 |
 |
 |
| |
Efficacy of DRV/r in the randomised, controlled, Phase III ARTEMIS trial in treatment-naive, HIV-1-infected patients is independent of HIV-1 subtype
|
| |
| |
Reported by Jules Levin
6th European HIV Drug Resistance Workshop, Budapest, Hungary, 26-28 March
2008.
Inge Dierynck,1 Sandra De Meyer,1 Erkki Lathouwers,1 Carline Vanden Abeele,1 Tom Van De Casteele,1 Ralph DeMasi,2 Andrew Hill,3 Sabrina Spinosa-Guzman,1
Gaston Picchio,2 Marie-Pierre de BŽthune1
1Tibotec BVBA, Mechelen, Belgium; 2Tibotec Inc., Yardley, PA, USA; 3University of Liverpool, Liverpool, UK
AUTHOR CONCLUSIONS
In ARV-naive patients enrolled in the ARTEMIS study, DRV/r 800/100mg once daily was
- highly effective, overall
- equally effective irrespective of HIV-1 subtype analysed.
The results of this analysis confirm previous in-vitro findings on the broad activity of DRV against different HIV-1 subtypes.
Introduction
· HIV-1 is characterised by extensive genetic heterogeneity. HIV-1 subtype B is the predominant genetic form found in Western and Central Europe, the Americas and Australia.1 Subtype C accounts for almost 50% of all HIV-1 infections worldwide.1 Circulating recombinant forms (CRFs) are reported in 18% of incident infections and represent the local predominant form in South-east Asia (CRF01_AE).1
· Darunavir (DRV; TMC114) with low-dose ritonavir (DRV/r) at a dose of 600/100mg twice daily has been approved in Europe2 and other countries including the USA3 for the treatment of HIV-1 infection in treatment-experienced adult patients. Based on the 24-week dose-finding results from the POWER 1 and 2 (TMC114-C213 and C202) studies,4,5 once-daily DRV/r 800/100mg was selected as the dose for treatment-naive, HIV-1-infected patients.
· Once-daily DRV/r 800/100mg is being examined in ARTEMIS (TMC114-C211; AntiRetroviral Therapy with TMC114 Examined In Naive Subjects), an ongoing Phase III study comparing the efficacy and safety of DRV/r versus lopinavir with low-dose ritonavir (LPV/r) in treatment-naive, HIV-1-infected adult patients across 26 countries.
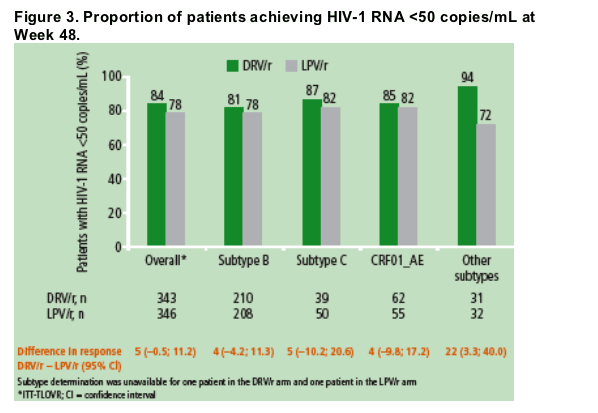
· In ARTEMIS, the primary 48-week analysis showed that 84% of patients in the DRV/r arm achieved HIV-1 RNA <50 copies/mL at Week 48 vs 78% of patients in the LPV/r arm (p value for noninferiority, p<0.001).6
· The ARTEMIS trial is characterised by a diverse representation of infecting HIV-1 subtypes among the enrolled population. Thus, it provides an opportunity to assess the effect of HIV-1 subtype on the virological response to DRV/r and LPV/r in the clinical setting and to confirm in-vitro findings on the activity of DRV against a broad panel of HIV-1 group M (A, B, C, D, E, F and G) and group O primary isolates.3
Methods
In ARTEMIS, treatment-naive, HIV-1-infected adult patients with HIV-1 RNA >5,000 copies/mL were randomised to receive DRV/r 800/100mg once daily or LPV/r 800/200mg total daily dose in addition to a fixed dose combination of tenofovir 300mg and emtricitabine 200mg once daily (Figure 1).

Sample HIV-1 subtype was derived as the best match in-between the determined protease-reverse transcriptase nucleotide sequence (determined by Virco BVBA, Mechelen, Belgium) and the corresponding ones from the Los Alamos National Laboratory subtype reference subset (2005).7
Virological response (defined as HIV-1 RNA <50 copies/mL) at Week 48 was determined using the time-to-loss of virological response (TLOVR) algorithm.
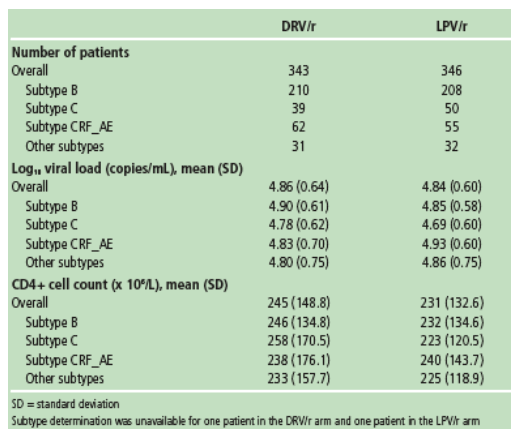
For subgroup analyses, demographics were summarised at baseline and virological (intent-to-treat [ITT]-TLOVR) and immunological responses (ITT non-completer status equals failure imputation algorithm [NC=F]) were summarised at Week 48 by subtype and by treatment arm.
Results
Subtype distribution in ARTEMIS
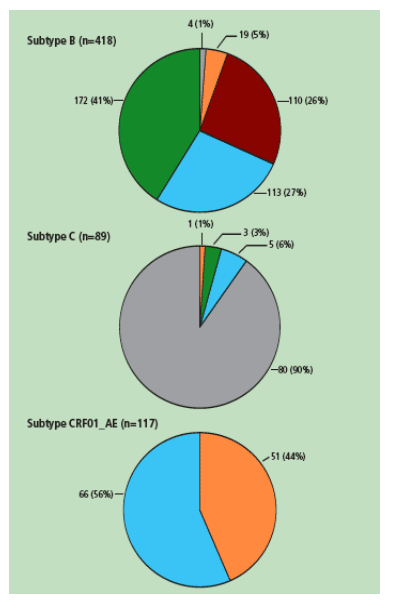
The majority (61%) of patients in ARTEMIS harboured HIV-1 subtype B; other prevalent subtypes found were subtype C (13%) and subtype CRF01_AE (17%); 9% of patients harboured other subtypes (Table 1).
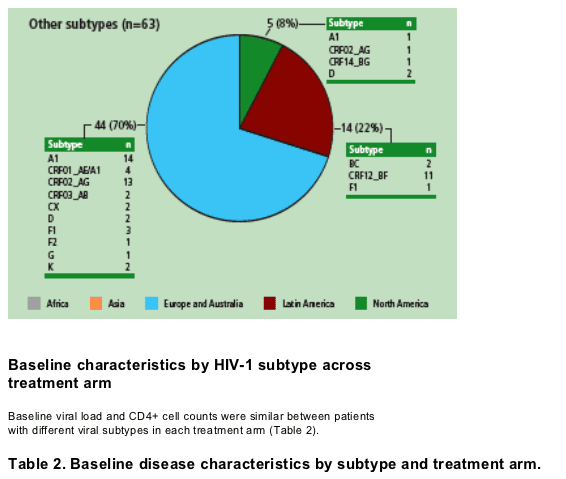
In-line with published data,1 HIV-1 subtype B was found primarily in patients in the Americas and Europe/Australia, subtype C was found primarily in Africa and subtype CRF01_AE was found in Europe/Australia and Asia (Figure 2). The increasing prevalence of non-B subtypes in Europe has been related to immigration and the strong relationships with former colonies.

Virological responses by HIV-1 subtype
Virological responses according to HIV-1 subtype are shown in Figure 3
- response rates in both treatment arms were comparable across patient groups harbouring subtypes B, C and CRF01_AE
- higher numerical response rates were observed in all non-B subtype groups versus the B subtype group in the DRV/r treatment arm
-- although the subgroup of patients with other subtypes was limited and diverse (13 different subtypes), the differences in response rates observed between the DRV/r and LPV/r arms for each subtype were consistent with the overall results.
Immunological responses by HIV-1 subtype
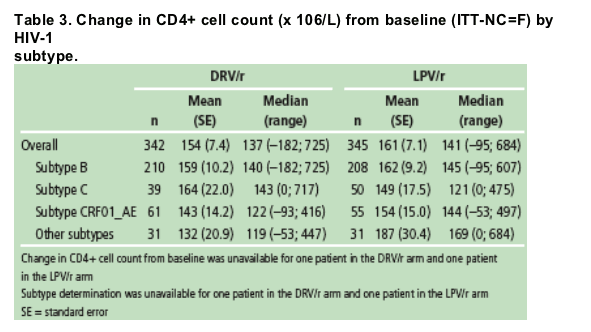
Table 3 shows the immunological responses in ARTEMIS patients by
subtype and by treatment arm.
Mean changes in CD4+ cell count from baseline through Week 48 were comparable
- in both treatment arms, overall
- across subtypes B, C, and CRF01_AE in both treatment arms.
No clinically relevant impact of subtype on immunological response was seen in both treatment arms.
References
1. Osmanov S, et al. J Acquir Immune Defic Syndr 2002;29:184-90.
2. PREZISTA (darunavir) Summary of Product Characteristics. February 2007 [accessed 10 March 2008]. Available from:
http://www.emea.europa.eu/humandocs/PDFs/EPAR/prezista/H-707-PI-en.pdf.
3. Tibotec Inc. PREZISTA (darunavir) Prescribing Information. October 2006 [accessed 10 March 2008]. Available from: http://www.prezista.com/prezista/.
4. Katlama C, et al. AIDS 2007;21:395-402.
5. Haubrich R, et al. AIDS 2007;21:F11-F18.
6. DeJesus E, et al. 47th Interscience Conference on Antimicrobial Agents and Chemotherapy, Chicago, IL, USA, 17-20 September 2007. Abstract H-718b.
7. Los Alamos National Laboratory. HIV-1 Subtype and Circulating Recombinant Form Reference Sequences. 2005 [accessed 10 March 2008]. Available from: http://hiv.lanl.gov.
|
| |
|
 |
 |
|
|