| |
Ten Years' Experience with Alendronate (5, 10 and 20 mg daily) for Osteoporosis in Postmenopausal Women
|
| |
| |
NEJM March 2004
Henry G. Bone, M.D., David Hosking, M.D., Jean-Pierre Devogelaer, M.D., Joseph R. Tucci, M.D., Ronald D. Emkey, M.D., Richard P. Tonino, M.D., Jose Adolfo Rodriguez-Portales, M.D., Robert W. Downs, M.D., Jayanti Gupta, Ph.D., Arthur C. Santora, M.D., Ph.D., Uri A. Liberman, M.D., Ph.D., for the Alendronate Phase III Osteoporosis Treatment Study Group
From Michigan Bone and Mineral Clinic, Detroit (H.G.B.); Medical Research Center, Nottingham City Hospital, Nottingham, United Kingdom (D.H.); Saint-Luc University Hospital, Universite Catholique de Louvain, Brussels, Belgium (J.-P.D.); Department of Medicine, Roger Williams General Hospital, Providence, R.I. (J.R.T.); Radiant Research-Reading, Wyomissing, Pa. (R.D.E.); Good Health Associates in Adult Medicine, South Burlington, Vt. (R.P.T.); Departamento de Endocrinologia, Escuela de Medicina, Universidad Catolica de Chile, Santiago, Chile (J.A.R.-P.); Virginia Commonwealth University, Richmond (R.W.D.); Merck Research Laboratories, Rahway, N.J. (J.G., A.C.S.); and Felsenstein Medical Research Center, Sackler Faculty of Medicine, Tel Aviv University, Petah-Tikva, Israel (U.A.L.).
ABSTRACT
Background Antiresorptive agents are widely used to treat osteoporosis. We report the results of a multinational randomized, double-blind study, in which postmenopausal women with osteoporosis were treated with alendronate for up to 10 years.
Methods The initial three-year phase of the study compared three daily doses of alendronate with placebo. Women in the original placebo group received alendronate in years 4 and 5 and then were discharged. Women in the original active-treatment groups continued to receive alendronate during the initial extension (years 4 and 5). In two further extensions (years 6 and 7, and 8 through 10), women who had received 5 mg or 10 mg of alendronate daily continued on the same treatment. Women in the discontinuation group received 20 mg of alendronate daily for two years and 5 mg daily in years 3, 4, and 5, followed by five years of placebo. Randomized group assignments and blinding were maintained throughout the 10 years. We report results for the 247 women who participated in all four phases of the study.
Results Treatment with 10 mg of alendronate daily for 10 years produced mean increases in bone mineral density of 13.7 percent at the lumbar spine (95 percent confidence interval, 12.0 to 15.5 percent), 10.3 percent at the trochanter (95 percent confidence interval, 8.1 to 12.4 percent), 5.4 percent at the femoral neck (95 percent confidence interval, 3.5 to 7.4 percent), and 6.7 percent at the total proximal femur (95 percent confidence interval, 4.4 to 9.1 percent) as compared with base-line values; smaller gains occurred in the group given 5 mg daily. The discontinuation of alendronate resulted in a gradual loss of effect, as measured by bone density and biochemical markers of bone remodeling. Safety data, including fractures and stature, did not suggest that prolonged treatment resulted in any loss of benefit.
Conclusions The therapeutic effects of alendronate were sustained, and the drug was well tolerated over a 10-year period. The discontinuation of alendronate resulted in the gradual loss of its effects.
Postmenopausal osteoporosis is a chronic, progressive disorder in which bone resorption exceeds formation, resulting in decreased bone mass and deterioration of the microarchitecture, with consequent decreased bone strength and increased susceptibility to fracture.1,2 Antiresorptive agents are widely used to treat osteoporosis. The reduction in the risk of fracture during antiresorptive treatment has been related to the magnitude of changes in bone mineral density and remodeling activity.3,4,5,6 Alendronate, a potent inhibitor of bone resorption, has produced sustained reductions in biochemical markers of bone remodeling into the premenopausal range7,8 and consistent dose-related increases in bone mineral density in a variety of populations, including elderly women.7,8,9,10,11,12,13,14 These effects have been associated with a substantially reduced risk of vertebral and nonvertebral fractures.3,4,5,15,16,17,18,19,20 Bone biopsy and histomorphometric analysis have confirmed that normal bone structure and mineralization are preserved and that bone turnover is reduced but not completely suppressed.21 Mineralization is increased but remains within the normal range.22,23 The favorable effects of alendronate on bone turnover, mass, and strength have been confirmed in animal models.24,25,26
In a pair of identical three-year randomized, placebo-controlled trials, alendronate increased bone mineral density, decreased bone turnover, and reduced the risk of vertebral fracture among women with osteoporosis.7,8,20 To investigate the effects of prolonged alendronate therapy as well as its discontinuation, these trials were extended for a total of 10 years. Results for the first seven years have been described.27,28 In this article we report the results through the final 3-year extension of the study, including 5 years of observation after the discontinuation of alendronate, and the cumulative, 10-year experience with alendronate.
Methods
Study Design
Two identical, concurrent multicenter, double-blind, randomized, placebo-controlled phase 3 studies,7,8,20 designed to permit pooling of results, enrolled a total of 994 postmenopausal women with osteoporosis that had been diagnosed on the basis of the bone mineral density of the lumbar spine.7,8,20,27
Initially, women were randomly assigned to receive 5, 10, or 20 mg of oral alendronate (Fosamax, Merck) or placebo daily. Figure 1 shows the treatment assignments for the original study and the extensions. Women in the placebo group were given open-label alendronate for years 4 and 5 and then discharged from the study. The original 5-mg and 10-mg alendronate groups continued to receive the same doses in all three extensions of the study (years 4 and 5, 6 and 7, and 8 through 10). Those in the original 20-mg group received 5 mg for years 3 through 5 and placebo for years 6 through 10 (the discontinuation group). Their cumulative exposure to alendronate was similar to the exposure in the 10-mg group after 5 years and to that in the 5-mg group after 10 years. Investigators and the women were aware that all long-term participants had received alendronate for at least five years and that the discontinuation group had been switched to placebo, but all remained unaware of each woman's current treatment.
The women were instructed to take the study medication daily, consistent with the instructions in the product insert. They received 500 mg of calcium daily. Vitamin D supplements were permitted but not required.
Twenty-nine of the original 37 centers carried out all three extension protocols, 17 within the United States and 12 in other countries. These sites contributed 804 of the original 994 study participants. Of the 482 women originally assigned to alendronate at those sites, 247 (51.0 percent) participated in all three extensions of the study. Protocols and extensions were approved by institutional review boards. Each woman gave written informed consent for the study and each extension.
Measurements
Efficacy End Points
The primary end point was the change in bone mineral density at the lumbar spine. Secondary end points were changes in bone mineral density at the femoral neck, trochanter, total proximal femur ("total hip"), total body, and forearm regions; changes in the levels of urinary N-telopeptides of type I collagen, a biochemical marker of bone resorption; and changes in the levels of serum bone-specific alkaline phosphatase and total serum alkaline phosphatase, indicators of the rates of bone formation. Bone mineral density was measured yearly by dual-energy x-ray absorptiometry (Hologic, Lunar, and Norland) and interpreted centrally by a quality-assurance center (Hologic MDM/Synarc) in a blinded fashion.20,27 Biochemical markers were measured annually, and specimens were analyzed as they were received during years 8 through 10, whereas earlier results were based on batched, archived specimens.7,8,27
Evaluation of Fractures
Data on morphometrically detected vertebral fractures, clinical fractures, and stature were collected as safety end points. No formal comparisons were planned, owing to the limited sample size and the fact that all the women in this portion of the study had previously received alendronate. Radiologically confirmed symptomatic nonvertebral fractures were considered adverse clinical events, with no attempt to exclude fractures related primarily to trauma. Stature was measured annually with Harpenden stadiometers.20,27
Standardized lateral radiographs of the thoracolumbar spine were scheduled at the end of each extension or at the time of early termination. Radiographs were read locally to facilitate patient care. Locally diagnosed vertebral fractures were reported as adverse events. A central evaluation was conducted after the study was completed (Synarc).28 The radiologist, who was unaware of the subjects' treatment assignments, evaluated the earliest technically satisfactory film obtained at the end of year 5 or thereafter and the latest subsequent film. Updated digitization methods were used.29 As in the original study,20 morphometric vertebral fracture was defined by a loss in vertebral height of at least 20 percent and at least 4 mm. The vertebral-fracture results presented here are the centrally read morphometric results for years 6 through 10 among women who continued in the study after year 7 (third extension).
Statistical Analysis
The preplanned analyses pooled data from U.S. and international centers. The results reported here are restricted to the 247 women who participated in the third extension of the study (years 8 through 10). The primary data are maintained by the sponsor as required by the Food and Drug Administration and other regulatory authorities. All authors had access to a complete set of results. The trial statistician analyzed the data and responded to all queries raised by the authors.
Bone Mineral Density
The modified intention-to-treat analysis included all women with measurements at base line, year 7, and at least one subsequent year in which a study drug was given; data from the last evaluation were carried forward in women who did not complete year 10. All bone density measurements of vertebrae at which new lumbar vertebral fractures were detected were censored for all time points.
Markers of Bone Turnover
Because analyses of biochemical markers were intended to evaluate pharmacodynamic effects, they were performed only in women who were in compliance with the protocol (per-protocol analysis). The effects of treatment are expressed as the percent change in values (geometric means). The distribution of changes was normalized by means of a natural-logarithmic transformation. Analysis of variance was used to examine treatment effects within groups, with factors for treatment and study center.
Safety
The proportions of women with clinical adverse events and laboratory adverse events were compared with the use of 95 percent confidence intervals. The incidence of laboratory values outside predefined limits was also examined. Analyses of the incidence of fracture were based on the first fracture within a specified period. Annualized height loss was estimated primarily from the mean loss during years 6 through 10, according to the modified intention-to-treat principle; a secondary analysis did not carry values forward. Using logistic-regression coefficients derived from the Study of Osteoporotic Fractures, we estimated that the rate of non-vertebral fracture for a placebo group after a 7-year increase in age would be 1.26 times the rate observed during years 1 through 3 (or 1.18 for a five-year age increase), and the expected increase in the risk of vertebral fracture attributable to advancing age for a placebo group over the interval from 6 to 10 years would be 1.53 times that observed in years 1 through 3.30,31
Results
The characteristics of the 247 women in the third extension of the study, at the time of their initial randomization, were similar among the groups and were similar to those of all 994 participants at base line. The prevalence of preexisting vertebral fracture and was 30.8 percent in the 5-mg group, 17.5 percent in the 10-mg group, and 27.2 percent in the discontinuation group. These differences were not significant. The prevalence of vertebral fracture was 20.6 percent in the original study population at base line (Table 1).
Bone Mineral Density
Bone mineral density at the lumbar spine continued to increase during years 6 through 10 and 8 through 10 in both alendronate groups (Table 2 and Figure 2A). The mean cumulative increase after 10 years of the 10-mg daily dose was 13.7 percent, as compared with the base-line value; smaller gains occurred in the 5-mg group. During years 6 through 10, the alendronate groups had no significant decline in bone mineral density at any skeletal site. In the discontinuation group, bone mineral density at the lumbar spine did not change significantly after year 5; significant decreases occurred at the total hip, femoral neck, and forearm, but bone mineral density at the lumbar spine, trochanter, total hip, and total body remained significantly above base-line values at year 10 (Table 2 and Figure 2A through 2E).
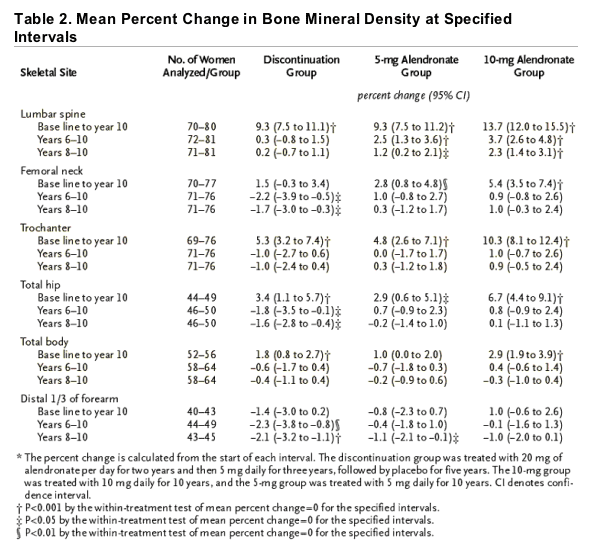
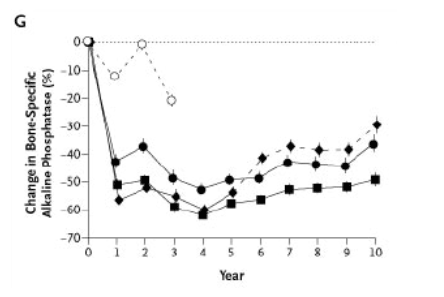
Figure 2. Mean (±SE) Percent Change in Bone Mineral Density (BMD) at the Lumbar Spine (Panel A), Femoral Neck (Panel B), Trochanter (Panel C), Total Hip (Panel D), and Total Body (Panel E) and in Urinary N-Telopeptides of Type I Collagen Values (Panel F) and Serum Bone-Specific Alkaline Phosphatase Levels (Panel G) over a 10-Year Period.
The discontinuation group was treated with 20 mg of alendronate per day for two years and then 5 mg daily for three years, followed by placebo for five years. The 10-mg group was treated with 10 mg of alendronate daily for 10 years, and the 5-mg group was treated with 5 mg of alendronate daily for 10 years. Ten years of alendronate treatment resulted in significant dose-dependent increases in bone mineral density at the lumbar spine and femur. In the discontinuation group, some net effects on bone mineral density and markers of bone turnover persisted through year 10, in a manner that was consistent with the cumulative dose received. Solid lines indicate the period during which alendronate was administered, and dashed lines the period of placebo administration.
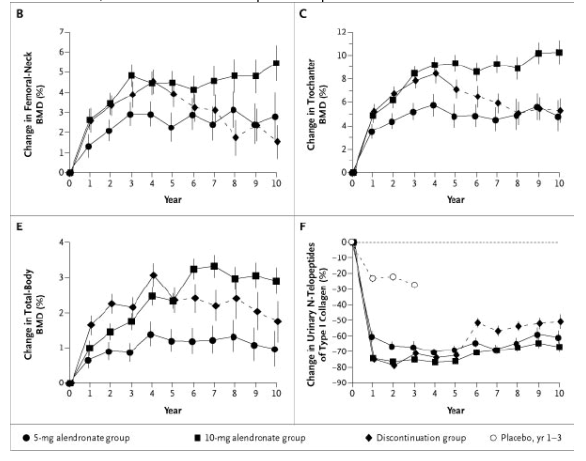
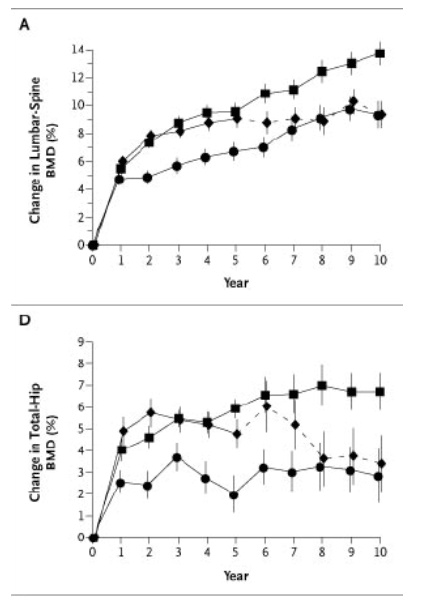
The vast majority of women who received alendronate had increases in bone mineral density during the 10-year study. For example, 89 percent of women who took the 10-mg dose daily had an increase (as indicated by a change greater than 0) in bone mineral density at the total hip.
Markers of Bone Remodeling
In the initial study, alendronate reduced the levels of markers of bone remodeling to stable nadir levels in the normal premenopausal range.7,8 This effect was sustained through 10 years of treatment (Figure 2F and Figure 2G). Mean levels of urinary N-telopeptides of type I collagen in the 10-mg group declined from 66.6 nmol of bone collagen equivalent per millimole of creatinine at base line to 22.0 nmol per millimole at the end of year 10 (the latter is similar to the published mean value for premenopausal women32). During years 8 through 10, levels of urinary N-telopeptides of type I collagen remained stable in alendronate groups. The mean serum bone-specific alkaline phosphatase level in the 10-mg group was 17.8 ng per milliliter at base line and 9.1 ng per milliliter at the end of year 10 (which is also similar to the published mean value for premenopausal women32). After the discontinuation of alendronate, levels of markers of bone remodeling increased within a year,27 but the mean values remained below base-line values. Although the mean total alkaline phosphatase levels were unchanged, small increases in mean serum bone-specific alkaline phosphatase values were measured during the final year of the study (Figure 2G).
Skeletal Safety
Vertebral Fracture
During the initial three-year study, 6.2 percent of women in the placebo group had new morphometric vertebral fractures, as compared with 3.2 percent in the pooled alendronate groups (P=0.03)20;proportions among the three alendronate groups were similar. For this analysis, 228 women could be evaluated for years 6 through 10. During those five years, the proportions of women with new morphometric vertebral fractures did not differ significantly among the three groups: 6.6 percent in the discontinuation group, 13.9 percent in the 5-mg group, and 5.0 percent in the 10-mg group. These proportions are difficult to compare with those for years 1 through 3 owing to the difference in the lengths of the observation periods. The rates of fracture (e.g., the number per 100 subject-years) could not be calculated accurately because the date of occurrence within the observation interval was usually not known.
Stature
Height loss in the 10-mg group during years 6 through 10 was similar to that among all alendronate-treated women during the first three years. The height loss was slightly but not significantly greater in the 5-mg and discontinuation groups than in the 10-mg group during years 6 through 10 (Figure 3A).
Nonvertebral Fracture
In years 6 through 10, the rate of radiologically confirmed nonvertebral fractures in the 10-mg group was similar to that in the pooled alendronate groups during the first three years of the study (Figure 3B). No insufficiency fractures or instances of fracture malunion were reported.
General Safety and Tolerability
During years 8 through 10, the safety profiles were similar among all three groups (Table 3). Four women died during years 8 through 10, all of whom were in the 5-mg group. None of the deaths were attributed to alendronate. The incidence of all upper gastrointestinal adverse events was similar among the three groups. One or two women in each group withdrew because of upper gastrointestinal adverse events. During years 8 through 10, 30 to 36 percent of women in each treatment group used aspirin, and 41 to 53 percent used nonsteroidal or glucocorticoid antiinflammatory agents. There appeared to be no adverse interaction between these drugs and alendronate.
Discussion
Alendronate appeared to be effective over the 10-year period of the study. The observed increases in bone mineral density at the lumbar spine during long-term alendronate therapy are consistent with models predicting that a positive bone-remodeling balance and increased secondary mineralization would be contributing factors.33,34 Nonstructural calcifications may have contributed to the measured increase in bone mineral density at the lumbar spine but are unlikely to explain most of the effect.35,36
After an initial reduction, the levels of bone-remodeling markers remained essentially stable within the premenopausal range during treatment. After the discontinuation of alendronate therapy, levels of these markers increased but remained below pretreatment levels. The small increase in serum bone-specific alkaline phosphatase levels during year 10 in all three groups could be due to subtle changes in the performance of the assay, particularly in view of the stable levels of total alkaline phosphatase.
The long retention time of alendronate in bone37 may result in gradual recycling and some residual suppression of bone resorption.38 The partial maintenance of the drug's effect after the discontinuation of therapy could be useful, particularly if adherence to therapy is inconsistent.39 In contrast, the discontinuation of estrogen results in a relatively rapid decline in bone mineral density and an increase in bone turnover.40,41,42,43,44,45 Accelerated bone resorption may cause microstructural weakness.3 This may explain the rapid diminution of antifracture efficacy after estrogen therapy is stopped.46 Increased rates of bone turnover and losses in bone mineral density also occur after the withdrawal of raloxifene.43
We used the incidence of fracture as a safety measure rather than an efficacy measure. Varying intervals between spinal radiographs, a change in analysis center, and the small number of events limit the interpretation of the incidence of vertebral fracture. Increases in age further confound comparison between the initial and final intervals. We calculated the increases in the incidence of fracture that would have been expected if our original placebo group had continued untreated. Our observations do not suggest any association between prolonged use of alendronate and an excess risk of fracture. Although the differences were not statistically significant, the fewest morphometric vertebral fractures, least height loss, and lowest rate of nonvertebral fractures occurred in the group given 10 mg of alendronate, which also had the greatest cumulative exposure to alendronate. Thus, there was no indication of any adverse cumulative effect. Other indicators of safety and tolerability were similar among the groups during years 8 through 10.
In summary, continuous treatment with 10 mg of alendronate daily for 10 years was associated with sustained therapeutic effects on bone density and remodeling, with no indication that the antifracture efficacy of the drug was diminished. The discontinuation of alendronate resulted in a gradual diminution of effect. Because each therapeutic agent used for the treatment of osteoporosis may have unique characteristics, our observations should not be assumed to apply to other treatments for osteoporosis.
Supported by Merck Research Laboratories. Presented in part at the Annual Meeting of the American Society for Bone and Mineral Research, San Antonio, Tex., September 20-24, 2002.
Dr. Bone reports having received honorariums from Merck; grant support from Merck, Amgen, NPS, Novartis, and Pfizer; and consulting fees from Merck, Novartis, Amgen, AstraZeneca, Aventis, Debio, En Pharma, GlaxoSmithKline, Roche, Nordic Bone, NPS, ProSkelia, Schering-Plough, Kyowa, Wyeth, and Zelos. Dr. Hosking reports having received honorariums from Merck, Procter & Gamble, and Novartis and grant support from Merck, Novartis, Eli Lilly, Procter & Gamble, Aventis, and Shive. Dr. Devogelaer reports having received grant support from Merck, Novartis, Eli Lilly, Procter & Gamble, and Cosucra. Dr. Tucci reports having received honorariums and consulting fees from Merck and Procter & Gamble and grant support from Merck and Bayer. Dr. Emkey reports having received honorariums and lecture fees from Merck. Dr. Rodriquez-Portales reports having received grant support from Wyeth. Dr. Downs reports having received grant support, consulting fees, and lecture fees from Merck and Lilly, as well as grant support from Wyeth. Dr. Santora reports holding equity in Merck and receiving several U.S. and international patents as inventor related to the use of bisphosphonates that are assigned to Merck. Dr. Liberman reports having received lecture fees from Merck.
|
|
| |
| |
|
|
|