 |
 |
 |
| |
CD4+ Cell Restoration after 48 Weeks in the Maraviroc Treatment-experienced Trials MOTIVATE 1 and 2
|
| |
| |
Reported by Jules Levin
IAC Mexico City Aug 3-8, 2008
D Asmuth1, J Goodrich2, D Cooper3, R Haubrich4, N Rajicic5, H Valdez5, B Hirschel6, and H Mayer2
1University of California Davis, Davis, USA; 2Pfizer Inc, New London, USA; 3University of New South Wales, Sydney, Australia; 4University of California San Diego, San Diego, USA;
5Pfizer Inc, New York, USA; 6Hospital Geneva University Hospital, Geneva, Switzerland
AUTHOR CONCLUSIONS
In MOTIVATE 1 and 2, highly TE patients receiving MVC (QD or BID) + OBT experienced early, rapid, greater, and persistent CD4+ cell count increases vs those receiving PBO + OBT (P=0.0182).
The CD4+ cell count advantage of MVC over PBO was driven not only by those patients who achieved undetectable HIV-1 RNA (<50 copies/mL at least once on therapy), but also by those who never achieved undetectable HIV-1 RNA. In addition, since treatment group was associated with CD4+ cell rises, these findings suggest that MVC increases CD4+ cell counts above and beyond what is expected for a given viral load reduction.
Greater early and rapid CD8+ cell count rises were also observed in the MVC arms vs the PBO arm (P<0.0001). CD8+ cell numbers subsequently declined in all three arms but had not returned to baseline by Week 48 in patients receiving MVC.
Predictors of CD4+ cell count rises among MVC-treated patients were similar to those observed among non-MVC-treated patients. Tropism at screening was also associated with CD4+ cell rises in MVC recipients.
MVC dose did not appear to influence CD4+ cell count outcome. Patients receiving MVC equivalent doses of 300 mg QD or 300 mg BID achieved
similar increases in CD4+ cell count after adjusting for other predictive variables.
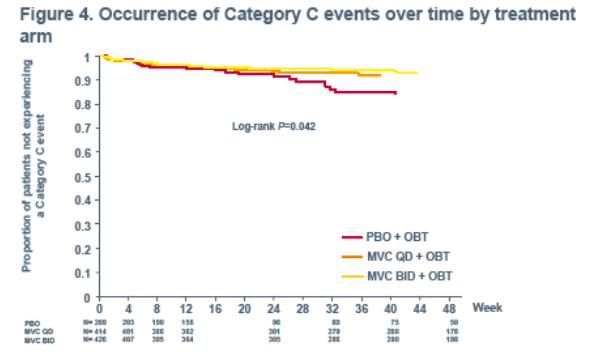
The time to Category C clinical event occurrence was significantly increased on treatment with MVC (QD and BID) + OBT vs PBO + OBT (P=0.042), which may be related to improved virologic suppression, additional CD4+ cell count gains, or both.
"Significantly greater (103 vs 24 CD4s) and faster increases in CD4+ cell count were observed in patients receiving MVC vs PBO (P=0.0182), with the majority of the difference established within the first 12 weeks of treatment. The early
MVC-associated increases in CD4+ cell count were sustained through
Week 48......In patients who achieved HIV-1 RNA <50 copies/mL at least once on therapy, increases in CD4+ cell count were significantly higher with MVC at
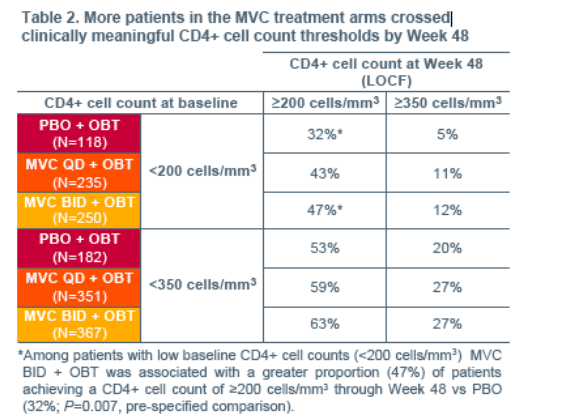
Week 48 vs PBO (P<0.05), suggesting that an effect in addition to the control of viral load was partially responsible for the CD4+ cell count rise observed in MVC recipients..... Among patients with low baseline CD4+ cell counts (<200 cells/mm3) MVC BID + OBT was associated with a greater proportion (47%) of patients achieving a CD4+ cell count of >/=200 cells/mm3 through Week 48 vs PBO (32%; P=0.007, pre-specified comparison)...... Even after accounting for viral load reduction, MVC treatment remained associated with CD4+ cell count increases..... In addition, R5 tropism at screening was associated with gains in CD4+ cell count..... The time to occurrence of a Category C event for patients receiving MVC (BID or QD) was significantly longer than for patients receiving PBO. ...... early CD8+ cell increases were observed in all three study arms, but were significantly greater (P<0.0001) in patients receiving MVC. In the PBO arm this early increase declined back to baseline by Week 24. CD8+ cell count levels in the MVC arms declined at a similar rate, but due to the greater early CD8+ cell count increases, did not return to baseline through 48 weeks."
BACKGROUND
Treatment-related increases in CD4+ cell count may be associated with
clinical benefits beyond decreasing the occurrence of AIDS-defining
illnesses:
-- HIV-infected patients who achieve CD4+ counts of >500 cells/mm3
after more than 6 years of HAART have a survival rate similar to that
of HIV-negative subjects.1
-- The risk of non-AIDS-defining malignancies is greater for patients with
prolonged periods of CD4+ count <200 cells/mm3 on treatment.2
MOTIVATE 1 and 2 are randomized, double-blind, placebo (PBO)-
controlled, Phase 3 studies of the CCR5 antagonist maraviroc (MVC), in
treatment-experienced (TE) patients with CCR5-tropic (R5) HIV-1.3
In pooled data from both studies (N=1,049), MVC once daily (QD) or twice
daily (BID) with an optimized background therapy (OBT) demonstrated
significantly better virologic suppression (P<0.0001) at 24 and 48 weeks
than PBO + OBT and a similar safety profile.3
For the primary endpoint of change from baseline in HIV-1 RNA at
Week 48, both MVC QD and BID were superior to PBO (-1.68 log10 copies/mL [difference vs PBO -0.87; 97.5% CI: -1.17, -0.62] and -1.84 log10 copies/mL [-1.05; -1.33, -0.78], respectively).3
For the secondary endpoint of proportion of patients with HIV-1 RNA <50 copies/mL at Week 48, MVC QD (43%) and MVC BID (46%) were also superior to PBO (17%; P<0.0001).3
A meta-analysis of 16 recent clinical studies in TE patients (including four CCR5 antagonist studies) showed an additional rise in CD4+ count of 32 cells/mm3 over 24 weeks in patients receiving CCR5 antagonists.4
The basis, predictors, and clinical benefit of this additional CD4+ cell count increase in TE patients receiving CCR5 antagonists are unknown.
OBJECTIVES
To describe CD4+ cell count rises after MVC administration to TE patients
and to identify predictors of CD4+ cell count restoration in TE patients
receiving MVC vs PBO in pooled data from the MOTIVATE 1 and 2 studies
at 48 weeks.
METHODS
∗ Missing CD4+ cell count values were handled using last observation carried forward (LOCF).
∗ Statistical methods included:
∗ Descriptive summaries (median; 25th, 75th percentiles).
∗ Shift table of number of subjects exceeding clinically meaningful CD4+ cell counts; P-value from a Wilcoxon Rank Sum test on medians for one pre-specified comparison of interest.
∗ Longitudinal model of inferred CD4+ and CD8+ cell count values over time, with effects of time and treatment in the model.
∗ Multivariate linear regression models to analyze CD4+ cell count change from baseline to Week 48, using the following variables:
∗ MOTIVATE 1 and 2: Baseline CD4+ cell count and HIV-1 RNA, HIV-1 RNA change from baseline to Week 48, age, gender, and treatment group (MVC arms vs PBO).
∗ MOTIVATE 1 and 2 plus Phase 2b study A4001029 (TE patients with non-R5 virus; model used for MVC recipients only): Baseline CD4+ cell count and HIV-1 RNA, HIV-1 RNA change at Week 48, average MVC concentration, MVC dose, age, gender, and tropism result (R5 vs non-R5). These analyses included MVC
recipients only to ascertain effects of tropism and MVC concentration on CD4+ cell count response.
∗ Cox proportional hazard regression model to analyze time to AIDSdefining (Category C) event, using the following variables:
∗ Baseline HIV-1 RNA, time-dependent CD4+ cell count values, age, gender, and treatment group (MVC arms vs PBO).
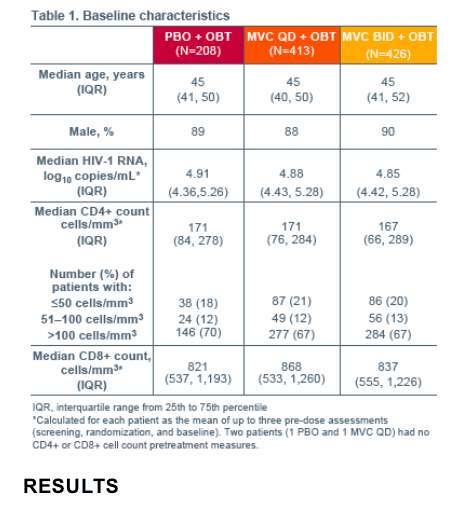
Significantly greater and faster increases in CD4+ cell count were observed
in patients receiving MVC vs PBO (P=0.0182), with the majority of the
difference established within the first 12 weeks of treatment. The early
MVC-associated increases in CD4+ cell count were sustained through
Week 48.
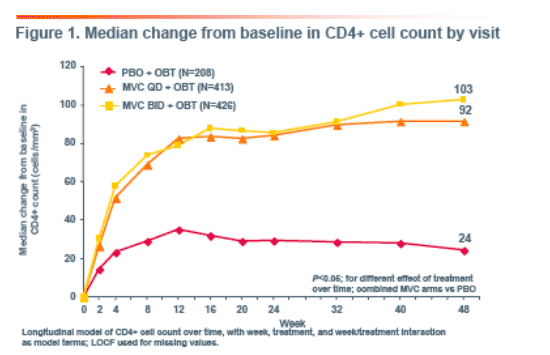
In patients who achieved HIV-1 RNA <50 copies/mL at least once on
therapy, increases in CD4+ cell count were significantly higher with MVC at
Week 48 vs PBO (P<0.05), suggesting that an effect in addition to the control of viral load was partially responsible for the CD4+ cell count rise observed in MVC recipients. In patients with persistent detectable viremia (never achieved HIV-1 RNA <50 copies/mL) significantly greater early CD4+ cell count increases were observed in those receiving MVC vs PBO which were preserved through 48 weeks (P=0.0013).
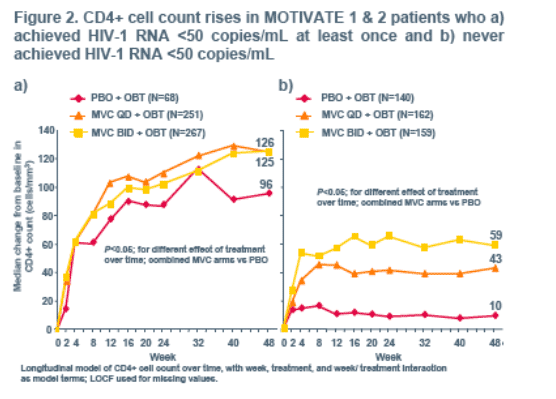
Early CD8+ cell increases were observed in all three study arms, but were
significantly greater (P<0.0001) in patients receiving MVC. In the PBO arm this early increase declined back to baseline by Week 24. CD8+ cell count levels in the MVC arms declined at a similar rate, but due to the greater early CD8+ cell count increases, did not return to baseline through 48 weeks.
Predictors of CD4+ cell count rises among all patients
A multivariate regression model of predictors of Week-48 CD4+ cell count
increases from baseline identified significant associations with:
Baseline CD4+ cell count: Higher baseline CD4+ cell counts were
associated with greater increases in CD4+ cell count (P=0.0007).
Baseline HIV-1 RNA level: Higher baseline HIV-1 RNA levels were
associated with greater increases in CD4+ cell count (P=0.0003).
Week-48 change in HIV-1 RNA level: Larger decreases from
baseline in HIV-1 RNA at Week 48 were associated with greater
increases in CD4+ cell count (P<0.0001).
MVC treatment: Least-squares estimates of Week-48 CD4+ cell count increase were 110 cells/mm3 in the combined MVC arms vs 85 cells/mm3 in the PBO arm (P=0.002). Even after accounting for viral load reduction, MVC treatment remained associated with CD4+ cell count increases.
Age: Older age was associated with smaller increases in CD4+ cell
count (P<0.0001).
Other than MVC treatment, all these factors have been associated with CD4+ cell count increases in previous studies of HIV-infected patients. Gender (P=0.0518) and gender/age interaction were not significantly associated in this model.
The finding that MVC treatment is associated significantly with CD4+ cell count outcome after adjusting for viral load change again suggests an effect above and beyond the antiviral effect.
Predictors of CD4+ cell count rises among MVC recipients
Similar modeling in only those patients receiving MVC in MOTIVATE 1 and
2 and in the Phase 2b A4001029 study identified the same significant associations as above, with the exception that male gender was also significantly associated with greater Week-48 CD4+ cell count increases. In addition, R5 tropism at screening was associated with gains in CD4+ cell count (P=0.042). By contrast, MVC dose (300 vs 600 mg/day) and average MVC concentration were not significantly associated with CD4+ cell count outcome.
Predictors of Category C events
Significant predictors of time to Category C event occurrence (Cox
proportional hazard modeling) were:
-- Baseline HIV-1 RNA (per log10 increase): HR=2.051 (P<0.001)
-- Time-dependent CD4+ cell count (per 50 cell/mm3 increase): HR=0.679 (P<0.001)
-- Sex (female vs male): HR=1.829 (P=0.042)
-- Baseline CD4+ cell count and ever achieving <50 HIV-1 RNA copies/mL were not significant predictors of time to Category C event.
-- Due to the association between MVC treatment and greater CD4+ cell
counts on therapy, treatment modality (MVC vs PBO) was only a significant predictor of time to Category C event when time-adjusted CD4+ cell count was not included in the model.
REFERENCES
1. Lewden C, et al. J Acquir Immune Defic Syndr 2007; 46:72-77.
2. Bruyand M, et al. 15th CROI 2008; Abstract 15.
3. Hardy D, et al. 15th CROI 2008; Poster 792.
4. Wilkin T, et al. 15th CROI 2008; Poster 800.
|
| |
|
 |
 |
|
|