| |
Efficacy and Safety of 3 Years Treatment with Entecavir (ETV) in Japanese Nucleoside-Naive Patients with Chronic Hepatitis B (CHB)
|
| |
| |
Reported by Jules Levin
The 18th Conference of the Asian Pacific Association for the Study of the Liver 2008
T Takehara1, N Hayashi1, K Takaguchi2, O Yokosuka3, S Fujioka4, M Shindo5, K Chayama6, J Toyota7, S Mochida8, H Ikeda9, E Tomita10, K Suzuki11, H Kumada12, T Okanoue13, M Sata14, H Tsubouchi15, H Ishikawa16, T Seriu17, M Omata18
1Department of Gastroenterology and Hepatology, Osaka University Graduate School of Medicine, Japan; 2Department of Internal Medicine, Kagawa Prefectural Central Hospital, Japan; 3Department of Medicine and Clinical Oncology, Graduate School of Medicine, Chiba University, Japan; 4Department of Internal Medicine, Okayama Saiseikai General Hospital, Japan; 5Division of Liver Disease, Department of Internal Medicine, Akashi Municipal Hospital, Japan; 6Department of Medicine and Molecular Science, Graduate School of Biomedical Sciences, Hiroshima University, Japan; 7Department of Hepatology, Sapporo Kosei General Hospital, Japan; 8Department of Gastroenterology and Hepatology, Saitama Medical School, Japan; 9Department of Gastroenterology and Hepatology, Kurashiki Central Hospital, Japan; 10Department of Gastroenterology, Gifu Central Hospital, Japan; 11First Department of Internal Medicine, Iwate Medical University, Japan; 12Department of Gastroenterology and Hepatology, Toranomon Hospital, Japan; 13Department of Gastroenterology and Hepatology, Kyoto Prefectural University of Medicine, Japan; 14Division of Gastroenterology, Department of Medicine, Kurume University School of Medicine, Japan; 15Department of Digestive Disease and Life-Style Related Disease, Kagoshima University Graduate School of Medical and Dental Science, Japan; 16Pharmaceutical Research Institute, Bristol-Myers Squibb Company, Japan; 17Pharmaceutical Research Institute, Bristol-Myers Squibb Company, USA; 18Department of Gastroenterology, Graduate School of Medicine, University of Tokyo, Japan
AUTHOR CONCLUSIONS
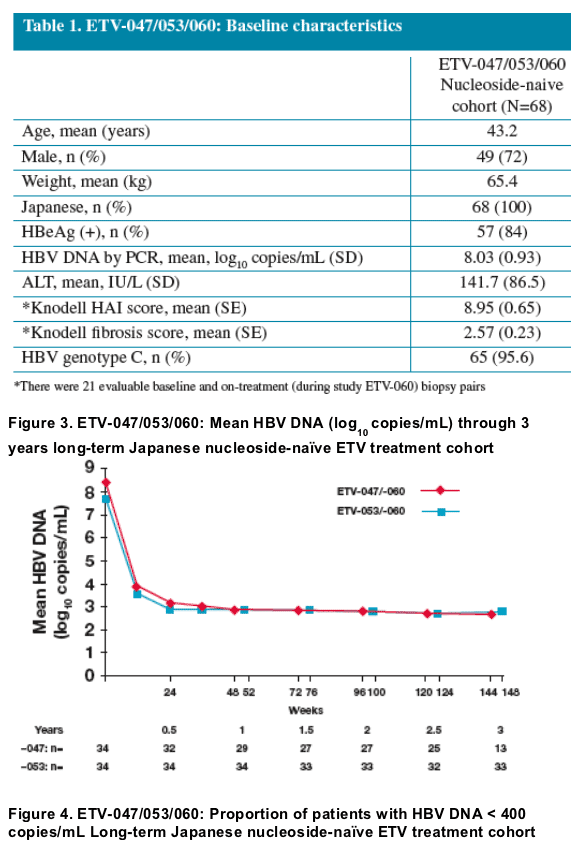
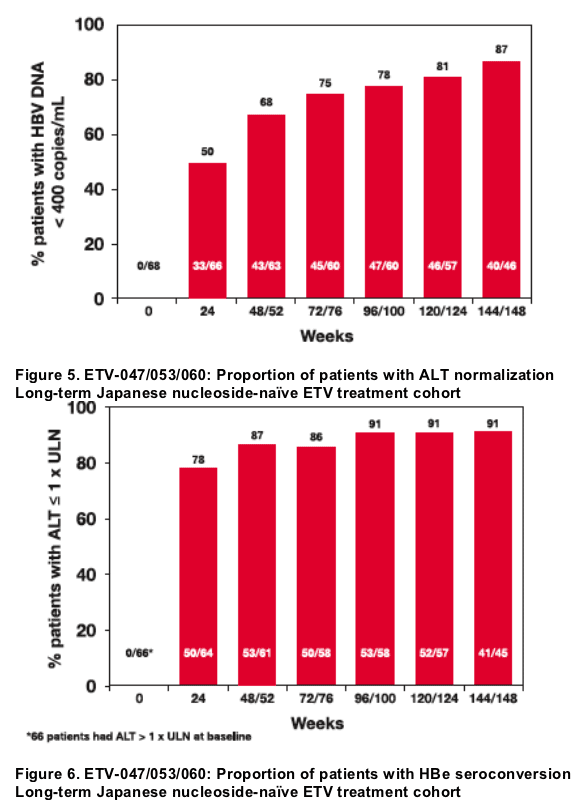
· 87% of nucleoside-naive patients treated continuously with entecavir 0.5 mg daily for 3 years achieved HBV DNA <400 copies/mL
· Continuous treatment through 3 years also resulted in ALT normalization in 91% of patients
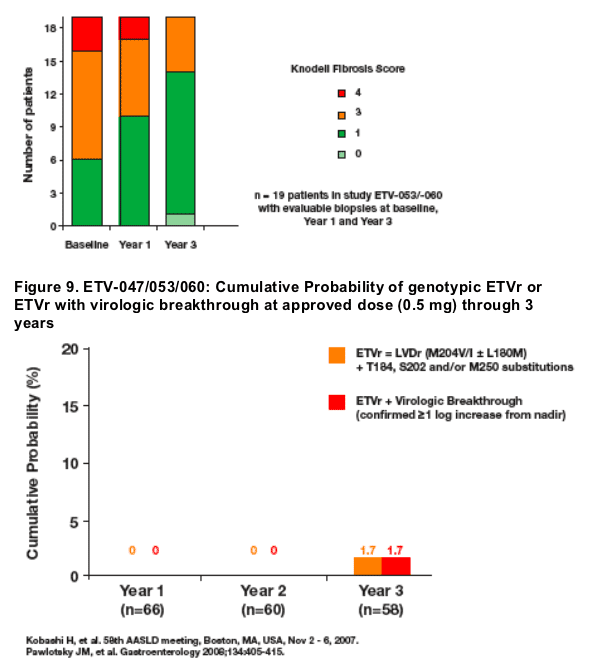
· Repeated histological assessment over 3 years demonstrated improvement in necroinflammation in 100% of patients and improvement in fibrosis in 63% of patients
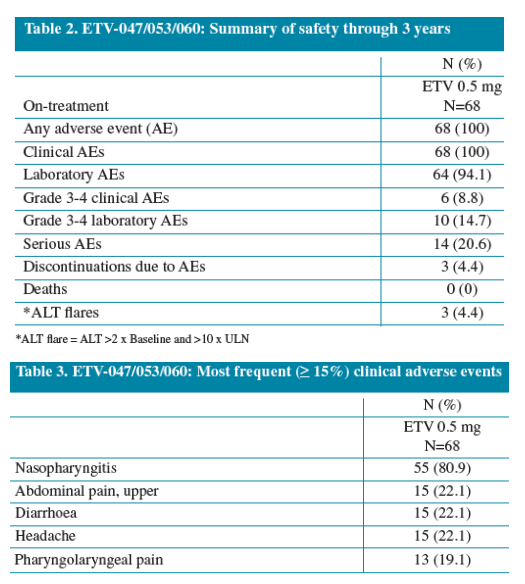
· The cumulative probability of entecavir resistance at the approved 0.5 mg dose through 3 years was 1.7%
· Entecavir was well-tolerated through 3 years of treatment
Introduction
· The goal of chronic hepatitis B (CHB) treatment is to achieve sustained suppression of HBV DNA and remission of liver disease
· Entecavir 0.5 mg daily for 48 weeks resulted in high rates of HBV DNA suppression, ALT normalization, and histologic improvement in study ETV-053 in Japan1
· Entecavir 0.5 mg daily for 24 weeks demonstrated superior HBV DNA reduction compared to lamivudine 100 mg daily in study ETV-047 in Japan2
· Patients from studies ETV-053 or -047 could enroll in rollover study ETV-060
· We present long-term efficacy, safety and resistance results of nucleoside-naive CHB patients who received up to 3 years of treatment with entecavir
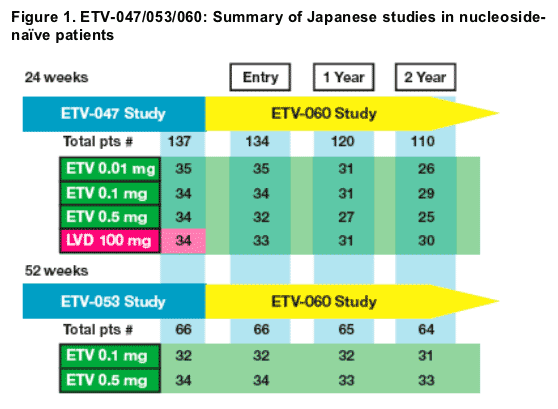
68 patients from prior nucleoside-naive studies received entecavir 0.5 mg daily
- ETV-047: n=34
- ETV-053: n=34
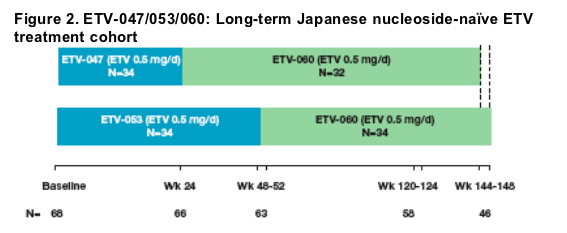
Program design
Eligibility criteria (studies ETV-047 and ETV-053)
- Chronic hepatitis B infection with compensated liver disease
- HBV DNA > 5 log10 copies/mL by PCR assay (> 7.6 log10 copies/mL for ETV 047)
- < 12 weeks prior treatment with anti-HBV nucleoside analogues
- ALT 1.25-10 x ULN
- HBeAg(+) or (-)
Study ETV-060
- Enrolment immediately after completion of ETV-047 or ETV-053 with no gap in dosing
- ETV 0.5 mg daily for a total treatment time of up to 148 weeks
Efficacy assessments through Week 148
- Mean change from baseline in HBV DNA by PCR
- Undetectable HBV DNA (< 400 copies/mL) by PCR
- ALT < 1.0 x ULN
- HBeAg seroconversion
- Histologic improvement by Knodell classification
- Resistance and virologic breakthrough
Safety
RESULTS

References
1. Takaguchi K, et al. J Clin Virol 2006;36(Suppl 2):S96.
2. Shindo M, et al. J Clin Virol 2006;36(Suppl 2):S94.
3. Kobashi H, et al. 58th AASLD meeting, Boston, MA, USA, Nov 2 - 6, 2007.
4. Pawlotsky JM, et al. Gastroenterology 2008;134:405-415.
|
|
| |
| |
|
|
|