced. In order to determine the prevalence of each mutation within the population, the region corresponding to the compound binding pocket (amino acids 340-539) was also amplified, cloned, and numerous clones sequenced. The phenotypic and the in vitro replication capacity analysis of variants were performed using the HCV replicon system.
Results: Analysis of pre-dosing sera has revealed a wild-type genotype for the positions 419, 423, and 482 of the NS5B when compared to the consensus sequence of genotypes 1a and 1b. A mixture of wild-type and M423T/V/I substitution was the common feature found by population sequencing in samples at day 11, 17, and 24. The M423T and M423V mutants were associated with a respective 18 to 21-fold increase in replicon EC50 values when compared to the wild-type. A less predominant mutation namely, L419M was detected by clonal analysis and was found to confer a 23-fold reduction in VCH-759 susceptibility. Overall, the selected mutants were found to have a reduced replication capacity when compared to the wild-type.
Conclusions: HCV variants with mutations conferring resistance to VCH-759 were selected during the course of a 10-day treatment. This study illustrates that VCH-759 should be used in a combination therapy to maintain viral suppression and prevent emergence of resistance.
BACKGROUND
VCH-759 is a thiophene 2-carboxylic acid derivative which binds in the thumb region of HCV NS5B polymerase at a site located at approximately 35 Å from the catalytic domain. It is therefore a non-competitive allosteric inhibitor associated with an IC50 in a low micromolar range against the enzyme.
VCH-759 is equipotent against HCV genotype 1a and 1b replicons with a therapeutic index (CC50/EC50) > 500.
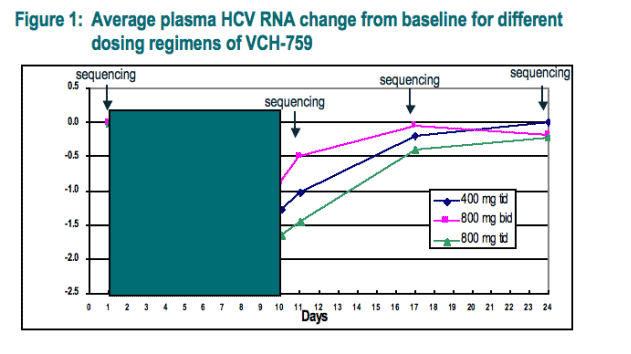
The antiviral activity, safety and tolerability of VCH-759 administered in monotherapy for 10 days to subjects infected with HCV genotype 1a or 1b was investigated. All patients who received VCH-759 among the three cohorts (400 mg tid, 800 mg tid, and 800 mg bid), had a sharp initial decline of approximately 1.5-log10 in plasma HCV-RNA concentration during the first two days of dosing (Fig. 1) and subsequent anti-viral responses could be divided into two major patterns: breakthrough or a sustained response.
OBJECTIVES
To characterize the in vivo selected resistant HCV variants to VCH-759 genotypically by "population sequencing" and by "clonal sequence analysis".
To characterize phenotypically the NS5B mutations found in the HCV variants by performing drug susceptibility testing of engineered HCV sub-genomic replicon genotype 1b cells and to investigate the in vitro replication capacity of selected mutants.
To investigate a potential correlation between the selection of resistant variants to VCH-759 with the viral kinetics (viral rebound) observed during the treatment.
METHODS
Population sequencing of VCH-759-resistant HCV variants: Plasma samples collected from baseline (day 1), days 11, 17, and 24 were analyzed and the HCV RNAs were isolated. cDNAs were synthesized using random primers and the SuperScript III reverse transcriptase (RT), and were subjected to amplifications by PCR using the Phusion High-fidelity DNA polymerase and combinations of upstream and downstream appropriate primers. Then, PCR products were analyzed by agarose gel electrophoresis and sent for sequencing.
Clonal analysis of VCH-759-resistant HCV variants (VCH-759 binding pocket sequencing): Shorter PCR products of 599 bp, encompassing the VCH-759-binding pocket (amino acids 340-539) were generated, cloned using the Zero Blunt¨ TOPO¨ PCR cloning kit, and numerous bacterial clones were sequenced per patient and per time point.
Engineering of recombinant replicons: All mutations were introduced into the pFKI389/NS3-3'/adapt vector obtained from Dr. R. Bartenschlager (Reblikon GmbH, Germany) by PCR-based site-directed mutagenesis and standard recombinant DNA technologies. In vitro transcripts were generated, and stable replicon cell lines were obtained upon selection with G418.
Drug susceptibility evaluation: The inhibitory effect of VCH-759 against wild type and recombinant replicon cells was determined by monitoring the levels of HCV RNA normalized to the 18S ribosomal RNA by quantitative real time PCR.
Replication capacity of HCV recombinant variants: Recombinant replicons were produced in the luciferase transient replicon system and RNAs were transfected in Huh7-derived cells side by side with the wild type replicon RNA. Cells were plated in 96-well plates, and luciferase activity was measured 96h post transfection using a luminometer. Transfection efficiency was normalized at 4h after electroporation and compared to the wild type replicon RNA.
RESULTS
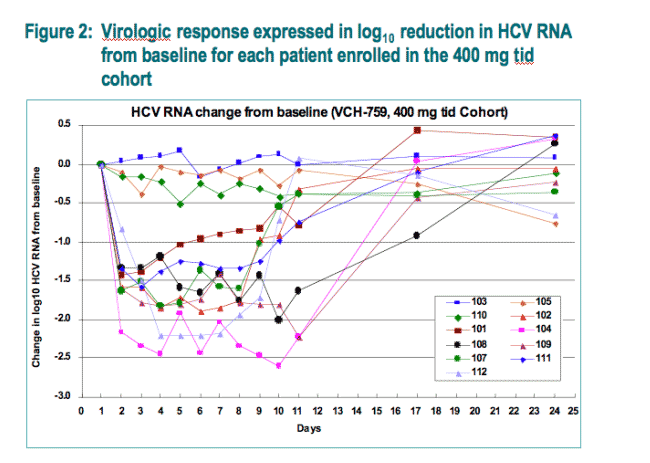
Viral response pattern to VCH-759: All patients who received VCH-759 among the three cohorts (400 mg tid, 800 mg tid, and 800 mg bid), had a sharp initial decline of approximately 1.5-log10 in plasma HCV-RNA concentration during the first two days of dosing (Fig. 1). Viral responses could be divided into two major patterns: breakthrough before the end of treatment (> 0.75 log10 IU/mL from nadir) or a sustained response (< 0.75 log10 IU/mL from nadir between day 1 and day 11) (Fig. 2, 400 mg tid cohort).
Population sequencing of NS5B: In addition to amino acids changes at position L419, M423, I482, several other amino acid changes were found only sporadically. The most prevalent mutation was a substitution found at M423 (data not shown).
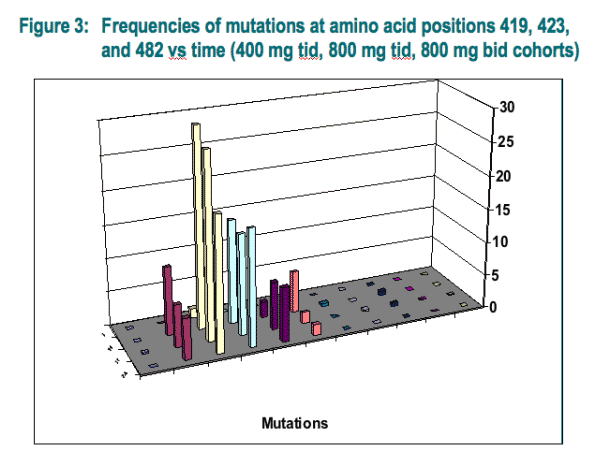
Clonal analysis of the VCH-759 drug binding pocket: An increase of amino acid change up to 8.4%, 44.1%, and 5.4% from baseline was found for positions 419, 423, and 482 respectively (Fig. 3). Double mutants were also observed but at a low frequency (0.2 - 0.6 %) (Fig. 3). In some patients who experienced a sustained viral response, genotypic analysis revealed a wild-type virus throughout the study (Fig. 4A). However, in a subset of patients experiencing a viral breakthrough, genotypic analysis has confirmed the presence of several variants which were found to be replaced by wild-type virus (Fig. 4B) or to remain predominant and persist at day 24 of the study (Fig. 4C).
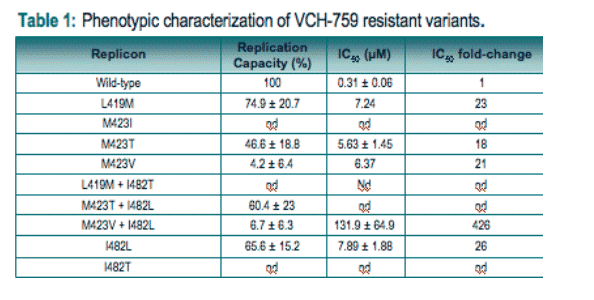
Drug susceptibility and replication capacity studies: A 18 to 26-fold increase of the compound's IC50 was recorded for the replicons harboring the L419M, M423V, I482L, and M423T single mutations whereas a 426-fold reduction in susceptibility (IC50) to the compound was noticed for the replicon harboring the M423V/I482L double mutation vs wild-type (Table 1). All mutants were associated with a reduced replication capacity when compared to the parental wild-type replicon in the absence of VCH-759 (Table 1).
|
| |
|
 |
 |
|
|