 |
 |
 |
| |
Efficacy and safety of peginterferon alfa-2a (40KD) in Chinese patients with HBeAg-positive chronic hepatitis B
|
| |
| |
Reported by Jules Levin
43rd EASL, April 2008, Milan Italy
Dao-zhen Xu1, Yao Xie2, Lai Wei3, Xin'yue Chen4, Mo'bing Wan5, Yu'ming Wang6, Lei Wang7, Jia Wei8, Jian'he Gan9, De'ying Tian10 Shu'lin Zhang11
1Infectious Disease Department, Beijing Ditan Hospital, Beijing, China; 2Infectious Disease Department, Beijing Ditan Hospital, Beijing, China; 3Hepatology Department, Beijing People Hospital, Beijing, China; 4Infectious Disease Department, Beijing Youan Hospital, Beijing, China;
5Infectious Disease Department, Changhai Hospital, Shanghai, China; 6Infectious Disease Department, Xinan Hospital, Chongqin, China; 7Hepatology Department, Jinan Infectious Disease Hospital, Jinan, China; 8Infectious Disease Department, Suzhou University Hospital, Suzhou, China;
9Infectious Disease Department, Suzhou University Hospital, Suzhou, China; 10Infectious Disease Department, Tonji Medical College Hospital, Huazhong University of Science and Technology, Wuhan, China; 11Infectious Disease Department, Xian Jiaotang University Hospital, Xian, China
This research was partly funded by Roche, Basel, Switzerland
STUDY AIM
To evaluate the efficacy and safety of peginterferon alfa-2a (PEGASYS) for 48 weeks in Chinese patients who were either treatment-naive or failures with IFN or LAM
INTRODUCTION
· Currently available treatments for chronic hepatitis B include conventional interferon (IFN) alfa-2a, nucleos(t)ide analogues (NAs), and peginterferon alfa-2a. Both IFN and NAs have low HBeAg seroconversion rates and high relapse rates during therapy and/or after stopping therapy. Few data are available on response to therapy in IFN and NA pre-treated patients
· Peginterferon has proved to be more efficacious than IFN in treatment-naive patients with HBeAg-positive chronic hepatitis B.1 Moreover, a 48-week course of peginterferon alfa-2a was shown to be significantly more effective than lamivudine (LAM) in patients with HBeAg-positive and HBeAg-negative CHB2,3
· Few data are available on response to peginterferon alfa-2a in patients who have failed IFN and LAM therapy, but small studies have suggested that peginterferon is just as effective in treatment-naive patients as it is in those with prior LAM therapy4,5
· We therefore evaluated the efficacy and safety of peginterferon alfa-2a in treatment-naive, IFN pre-treated and nucleos(t)ide analogue pre-treated Chinese patients with HBeAg-positive CHB
AUTHOR SUMMARY
· There were no statistically significant differences in response rates between treatment-naive Chinese patients and those who had failed with IFN or LAM for any of the study endpoints
· Among patients with available data at week 72, rates of HBeAg seroconversion were similar to those observed in previous studies:
- 39%, 26% and 31% in Groups A, B and C, respectively
· Peginterferon alfa-2a was well tolerated. The adverse events rates were similar in groups A, B and C, respectively, and no unexpected
adverse events were reported
AUTHORCONCLUSION
· After 48 weeks of peginterferon alfa-2a treatment, the HBeAg seroconversion rate observed 24 weeks post-treatment in treatment-naive Chinese patients, was consistent with the result of a previously reported Phase 3 trial
· There were no significant differences in HBeAg seroconversion or HBV DNA response rate between HBeAg-positive CHB patients who were treatment-naive and those who had failed with previous treatments
· The percentage of patients with HBeAg seroconversion increased during the treatment-free follow-up period
METHODS
· This was a multi-centre study of Chinese patients who were either naive or failures (relapsers/non-responders) with conventional IFN or LAM
- Patients who relapsed on conventional IFN were those who received 3-5 MIU IFN tid for 4-6 weeks and had an end of treatment response (HBV DNA <500,000 copies/ml or HBeAg seroconversion) but tested positive for HBV DNA or HBeAg after 24 weeks' follow-up.
Non-responders were defined as patients who had no change from baseline in HBV DNA levels or HBeAg during the treatment period and follow-up
- Patients who relapsed on lamivudine were those who received 100 mg qd LAM for at least 1 year and had an end of treatment response but tested positive for HBV DNA or HBeAg after 24 weeks' follow-up. Lamivudine non-responders are defined as those with no change from baseline in HBV DNA levels or HBeAg during the treatment and follow-up periods
· 307 patients were enrolled at 46 study centres across China over a period of 23 months. Patients were aged 18+, had been HBeAg-positive, HBsAg-positive and anti-HBs negative for at least 6 months and had serum HBV DNA >500,000 copies/mL and ALT >1 x upper limit of normal (ULN) and <10 x ULN
· Patients were stratified according to their previous treatment history (Figure 1)
· All patients received peginterferon alfa-2a 180 _g qw for 48 weeks
· Response was measured 24 weeks after end of treatment. Primary endpoints were HBeAg seroconversion and HBV DNA suppression to <105 copies/mL. Secondary endpoints included HBV DNA <500 copies/mL; HBeAg clearance; HBsAg clearance and presence of anti-HBs; ALT normalisation; and a combined endpoint of HBeAg clearance, HBV DNA <100,000 copies/mL and ALT normalisation and were determined at weeks 48 and 72
· Patients were evaluable for per protocol analysis if they satisfied all inclusion criteria and received at least 4 weeks of treatment
- 16, 18 and 16 patients in Groups A, B and C, respectively, were excluded from the per protocol population
- Results shown are for patients with available data at weeks 48 and 72
· Assays of HBeAg, anti-HBe, HBsAg and anti-HBs were conducted using the AXSYM automated enzyme immunoassay system (Abbott Laboratories, USA). HBV DNA levels were determined by PCR (ShenZhen PG Biotech)
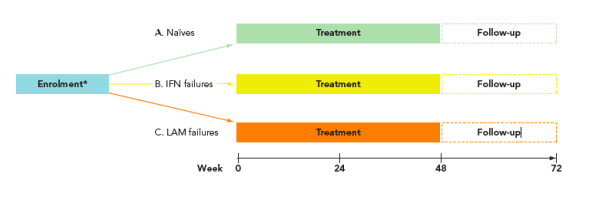
* Eligible patients were; HBeAg+; HBsAg+; HBV DNA >105 cp/mL; ALT >1 x ULN <10 x ULN (determined by two abnormal values taken >14 days apart, the latter taken within 35 days of the first dose of study drug and the second no more than 5 months before that)
RESULTS
Patient disposition
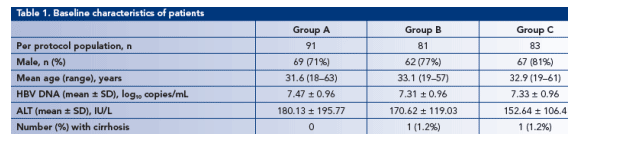
· Baseline patient characteristics in each of the three groups in the per protocol population are shown in Table 1
· The safety population included all patients who received at least one dose of peginterferon alfa-2a (n=305)
Efficacy results
Efficacy analysis was based on the per protocol population
Primary endpoints
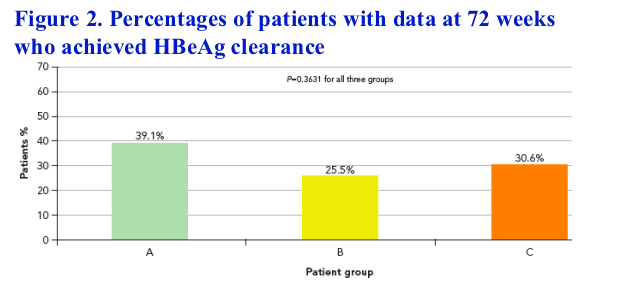
Among patients with data available at 72 weeks, HBeAg seroconversion rates were 39.1%, 25.5% and 30.6% for groups A, B and C, respectively (P=0.3631) (Figure 2). 63.0%, 57.5% and 42.8% of patients, respectively, had HBV DNA <105 copies/mL
Secondary endpoints
At week 72, there were no statistically significant differences in response rates in patients with data across all 3 groups (Figure 3A-C)
1 patient achieved HBsAg seroconversion
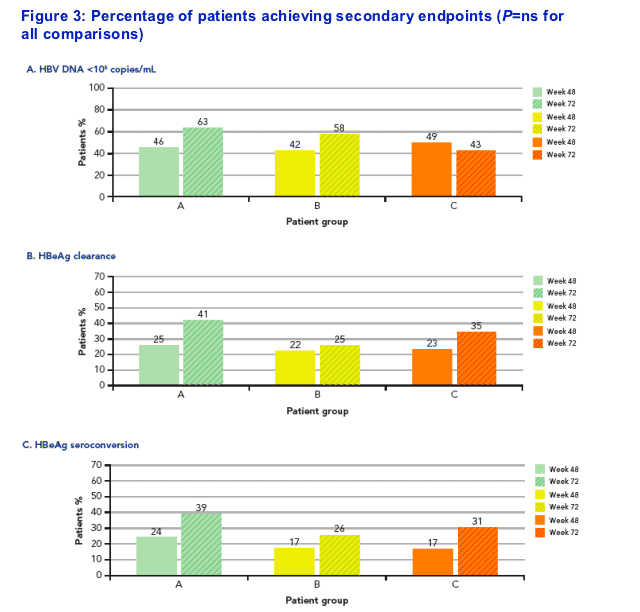
Group A: n=79 at wk 48, n=46 at wk 72; Group B: n=69 at wk 48, n=47 at wk 72; Group C: n=71 at wk 48, n=49 at wk 72
Safety results
Tolerability
· Peginterferon alfa-2a was well tolerated, with 94%, 90% and 93% of patients in Groups A, B and C, respectively, completing the 48-week study period
· The numbers of patients in each group requiring dose modification were very similar, 25.3%, 19.2% and 25.3% in groups A, B and C, respectively
Adverse events
· The overall incidence of adverse events in the safety population did not differ between the three groups and the most frequent symptoms in all groups were those generally associated with IFN therapy (ie 'flu-like' symptoms such as pyrexia, myalgia and fatigue)
· Only three serious adverse events requiring hospitalisation and discontinuation of the study drug occurred during the treatment period, one in each group. All resolved without sequelae
· A further four patients discontinued treatment due to clinical adverse events, two in Group A and one each in Groups B and C
Laboratory abnormalities
· The most frequent laboratory abnormalities were observed for leukocyte, platelet and haematocrit counts
· There were no statistically significant differences in the frequency of laboratory abnormalities between the three groups
REFERENCES
1. Cooksley WGF et al. Peginterferon alpha-2a (40 kDa): an advance in the treatment of hepatitis B e antigen-positive chronic hepatitis B. J Viral Hepatitis 2003;10:298-305.
2. Marcellin P et al. Peginterferon alfa-2a alone, lamivudine alone, and the two in combination in patients with HBeAg-negative chronic hepatitis B. N Engl J Med 2004;351:1206-17.
3. Lau GKK et al. Peginterferon alfa-2a, lamivudine, and the combination for HBeAg-positive chronic hepatitis B. New Engl J Med 2005;352(26):2682-95.
4. Marcellin P et al. Efficacy and safety of peginterferon alfa-2a (40KD) (PEGASYS) in patients with chronic hepatitis B who had received prior
treatment with nucleos(t)ide analogues: the PEGaLAM cohort. EASL 2006. Abstract 502.
5. Shi X.F. et al. Peginterferon alfa-2a for patients with HBeAg-positive CHB who failed prior lamivudine treatment. APASL 2007. Abstract.
|
| |
|
 |
 |
|
|