 |
 |
 |
| |
SUSTAINED DURABILITY OF HBeAg SEROCONVERSION IN CHRONIC HEPATITIS B PATIENTS AFTER TREATMENT WITH TELBIVUDINE AND LAMIVUDINE
|
| |
| |
Reported by Jules Levin
EASL, April 2008, Milan, Italy
T. Poynard,1 J-L. Hou,2 A. Chutaputti,3 M. Manns,4 N. Naoumov5
1Groupe Hospitalier Pitie-Salpetriere, Paris, France; 2NanFang Hospital, First Medical University of the PLA, Guangzhou, China; 3Phramongkutklao Hospital, Bangkok, Thailand;
4Medizinische Hochschule Hannover, Hannover, Germany; 5Novartis Pharma AG, Basel, Switzerland
AUTHOR CONCLUSIONS
Telbivudine achieved a proportionally higher rate of greater HBeAg responses than lamivudine, with a greater proportion of HBeAg loss and seroconversion over a 2-year treatment period. A greater proportion of patients treated with telbivudine therefore qualified for treatment discontinuation due to HBeAg seroconversion, compared with lamivudine.
The post-treatment durability of HBeAg seroconversion with telbivudine was more than 85% by KaplanĐMeier estimate after 52 weeks of off-treatment follow-up, and was comparable to lamivudine.
These results suggest that most patients will have sustained off-treatment seroconversion after at least 6 months of treatment consolidation following HBeAg seroconversion with either telbivudine or lamivudine. These results also provide evidence to support the use of consolidation treatment to optimize durability of response and are in line with recent guideline recommendations for cessation of treatment in this patient group.4,5
Background
· HBeAg seroconversion is an important therapeutic milestone for patients with HBeAg-positive chronic hepatitis B (CHB).
· HBeAg seroconversion is indicative of good prognosis, including lower rates of cirrhosis and slower disease progression.1,2 Moreover, outcomes for patients who achieve HBeAg seroconversion are better if seroconversion is achieved earlier in the course of infection.3
· HBeAg seroconversion also is a clinically meaningful treatment goal because international treatment guidelines indicate that patients who achieve HBeAg seroconversion and who have a period of treatment (6Đ12 months) after seroconversion (ŇconsolidationÓ) and who have sustained disease remission after consolidation may be able to stop anti-hepatitis B virus (HBV) therapy.4,5 More data are needed, however, to ascertain durability of HBeAg seroconversion among therapies for CHB, including during off-treatment follow-up.
· Two large phase III trials comparing treatment with telbivudine and lamivudine over a 2-year period were conducted in adult patients with CHB. The GLOBE study (NV-02B-007, Ň007Ó), conducted in 20 countries, compared these two antiviral agents in 1367 (921 HBeAg-positive) patients.6 A similarly designed study (NV-02B-015, Ó015Ó) was conducted in China and enrolled 332 (290 HBeAg-positive) patients.7 In both studies, treatment with telbivudine for 2 years demonstrated significantly greater antiviral effects, compared with lamivudine, in both HBeAg-positive and HBeAg-negative patients.
· Historically, trials reporting durability of HBeAg seroconversion with lamivudine treatment have reported limited durability (relapse rate 56%) of HBeAg seroconversion.8 These data may not be relevant to modern clinical practice, because standard methodology was not used, and in particular, no consolidation treatment was offered to patients. More recently, 96-week data for entecavir and lamivudine have shown off-treatment HBeAg seroconversion sustained in 77% and 72% of patients, respectively.9
Aim
· The aim of this analysis was to assess off-treatment durability of response in HBeAg-positive patients followed for up to 52 weeks after discontinuation of telbivudine or lamivudine therapy in the combined populations of studies 007 and 015.
Methods
· The efficacy of telbivudine 600 mg daily was compared with that of lamivudine 100 mg daily in two 104-week phase III studies in patients with CHB, HBV DNA 36 log10 copies/mL, ALT 1.3Đ10 x ULN, and compensated liver disease.6,7
· A total of 1211 HBeAg-positive patients were enrolled into these two trials: 458 and 463 HBeAg-positive patients were treated with telbivudine and lamivudine, respectively, in the GLOBE study, and 147 and 143 telbivudine- and lamivudine-treated HBeAg-positive patients, respectively, were studied in the 015 trial.
· The HBeAg-positive patients followed in these two trials were eligible for treatment discontinuation (based on investigator discretion) if they received at least 1 year of antiviral treatment and had HBV DNA <5 log10 copies/mL, with HBeAg loss maintained on treatment for at least 24 weeks.
· Patients continued regular follow-up visits for up to 52 weeks post-treatment in order to assess durability of HBe-Ag seroconversion.
Results
Baseline Demographics
· The HBeAg-positive groups in both studies were well matched at baseline (Table 1).
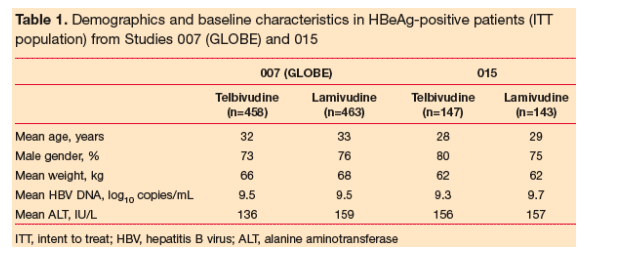
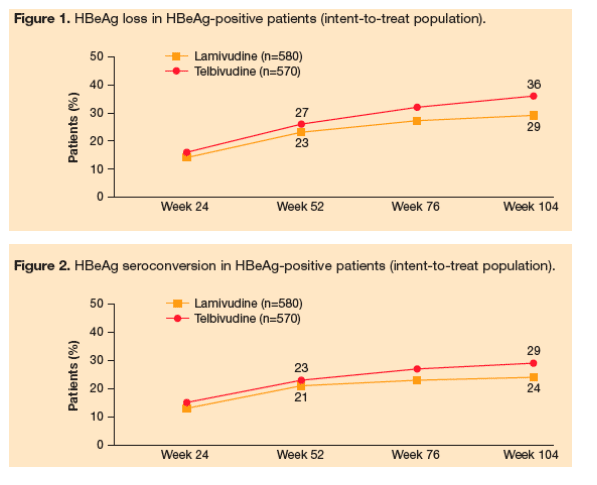
HBeAg Loss and Seroconversion
A numerically greater proportion of patients receiving telbivudine achieved HBeAg loss (Figure 1) and HBeAg seroconversion (Figure 2) at each study visit, compared with lamivudine (not statistically significant); a numerically greater proportion of telbivudine rather than lamivudine recipients therefore met this important criterion for treatment discontinuation.
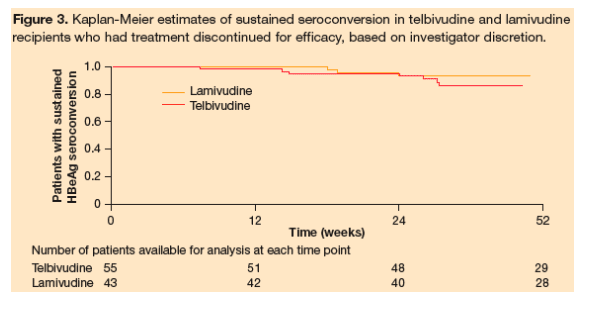
Off-Treatment Durability of HBeAg Responses
Among patients who discontinued treatment due to efficacy (treatment discontinued at the discretion of the investigator, after at least 6 months of treatment consolidation) and who had retained HBeAg seroconversion at the time of treatment cessation, the KaplanĐMeier estimates of sustained off-treatment HBeAg seroconversion during the follow-up period were 98%, 94%, and 86% after 12, 24, and 52 weeks, respectively, in the telbivudine treatment group, and 100%, 95%, and 93%, respectively, in the lamivudine group (Figure 3). There was no significant difference between the two arms.
|
| |
|
 |
 |
|
|