 |
 |
 |
| |
POTENT ANTIVIRAL ACTIVITY OF THE HCV NUCLEOSIDE POLYMERASE INHIBITOR, R7128, IN COMBINATION WITH PEG-IFN a-2a AND RIBAVIRIN
|
| |
| |
Reported by Jules Levin
43rd Annual Meeting of EASL, Milan, Italy, April 23-27, 2008
J. Lalezari, E. Gane, M. Rodriguez-Torres, E. DeJesus, D. Nelson, G. Everson, I. Jacobson, R. Reddy, G.Z. Hill, A. Beard, W.T. Symonds, M.M. Berrey, and J.G. McHutchison
AUTHOR CONCLUSIONS
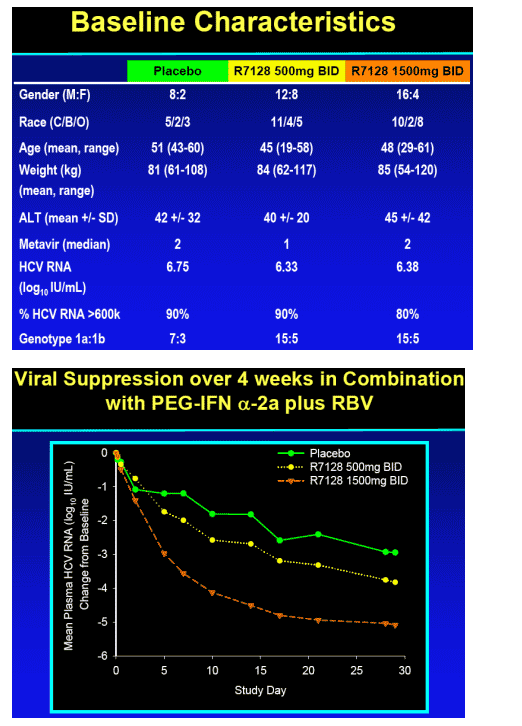
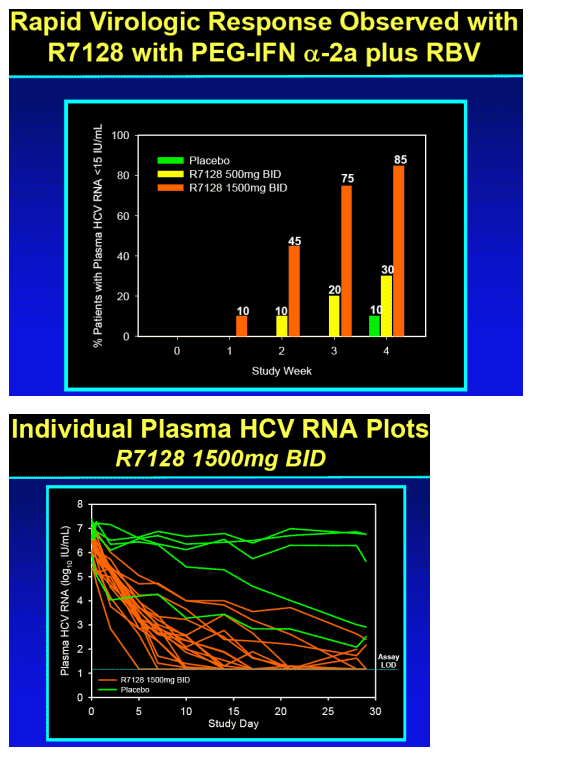
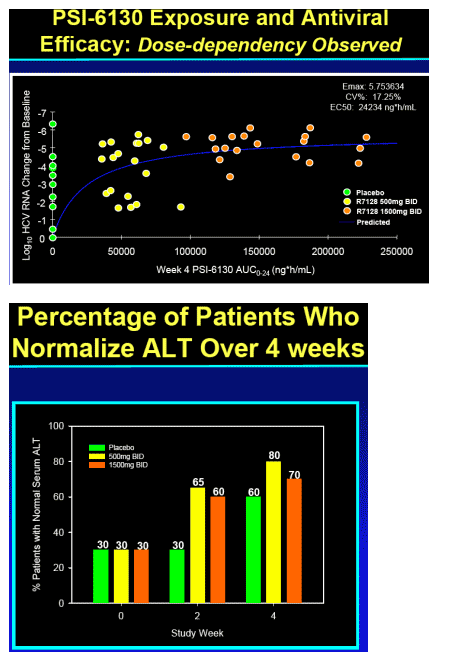
Significant antiviral effect of R7128 in combination with PEG-IFN alpha-2a and RBV over 4 weeks
-- 85% patients with undetectable HCV RNA by week 4 (R7128 1500 mg BID + PEG-IFN_-2a/RBV)
-- 5.1 log10 mean decrease in HCV RNA
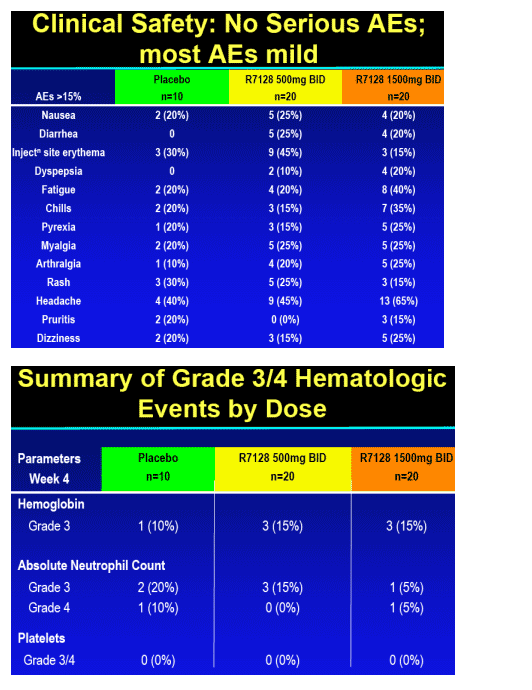
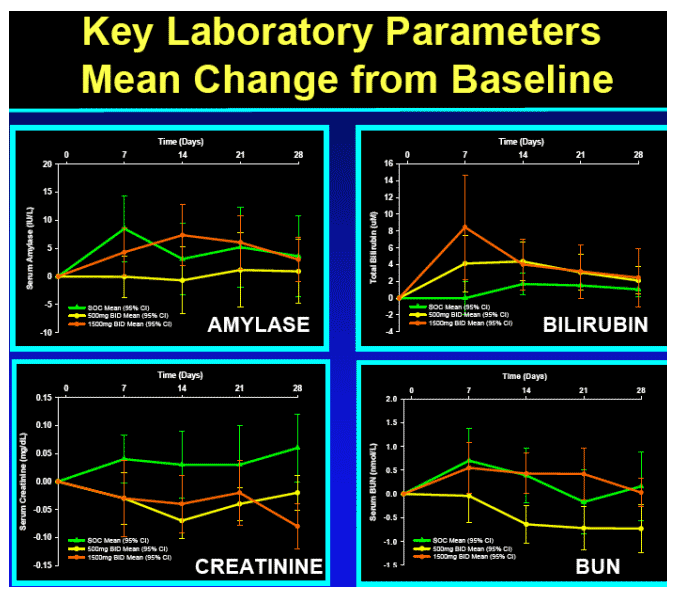
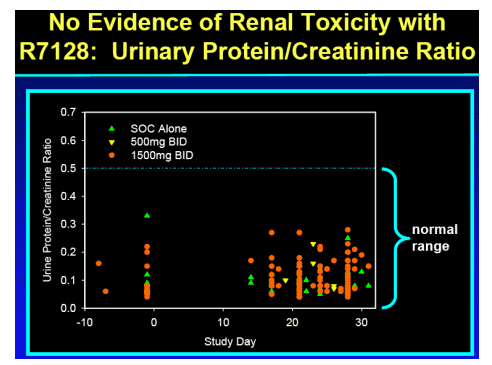
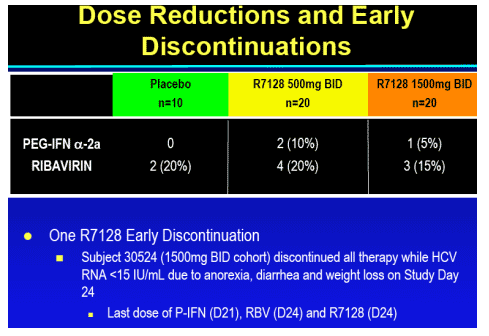
R7128 safe and well tolerated over the 4 weeks:
-- no substantial differences from expected events observed with PEG-IFN_-2a/RBV
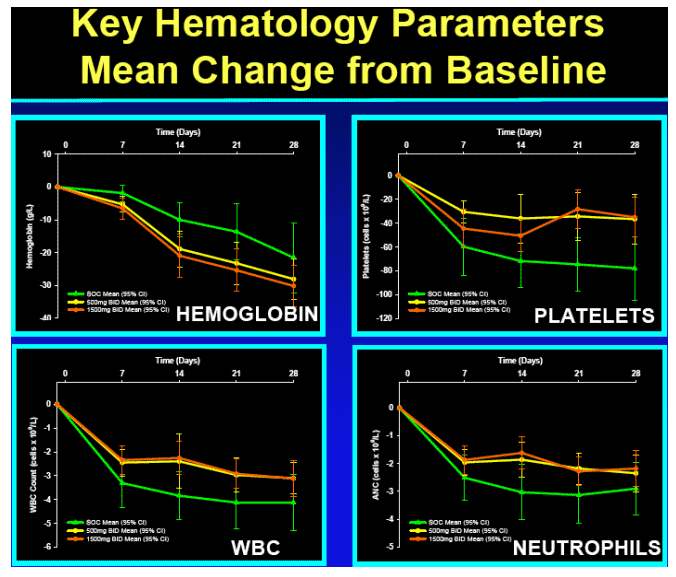
-- No hematologic or other toxicity identified
R7128: Next steps
Two additional cohorts will be enrolled in the current 4-week triple combination study
-- R7128 1000 mg BID + PEG-IFN_-2a/RBV in GT-1a/b Rx-naive
-- R7128 1500 mg BID + PEG-IFN_-2a/RBV in GT-2/3 prior non-responders
International phase 2b study to evaluate R7128 in triple combination for up to 12 weeks
-- R7128 at doses of 1000 and 1500 mg BID
-- PEG-IFN_-2a and RBV
R7128: Specific Nucleoside Polymerase
Inhibitor of HCV
R7128 is a prodrug of PSI-6130 and PSI-6206
Mode of action via chain termination
Completed single ascending oral dose studies in healthy volunteers for PSI-6130 and R7128
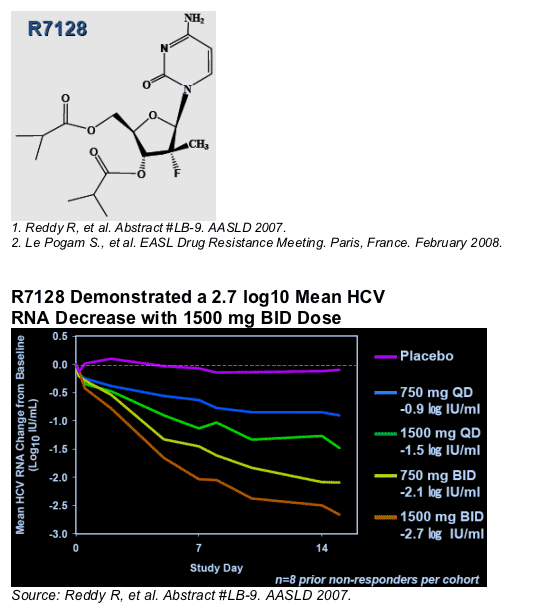
Completed 14-day multiple ascending oral dose study in HCV patients who failed interferon
-- 2.7 log10 (>99%) Mean HCV RNA decrease with 1500mg twice-daily (BID) dose1
-- No maximum tolerated dose identified
-- No evidence of viral resistance detected to date2
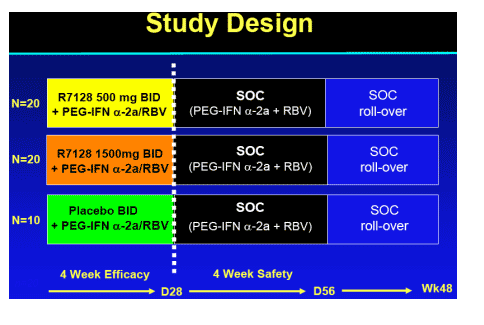
Study Objectives and Design
Objective
-- Evaluate safety/tolerability, antiviral activity and viral resistance
Design
-- Multi-center, randomized, double-blind (R7128)
-- 4-week combination with peginterferon _-2a (PEGASYS)+ RBV (COPEGUS) with follow-up safety data on SOC to day 56
-- Patient population
Treatment-naive, genotype 1
Non cirrhotic (Metavir ≦ 2, Knodell ≦ 3, Ishak ≦ 4)
--HCV RNA Assay: COBAS AmpliPrep/COBAS TaqMan HCV Test, RUO Version (lower limit of detection 15 IU/mL)
-- Samples for HCV NS5B resistance testing collected before, during and after the treatment period.
|
| |
|
 |
 |
|
|