 |
 |
 |
| |
EVALUATION OF A 4 WEEK LEAD-IN PHASE WITH NITAZOXANIDE PRIOR TO NITAZOXANIDE+PEGINTERFERON IN TREATING CHRONIC HEPATITIS C
|
| |
| |
Reported by Jules Levin
43rd EASL, April 200, Milan Italy
Jean-Francois Rossignol1,2, Asem Elfert3, Emmet B. Keefe1,2
1The Romark Institute for Medical Research, Tampa, FL, USA, 2Division of Gastroenterology and Hepatology, Department of Medicine, Stanford University School of Medicine, Stanford, California; 3Department of Gastroenterology and Hepatology, University of Tanta, Faculty of Medicine, Tanta, Egypt
AUTHOR SUMMARY
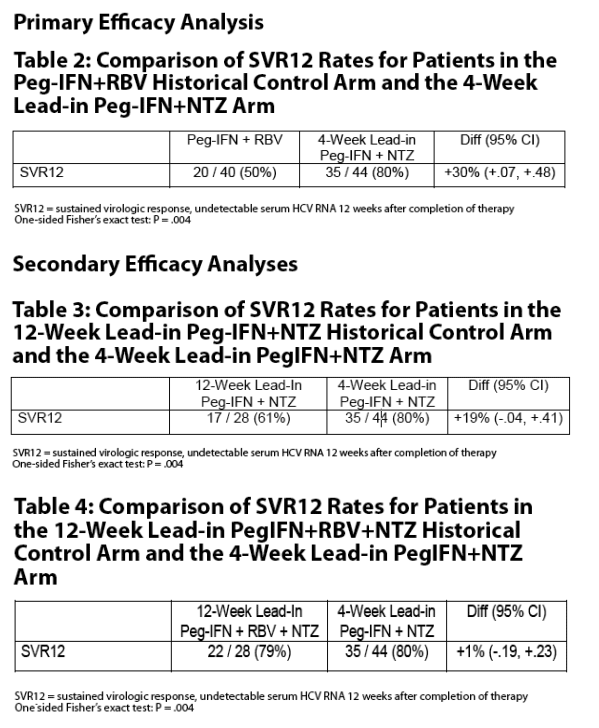
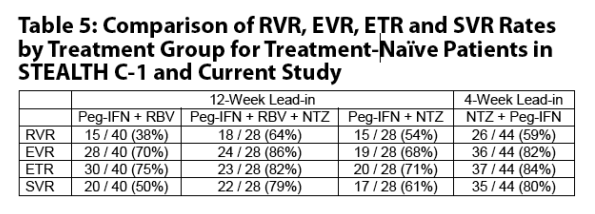
The nitazoxanide lead-in phase of 12 weeks used in the STEALTH C-1 trial can be reduced to 4 weeks without compromising RVR, EVR and ETR rates.
SVR rates using triple therapy with peginterferon alfa-2a plus ribavirin plus nitazoxanide (79% in STEALTH C-1) may not be different from SVR rates using combination peginterferon alfa-2a plus nitazoxanide (61% in STEALTH C-1 and 80% in current study).
AUTHOR CONCLUSIONS
The nitazoxanide lead-in phase in future studies of chronic hepatitis C with nitazoxanide added to peginterferon or peginterferon plus ribavirin can be reduced from 12 weeks to 4 weeks without compromising RVR, EVR and ETR rates.
Future studies comparing triple therapy with peginterferon plus ribavirin and nitazoxanide versus combination therapy with peginterferon and nitazoxanide are warranted.
INTRODUCTION
Nitazoxanide (Alinia, Romark Laboratories, Tampa, Florida) is licensed in the United States for the treatment of diarrhea caused by Cryptosporidium parvum and Giardia lamblia. The mechanism of action of nitazoxanide against anaerobic bacteria and protozoa is attributed to interference with anaerobic energy metabolism via pyruvate: ferredoxin oxidoreductase.
Nitazoxanide is also active against a range of viruses in cell culture models, and is currently in phase II clinical development for the treatment of chronic hepatitis C and chronic hepatitis B. The mechanism of action of nitazoxanide against viruses is different from its action against anaerobic bacteria and protozoa, and is related to enhancement of the intracellular activity of interferon by inducing eIF2alfa phosphorylation. In addition, recent laboratory studies suggest that nitazoxanide does not induce resistance.
In a randomized controlled trial (STEALTH C-1) in patients with chronic hepatitis C genotype 4, patients treated with nitazoxanide with a 12-week lead-in phase followed by 36 weeks of nitazoxanide and peginterferon alfa-2a plus ribavirin (SVR = 79%) or nitazoxanide and peginterferon alfa-2a (SVR = 61%) responded better than patients treated for 48 weeks with peginterferon alfa-2a plus ribavirin (SVR = 50%).
The rationale for a lead-in phase with nitazoxanide alone before combination therapy was based on early pilot studies (unpublished) that showed greater efficacy if nitazoxanide was administered before peginterferon plus ribavirin versus starting all three drugs at the same time. In addition, laboratory studies demonstrated that pretreatment of HCV replicon-containing cells with nitazoxanide potentiated subsequent treatment with nitazoxanide plus interferon.
OBJECTIVE
The primary objective was to evaluate the effect of reducing the duration of nitazoxanide lead-in therapy from 12 weeks to 4 weeks in patients with chronic hepatitis C treated with combination therapy with nitazoxanide plus peginterferon alfa-2a.
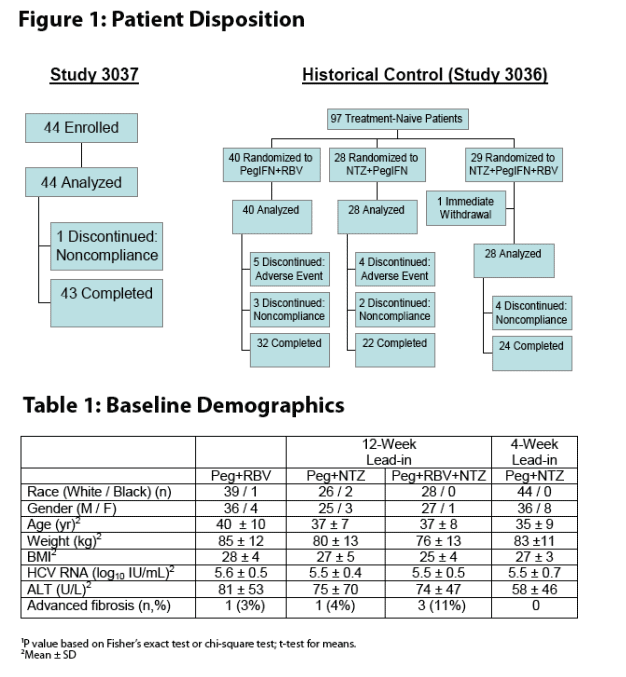
Study
44 treatment-naive patients with chronic hepatitis C were enrolled at the University of Tanta in Egypt in this open label study (RM01-3037)
Patients received nitazoxanide 500 mg twice daily with food for 4 weeks followed by nitazoxanide plus peginterferon alfa-2a, 180 μg/wk, for 36 weeks
Physical examination, hematologic and chemistry tests and serum HCV RNA (Abbott m2000, LLD 10 IU/mL) were performed every 4 weeks during treatment
Efficacy Parameters
Primary endpoint was SVR (serum HCV RNA <10 IU/mL 24 weeks following end of treatment)
The study was designed to use historical control data from the STEALTH C-1 trial (Peg-IFN+RBV versus Peg-IFN+RBV+NTZ versus Peg-IFN+NTZ)
Secondary endpoints included serum HCV RNA <10 IU/mL after 4 weeks (RVR) and 12 (EVR) weeks of combination therapy, and at end of treatment (ETR).
Analysis of data was by intent to treat.
Changes in Serum HCV RNA during Lead-in Phase
Adverse Events
12-week lead-in phase in STEALTH C-1 study: -0.26 log10 IU/mL
4-week lead-in phase in current study: -.0.25 log10 IU/mL
Adverse Events
Adverse events were similar to those seen in STEALTH C-1. Side effects were those expected with peginterferon therapy and were mild to moderate in severity. There were no dose reductions, drug discontinuations, or serious adverse events.
|
| |
|
 |
 |
|
|