 |
 |
 |
| |
Clinical Predictors of Tenofovir-Associated Nephrotoxicity
|
| |
| |
IMPORTANT CORRECTION
The authors of this study presented at the Intl AIDS Conference in Mexico City contacted NATAP last night and others as well to inform us they made a error in calculation regarding the percent treated or not treated with tenofovir who experienced nephrotoxicity. The original slide presentation at the conference reported 7.5% who were taking TDF experienced nephrotoxicity compared to 4.2% who were not taking TDF & experienced nephrotoxicity, and the p-value was 0.052, which is they had stated was of borderline significance. BUT last night I received an email from the authors making a correction, that was not noticed by them or any observers until after the meeting. The actual percentage is 4.7% (35/744) for TDF-receiving patients who experienced nephrotoxicity and 4.2% for those not taking TDF who experienced nephrotoxicity. So it appears to be a mistake in math because if you divide 35 by 744 you actually get 4.7% not 7.5%, but apparently no one noticed this until after the meeting. Now, while the incorrect originally presented numbers were not significant it did make a certain impact that 7.5% appeared greater than 4.2% and the p-value was close to significance. However, now there appears to be little difference (4.7% vs 4.2%) in rates of nephrotoxicity between those taking or not taking TDF. Sorry for the inconvenience, but this is important information so I'm pleased it is corrected. If we receive any infurther notice from the authors about the study I will forward it to you.
Jules Levin
Concurrent Nephrotoxic Drugs, PI Use and Hypertension Inflate Risk of Kidney Toxicity Among Patients on Tenofovir
XVII International AIDS Conference
August 3-8, 2008
Mexico City
Mark Mascolini
From Jules: This study is a pilot study, although a large one with 1600 patients at Duke. The purpose is to identify factors that might affect the patientŐs kidney and potential kidney disease. So the study is trying to identify factors such as use of concurrent meds that might increase the risk for affecting kidney function so the clinician can be assisted by these study findings in making treatment decisions. For example, if a patient is on certain drugs that may cause nephrotoxicity, or if a patient has diabetes or hypertension, or if a patient is on an ace inhibitor, then the clinician can use this information in deciding perhaps I should remove the ace inhibitor and substitute another drug or perhaps I should not use a protease inhibitor but use a NNRTI instead, or perhaps I should not use tenofovir, or if the clinician wants to use tenofovir then perhaps they should minimize or try to eliminate the other drugs that can contribute to nephrotoxicity. But in the end the results of this study are intended to help the clinician identify factors or drugs that can contribute to nephrotoxicity so the clinician can make treatment decisions that can help to prevent or minimize the risk for nephrotoxicity. As I said this is a pilot study and the researchers intend to continue and expand this study so I think interesting and I expect very helpful information will come from this research. Also, an important note. You will see in this report and in the slides following that 20 patients discontinued tenofovir and 15 remained on tenofovir. Of the 20 16 were on nephrotoxic drugs, and of the 15 who continued on tenofovir 14 were on nephrotoxic drugs. As well, 19/20 in the TDF discontinuation group were treatment experienced, and 12/15 who continued TDF were treatment-experienced (however, for 2 patients in this group, it was unlear from the records if the patients had been on ARV previously).
The purpose of this study was to identify risk factors for developing kidney disease so if tenofovir is being considered for a patient the risk factors identified in this study can be scrutinized before taking TDF, so an alternative regimen can be considered you can address the risk factors such as the nephrotoxic drugs the patient may be taking. In this study clearly a significant number of patients were treatment experienced and taking nephrotoxic drugs along with tenofovir.
Taking tenofovir with a protease inhibitor (PI) almost quadruples the risk of kidney toxicity, according to results of a large case-control study [1]. But taking a nonnucleoside with tenofovir cut the risk by two thirds. Hypertension, nephrotoxic drugs, other medical conditions, and a history of opportunistic infection also heightened kidney toxicity risk in tenofovir takers.
Randomized controlled trials involving tenofovir are an imperfect gauge of kidney toxicity rates, suggested Chelsea Castellano and Duke University colleagues, because they limit enrollment to people with normal renal function and do not have the statistical power to discriminate between side effect rates in different treatment arms.
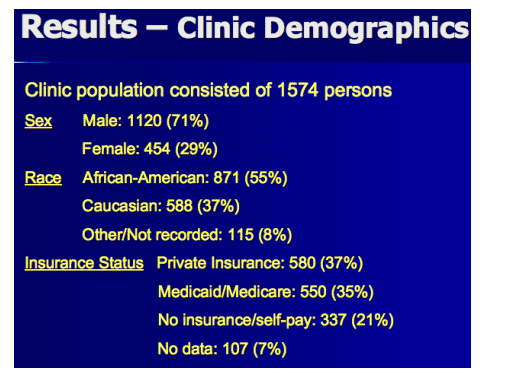
The Duke group analyzed kidney toxicity trends in 744 HIV clinic patients (47%) who took tenofovir for more than 3 months and had their creatinine measured less than 1 year before starting tenofovir. From the 830 clinic patients who never took tenofovir (53%), the investigators randomly selected 191 controls who had taken antiretrovirals for at least 1 year. People with renal toxicity did not differ from controls in age, gender, race, or type of insurance.
Castellano and colleagues defined nephrotoxicity as (1) more than a 50% drop in glomerular filtration rate or (2) an absolute drop in creatinine clearance of 25 mL/min or more. The researchers added the second definition to include people who start tenofovir with high creatinine clearance.
These criteria identified 35 tenofovir takers (7.5% of 744) with nephrotoxicity, 20 (57% of 35) of whom had to stop tenofovir. Kidney function improved significantly in 16 of those 20 (80%) after they eliminated tenofovir from their regimen. Of the 15 people with renal toxicity who continued tenofovir, 10 (67%) had persistently abnormal renal function. Most of these people did not lower their tenofovir dose. Among the 191 controls, only 8 (4.2%) met toxicity criteria, a rate that differed from the tenofovir group rate by a nearly significant margin (P = 0.052).
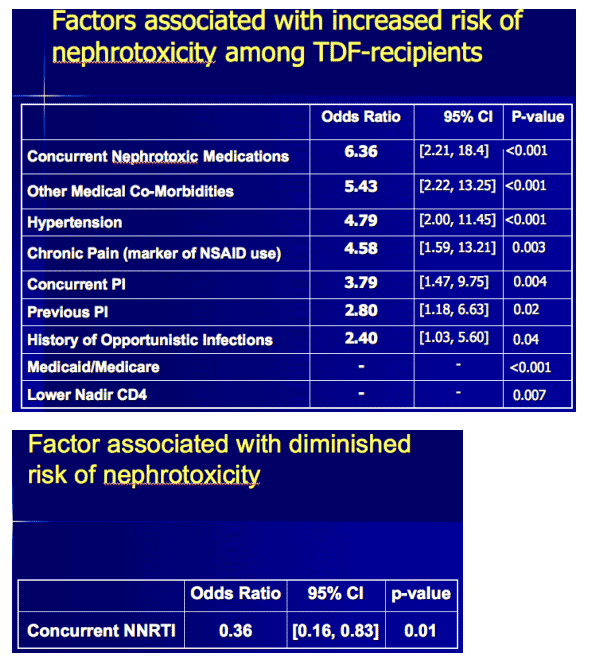
Comparing the kidney-impaired group with 74 randomly selected tenofovir takers without kidney toxicity, the Duke team identified seven independent predictors of toxicity at the following odds ratios (OR) and 95% confidence intervals (CI):
- Current nephrotoxic drug use: OR 6.36, 95% CI 2.21 to 18.4, P < 0.001
- Other medical conditions: OR 5.43, 95% CI 2.22 to 13.25, P < 0.001
- Hypertension: OR 4.79, 95% CI 2.00 to 11.45, P < 0.001
- Chronic pain (a marker of nonsteroidal antiinflammatory use): OR 4.58, 95% CI 1.59 to 13.2, P = 0.003
- Current PI use: OR 3.79, 95% CI 1.47 to 9.75, P = 0.004
- Previous PI use: OR 2.80, 95% CI 1.18 to 6.63, P = 0.02
- History of opportunistic infection: OR 2.40, 95% CI 1.03 to 5.60, P = 0.04
Taking a nonnucleoside during tenofovir therapy independently lowered the risk of kidney toxicity 64% (P < 0.01).
Castellano noted that limiting the analysis to people who took tenofovir for at least 3 months could result in an underestimate of kidney toxicity because some patients may stop the drug for that very reason within 3 months. The Duke investigators recommended avoiding tenofovir in people taking nephrotoxic drugs for hypertension, especially if they are also taking a PI.
Reference
1. Castellano C, Williams W, Kepler TB, et al. Clinical predictors of tenofovir-associated nephrotoxicity in HIV-1-infected patients. XVII International AIDS Conference. August 3-8, 2008. Mexico City. Abstract WEAB0104.

1. Gallant JE, DeJesus E, Arribas JR, et al. Tenofovir DF, emtricitabine, and efavirenz vs zidovudine, lamivudine, and efavirenz for HIV.NEngl J Med. 2006;354:251-260.
2. Saag MS, Cahn P, Raffi F, et al. Efficacy and safety of emtricitabine vs stavudine in combination therapy in antiretroviral-naive patients: a randomized trial. JAMA. 2004;292:180-189.
3. ACTG 5202 clinical trial
4.Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. Department of Health and Human Services. January 29, 2008; 1-128.

3. Cheng A, Barriere S, Coakley DF et al. Safety profile of tenofovir DF in antiretroviral-experienced patients from randomized, double-blind, placebo-controlled clinical trials. Paper presented at the Nineth Conference on Retroviruses and Opportunistic Infections. Seattle, WA; 2002 Feb 27.


Demographic and clinical information for all HIV-1 infected patients being cared for at the Duke HIV clinic are stored in a clinic database (DART) for clinical and quality improvement purposes. This database will be used to determine the number of patients seen at the clinic for at least 3 months receiving tenofovir treatment within the time frame of October 2001 (when tenofovir was FDA-approved) to the present

The first consensus definition of acute renal failure (ARF) has now been developed. It is called RIFLE, the initials reflecting the terms Risk, Injury, Failure, Loss and End Stage in relation to kidney function. It is presented in this article.
The body responsible for the consensus definition of ARF is the Acute Dialysis Quality Initiative (ADQI) group, which conducted a consensus conference for this purpose.
Rinaldo Bellomo1, Claudio Ronco2, John A Kellum3, Ravindra L Mehta4, Paul Palevsky5 and the ADQI workgroup6
Critical Care 2004, 8:R204-R212 (DOI 10.1186/cc2872)
2. added to make it more sensitive to those that start out at normal kidney function. This latter criterion was added to capture significant changes in renal function among persons who initiated TDF w/ high CrCl.

Generated a list of potentially nephrotoxic drugs from two previously published papers from clinical infectious diseases regarding renal dysfunction in HIV-infected patients.


for most patients it was not one single isolated measurement of Cr that was elevated; most patients had multiple measurements of serum Cr Đ makes lab error or spurious finding less likely.


|
| |
|
 |
 |
|
|