| |
Effects of Alendronate on BMD and Fracture Risk: The FOSIT Study
|
| |
| |
Multinational, Placebo-Controlled, Randomized Trial of the Effects of Alendronate on Bone Density and Fracture Risk in Postmenopausal Women with Low Bone Mass: Results of the FOSIT Study
Subsequent to this study alendronate (Fosamax) was administered once weekly.
Osteoporos Int (1999) 9:461-468
1999 International Osteoporosis Foundation and National Osteoporosis Foundation
H. A. P. Pols1, D. Felsenberg2, D. A. Hanley3, J. Stepa'n4, M. Munoz-Torres5, T. J. Wilkin6, G. Qin-sheng7, A. M. Galich8, K. Vandormael9, A. J. Yates10 and B. Stych11 for the Fosamax1 International Trial Study Group
1Erasmus University Medical School, Rotterdam, The Netherlands; 2Free University of Berlin, Berlin, Germany; 3University of Calgary, Calgary, Alberta, Canada; 4Charles University, Prague, Czech Republic; 5Hospital Clinico San Cecilio, Granada, Spain; 6Freedom Fields Hospital, Plymouth, Devon, UK; 7Peking Union Medical College Hospital, Beijing, People's Republic of China;
8Hospital Italiano de Buenos Aires, Buenos Aires, Argentina; 9Merck Research Laboratories, Brussels, Belgium; 10Merck Research Laboratories, Rahway, New Jersey, USA; 11Merck & Co., Inc., Whitehouse Station, New Jersey, USA
Abstract.
This randomized, double-masked, placebo-controlled trial evaluated the safety, tolerability and effects on bone mineral density (BMD) of alendronate in a large, multinational population of postmenopausal women with low bone mass. At 153 centers in 34 countries, 1908 otherwise healthy, postmenopausal women with lumbar spine BMD 2 standard deviations or more below the premenopausal adult mean were randomly assigned to receive oral alendronate 10 mg (n = 950) or placebo (n = 958) once daily for 1 year.
All patients received 500 mg elemental calcium daily. Baseline characteristics of patients in the two treatment groups were similar. At 12 months, mean increases in BMD were significantly (p<0.001) greater in the alendronate than the placebo group by 4.9% (95% confidence interval 4.6% to 5.2%) at the lumbar spine, 2.4% (2.0% to 2.8%) at the femoral neck, 3.6% (3.2% to 4.1%) at the trochanter and 3.0% (2.6% to 3.4%) for the total hip.
The incidence of nonvertebral fractures was significantly lower in the alendronate than the placebo group (19 vs 37 patients with fractures), representing a 47% risk reduction for nonvertebral fracture for alendronate-treated patients (95% confidence interval 10% to 70%; p = 0.021). Incidences of adverse events, including upper gastrointestinal adverse events, were similar in the two groups.
Therefore, for postmenopausal women with low bone mass, alendronate is well tolerated and produces significant, progressive increases in BMD at the lumbar spine and hip in addition to significant reduction in the risk of nonvertebral fracture.
Introduction
Osteoporosis is characterized by low bone mass and microarchitectural deterioration of bone with consequent bone fragility and thus an increased risk of fracture, most commonly vertebral, wrist (Colles') and hip fractures [1,2]. Osteoporosis in postmenopausal women is an important public health issue in industrialized countries. A 50-year-old white woman has a 40% risk of experiencing a clinically apparent fracture during her remaining lifetime [3]. In the United States and Europe, approximately 30% of all postmenopausal white women have osteoporosis, as defined by bone mineral density (BMD) measurements [1]. This high prevalence of osteoporosis occurs because of low postmenopausal estrogen levels, which result in progressive bone loss secondary to a combination of increased bone turnover and an excess of bone resorption relative to bone formation [4].
Alendronate, a potent inhibitor of osteoclast-mediated bone resorption, normalizes the rate of bone turnover to premenopausal levels [5]. In both animal and human studies administration of alendronate increases bone mass and maintains histologically normal bone [6-9]. Liberman and colleagues [10] reported that treatment with 10 mg alendronate daily for 3 years resulted in mean increases, relative to placebo, of 8.8%, 5.9% and 7.8% in BMD of the lumbar spine, femoral neck and trochanter, respectively, in addition to marked decreases in vertebral and nonvertebral fracture incidence among approximately 200 women with osteoporosis. The objective of the current double-masked, placebo-controlled study was to evaluate efficacy and tolerability of alendronate over 1 year in a large and diverse multinational population of postmenopausal women with low bone mass.
Patients and Methods
Patients
We recruited patients from 153 centers in 34 countries (in Europe, Latin America, Australia, Canada, South Africa and China). Women eligible for study participation had been postmenopausal for at least 3 years, were not older than 85 years, and had BMD of the lumbar spine (L2-4) at least 2 standard deviations (SD) below the mean for mature, premenopausal women - a value that approximately corresponds to the median BMD of 65-year-old women. Lumbar spine BMD, as measured using dual-energy X-ray absorptiometry (DXA), was <0.86 g/cm2 by Hologic QDR densitometry (Hologic, Waltham, MA) or <0.98 g/cm2 by Lunar DPX densitometry (Lunar, Madison, WI). Eligible patients were otherwise in good health and were between 20% below and 50% above ideal body weight as defined in the Metropolitan Life Insurance Company Height and Weight Table. Levels of 25-hydroxyvitamin D were determined before study entry.
Excluded from participation were women with metabolic bone disease other than postmenopausal osteoporosis; disturbed parathyroid or thyroid function; major gastrointestinal disease (for example, peptic ulcer or malabsorption) within the year before enrollment or use of a drug to inhibit gastric acid secretion for >2 weeks within 3 months of study entry; myocardial infarction within the year prior to enrollment; uncontrolled hypertension or untreated angina; significantly impaired renal function (serum creatinine >150 mmol/l); or evidence of significant end organ disease. Also excluded were women who had received a bisphosphonate or fluoride (>8 mg/day) during the previous 6 months; estrogen (except vaginal 43 times/week), ipriflavone or calcitonin during the previous 4 months; or any anabolic steroid, glucocorticoid or progestin for >2 weeks within the previous 6 months. Participants could not be receiving any medications that might alter bone or mineral metabolism, including vitamin A in excess of 10.000 U/day, vitamin D in excess of 1000 U/day, anticonvulsants or phosphate-binding antacids. Finally, at least three vertebrae from L1 to L4 had to be evaluable by DXA to determine BMD in this region. Spinal radiographs were not obtained before study entry.
The study protocol was approved by each local ethics committee, and all participants signed a written informed consent form.
Study Design
After a 2- to 4-week baseline period, study participants were randomly assigned to receive alendronate 10 mg or matching-image placebo tablets once daily for 12 months. Patients were instructed to take one tablet of study drug with a glass of water each morning, after an overnight fast, and to refrain from lying down or taking any other beverage or food for at least 30 min thereafter. All patients received tablets containing 500 mg of elemental calcium as the carbonate or citrate salt with instructions to take one tablet daily at the evening meal.
Efficacy Criteria
Bone densitometry was performed during the baseline period and at 3, 6 and 12 months after starting the study drug. BMD was measured, using Hologic QDR densitometers (QDR-1000, -1000/W, -1500 or -2000; Hologic, Waltham, MA) or Lunar DPX densitometers (DPX, DPX-L or DPX-a; Lunar, Madison, WI), at the following four sites: lumbar spine (primary endpoint), femoral neck, trochanter and total hip. The mean BMD from at least three evaluable vertebrae from L1 to L4 was used to determine lumbar spine BMD. All BMD measurements were reviewed at a central quality assurance site.
Bone-specific alkaline phosphatase, a marker of bone formation, and urine N-telopeptide crosslinks of type I collagen/creatinine ratio, a marker of bone resorption, were measured at each clinic visit using previously described assays [11,12]. Assays were performed at a central laboratory.
Safety Criteria
Adverse events were recorded at each visit using nonleading questioning. An electrocardiogram and complete physical examination (repeated at 12 months) were performed at the screening visit; limited physical examinations were performed at subsequent visits. Clinical laboratory tests were performed at each visit. The occurrence of clinical fractures was captured through adverse event reporting. To evaluate whether all fractures had been correctly coded by the investigators, supporting documentation for each fracture consisting of radiographs and/or radiology reports, hospital discharge reports with clinical diagnosis and/or confirmation by the investigator/treating physician was sought after completion of the study.
Statistical Methods
The primary evaluation of the efficacy data was based on the intention-to-treat principle. The effect of treatment on BMD and biochemical markers of bone turnover was assessed using analysis of variance with factors for treatment and center.
Fisher's exact test was used to compare the incidence of adverse events. The analysis of clinical fracture incidence as an efficacy parameter, which was not predefined because of limited power, was based on the log rank test. The estimate of the relative hazard was derived from a proportional hazards model by the likelihood ratio method. Sensitivity and subgroup analyses were performed to evaluate the robustness and consistency of results.
The study had >99% power to detect a 3.5% difference between alendronate and placebo in mean percentage change from baseline in BMD of the lumbar spine, assuming a standard deviation of 4.5%. All treatment comparisons were two-sided, and statistical significance was defined as p<0.05.
Results
Of 1908 randomized patients, 950 were assigned to treatment with alendronate and 958 to placebo. The two treatment groups were comparable with regard to patient demographic characteristics, BMD and biochemical markers of bone turnover at baseline (Table 1); 94% of randomized patients in each group were white. A total of 1697 (89%) patients completed the study: 832 (88%) in the alendronate groups and 865 (90%) in the placebo group.
Bone Mineral Density
Marked and progressive mean increases in BMD were recorded for alendronate-treated patients. Table 2 presents changes from baseline in BMD at 12 months. At all sites and at each time point, including at 3 months, mean percentage changes in BMD among patients in the alendronate group were significantly (p<0.001) greater than those among patients in the placebo group; changes at the lumbar spine are illustrated in Fig. 1. At 12 months the difference in least square means between alendronate and placebo groups in percentage changes from baseline in BMD was 4.9% (95% confidence interval 4.6% to 5.2%) at the lumbar spine, 2.4% (2.0% to 2.8%) at the femoral neck, 3.6% (3.2% to 4.1%) at the trochanter and 3.0% (2.6% to 3.4%) for the total hip.
The treatment-by-center and treatment-by-densitometer- type interactions were not significant, indicating that the treatment effect on BMD was consistent among investigators and independent of densitometer type.
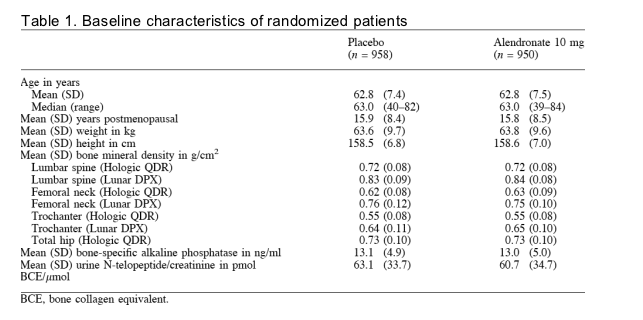
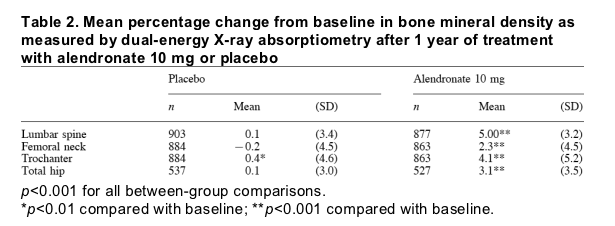
Fig. 1. Mean percentage changes from baseline and 95% confidence intervals for bone mineral density of the lumbar spine in women receiving alendronate 10 mg or placebo for 1 year (p50.001 for between-group comparisons at 3, 6 and 12
months).
Biochemical Markers of Bone Turnover
Both bone-specific alkaline phosphatase and urine Ntelopeptide/creatinine decreased significantly (p<0.001) compared with baseline in the alendronate and placebo groups (alkaline phosphatase by 52% vs 11%, respectively, and urine N-telopeptide/creatinine by 74% vs 21%, respectively, at 12 months). For both parameters, decreases were significantly (p<0.001) greater in the alendronate group than in the placebo group at all time points (data not shown). The reductions seen in the placebo group reflect the antiresorptive effect of calcium.
Clinical Features
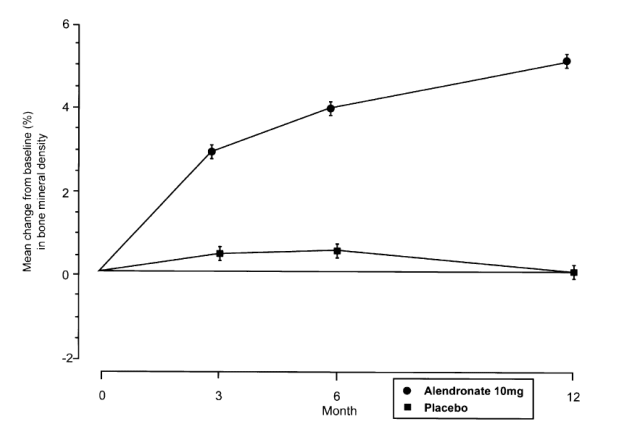
The risk of clinical nonvertebral fracture was significantly (p = 0.021) lower in the alendronate group than in the placebo group: 19 patients receiving alendronate experienced at least one nonvertebral fracture compared with 37 patients receiving placebo. The cumulative incidence of patients experiencing nonvertebral fracture is plotted in Fig. 2. The estimated cumulative incidence of nonvertebral fracture after 1 year was 2.4% in the alendronate group and 4.4% in the placebo group, yielding a relative hazard of 0.53 (95% confidence interval, 0.30 to 0.90). Figure 3 shows the percentage of women treated with alendronate or placebo, by fracture site, who experienced each specific type of nonvertebral fracture.
In all cases except one there was clear clinical or radiographic evidence, or both, supporting a definite fracture at the time the fracture was reported. The exception was a placebo-treated patient for whom the radiology report indicated that a malleolar fracture could not be excluded, but the radiograph did not allow for a definitive diagnosis. Analysis of the data excluding this fracture continued to support a significant reduction in nonvertebral fractures (p = 0.028). Since spine radiographs were not obtained before study entry, it was not possible to be certain that a compressed vertebra observed during the study represented a new fracture as opposed to a pre-existing fracture. Vertebral fractures, therefore, could not be evaluated.
Fig. 2. Cumulative proportion of patients with any clinical nonvertebral fracture. Alendronate treatment for 1 year was associated with a 47% reduction in the risk of nonvertebral fracture relative to placebo (95% confidence interval, 10% to 70%; p = 0.021).
Safety and Tolerability
Alendronate was generally well tolerated. No statistically significant differences between treatment groups were found in the overall incidence of adverse events (alendronate, 67.1%; placebo, 69.7%), the incidence of adverse events considered by the investigator to be possibly, probably or definitely drug-related (19.1% vs 18.0%), or the incidence of adverse events resulting in permanent discontinuation of study medication (6.4% vs 5.6%). Serious adverse events, specifically those resulting in hospitalization or permanent disability or cancers, were also equally common between groups (alendronate 6.5%; placebo 6.3%).
There were no significant differences between treatment groups in the overall incidence of upper gastrointestinal adverse events (alendronate, 21.3%; placebo, 19.3%) or of specific upper gastrointestinal adverse events such as abdominal pain, dyspepsia, or nausea or esophageal events (Table 3).
Discussion
One year of treatment with alendronate produced significant and progressive increases in BMD among patients enrolled in this large, multinational study. The effects observed were independent of study center. The increases recorded in BMD at the spine and hip are very similar to those observed with alendronate 10
mg at 12 months in previous studies. The weighted average effect on lumbar spine BMD in patients from four previous clinical, randomized studies [10,13,14]
was 5.06%, which is highly comparable to the mean 5.00% observed in this study (unpublished data on file, Merck & Co., Inc.). The consistency of response across studies is remarkable when the differences in geography (USA only, or multinational excluding USA) and patient definition are considered.
A major finding in this study is that alendronate 10 mg was associated with a significant decrease in the incidence of nonvertebral fractures over just 1 year of treatment. At the time this study was initiated there were no data available on potential fracture risk reduction associated with alendronate treatment. It was considered unlikely that a significant difference in clinical fractures could be demonstrated in such a short time period, as the expected number of events was low. Therefore, there was no formal hypothesis that nonvertebral fracture incidence would be reduced. Since that time, other alendronate studies have demonstrated decreases in vertebral and nonvertebral fracture incidence, albeit over a longer period of study. In each of these previous studies, time-to-event plots demonstrated a lower incidence of fractures in the alendronate group over the entire duration of the study (including the 1-year time point), although significance at 1 year has not previously been reported [10,15,16]. Moreover, the fracture incidence in the placebo group in this study was similar to that seen in prior studies. Therefore, the consistency of these data across studies provide compelling evidence to support fracture reduction with alendronate, even within the first year of therapy. The fact that all patients in the current study received the dose of alendronate that produces optimal effects on BMD (10 mg) from the outset may have contributed to the substantial (47%) risk reduction for nonvertebral fractures.
A limitation of the current study is that fractures were captured through adverse event reporting only and no vertebral radiographs were obtained; thus, the impact on radiographically (or morphometrically) defined vertebral fractures could not be assessed. However, the fact that the study population represents patients with BMD 2 SD or more below the young adult means without regard to presence or absence of previous vertebral fractures is in itself important to the generalizability of the results. Moreover, the findings of the current study are consistent with those of a meta-analysis of five prospective, randomized, placebo-controlled studies of alendronate treatment of osteoporosis, which found a 29% reduction in risk of nonvertebral fracture in patients treated with doses of 52.5 mg/day alendronate for 2 years or more [16].
Compared with previous alendronate trials, the subjects in the current study represent a relatively unselected group of patients, recruited on the basis of
low bone mass without regard to history of vertebral fracture. The vertebral fracture arm of the Fracture Intervention Trial [15] enrolled 2027 women with low BMD and pre-existing vertebral fractures at baseline. These women constitute a very high-risk population, as the presence of a vertebral fracture greatly increases the risk of future hip and vertebral fractures [17,18]. In the Fracture Intervention Trial, administration of alendronate 5 mg for 2 years followed by 10 mg in the third year reduced the incidence of morphometrically measured vertebral fracture by 47% and of any clinical fracture by 28%.
Alendronate was well tolerated in the present study. Adverse events (other than fractures), including upper gastrointestinal adverse events, occurred with similar incidence in the alendronate and placebo groups, a finding that is consistent with earlier studies [10,13-15]. This finding is, however, in contrast to the perception that alendronate may cause upper gastrointestinal complaints more frequently in general practice. One important consideration is that upper gastrointestinal complaints are common in the general population, particularly among women of this age range. Indeed, approximately 20% of patients in this study had complaints whether they were on placebo or alendronate, which is consistent with the incidence reported in a population-based survey in the USA [19].
Although there have been occasional reports of esophagitis associated with alendronate [20], no excess in esophageal adverse events was found in the present study. Moreover, patients were not excluded from the study for a history of gastrointestinal disease, excepting those with major gastrointestinal disease during the past year. All study participants were instructed to take study medication with a full glass of water and to refrain from lying down for 30 min afterwards, consistent with the recommendations in the product label.
We conclude that alendronate is well tolerated and produces significant and progressive increases in BMD at both the lumbar spine and hip among a diverse population of postmenopausal women with osteoporosis defined on the basis of low spine BMD without regard to prior fracture history. This is the first study in which treatment with 10 mg alendronate is associated with a significant reduction in nonvertebral fracture after only 1 year of therapy.
References
1. Melton LJ III. How many women have osteoporosis now? J Bone Miner Res 1995;10:175-7.
2. Kanis JA, Melton LJ III, Christiansen C, Johnston CC, Khaltaev N. The diagnosis of osteoporosis. J Bone Miner Res 1994;9:1137-41.
3. Melton LJ III, Chrischilles EA, Cooper C, Lane AW, Riggs BL. Perspective. How many women have osteoporosis? J Bone Miner Res 1992;7:1005-10.
4. Riggs BL, Melton LJ III. The prevention and treatment of osteoporosis. N Engl J Med 1992;327:620-7.
5. Garnero P, Shih WJ, Gineyts E, Karpf DB, Delmas PD. Comparison of new biochemical markers of bone turnover in late-postmenopausal osteoporotic women in response to alendronate treatment. J Clin Endocrinol Metab 1994;79:1693-700.
6. Guy JA, Shea M, Peter CP, Morrissey R, Hayes WC. Continuous alendronate treatment throughout growth, maturation, and aging in the rat results in increases in bone mass and mechanical properties. Calcif Tissue Int 1993;53:283-8.
7. Balena R, Markatos A, Seedor JG, et al. Long-term safety of the aminobisphosphonate alendronate in adult dogs. II. Histomorphometric analysis of the L5 vertebrae. J. Pharmacol Exp Ther 1996;276:277-83.
8. Balena R, Toolan BC, Shea M, et al. The effects of 2-year treatment with the aminobisphosphonate alendronate on bone metabolism, bone histomorphometry, and bone strength in ovariectomized nonhman primates. J Clin Invest 1993;92:2577-86.
9. Chavassieux PM, Arlot ME, Reda C, Wei L, Yates AJ, Meunier PJ. Histomorphometric assessment of the long-term effects of alendronate on bone quality and remodeling in patients with osteoporosis. J Clin Invest 1997;100:1475-80.
10. Liberman UA, Weiss SR, BroÅNll J, et al. Effect of oral alendronate on bone mineral density and the incidence of fractures in postmenopausal osteoporosis. N Engl J Med 1995;333:1437-43.
11. Garnero P, Delmas PD. Assessment of the serum levels of bone alkaline phosphatase with a new immunoradiometric assay in patients with metabolic bone disease. J Clin Endocrinol Metab 1993;77:1046-53.
12. Hanson DA, Weiss MAE, Bollen AM, Maslan SH, Singer FR, Eyre DR. A specific immunoassay for monitoring human bone resorption: quantitation of type I collagen crosslinked Ntelopeptides in urine. J Bone Miner Res 1992;7:1251-8.
13. Chesnut CH III, McClung MR, Ensrud KE, et al. Alendronate treatment of the postmenopausal osteoporotic woman: effect of multiple dosages on bone mass and bone remodeling. Am J Med 1995;99:144-52.
14. Adami S, Passeri M, Ortolani S, et al. Effects of oral alendronate and intranasal salmon calcitonin on bone mass and biochemical markers of bone turnover in postmenopausal women with osteoporosis. Bone 1995;17:383-90.
15. Black DM, Cummings SR, Karpf DB, et al. Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Lancet 1996;348:1535-41.
16. Karpf DB, Shapiro DR, Seeman E, et al. Prevention of nonvertebral fractures by alendronate: a meta-analysis. JAMA 1997;277:1159-64.
17. Ross PD, Davis JW, Epstein RS, Wasnich RD. Pre-existing fractures and bone mass predict vertebral fracture incidence in women. Ann Intern Med 1991;114:919-23.
18. Kotowicz MA, Melton LJ III, Cooper C, Atkinson EJ, O'Fallon WM, Riggs BL. Risk of hip fracture in women with vertebral fracture. J Bone Miner Res 1994;9:599-605.
19. Talley NJ, Weaver AL, Zinsmeister AR, Melton LJ III. Onset and disappearance of gastrointestinal symptoms and functional gastrointestinal disorders. Am J Epidemiol 1992;136:165-77.
20. de Groen PC, Lubbe DF, Hirsch LJ, et al. Esophagitis associated with the use of alendronate. N Engl J Med 1996;335:1016-21.
|
|
| |
| |
|
|
|