 |
 |
 |
| |
Maraviroc Exposure-safety Biomarker (Transaminases and Creatine Kinase) Analyses in MOTIVATE 1 and 2 Studies in HIV-1-infected Treatment-experienced Subjects
|
| |
| |
Reported by Jules Levin
IAC Mexico City Aug 3-8 2008
L McFadyen1, P Jacqmin2, JR Wade2, and B Weatherley1
1Pharmacometrics, Pfizer, PGRD, Sandwich, UK; 2Exprimo NV, Mechelen, Belgium
BACKGROUND
Maraviroc (MVC), a CCR5 antagonist, has been approved for combination treatment of HIV-1-infected treatment-experienced subjects with exclusively CCR5-tropic (R5) virus. MVC plus Optimized Background Therapy (OBT) has shown statistically superior virologic suppression and improvement in CD4 count
compared with OBT plus placebo (PBO) in the MOTIVATE 1 and 2 studies. Favorable efficacy has been sustained through 48 weeks on treatment [1,2].
No clinically relevant differences in safety or tolerability have been observed between the MVC+OBT arms and the OBT+PBO arms in the MOTIVATE studies [1].
No imbalance between arms in the incidence of liver enzyme abnormalities has been found in the MOTIVATE studies, even before correction for duration of exposure [3]. However concerns about the hepatic safety of CCR5 antagonists as a class have been raised subsequent to the termination of the development of aplaviroc. Aplaviroc was associated with idiosyncratic hepatic toxicity [4].
Creatine kinase (CK) is of interest in this analysis because of reports of myalgia and increased CK in the MOTIVATE studies. These are difficult to interpret due to differences in time on treatment between the MVC+OBT and OBT+PBO groups.
OBJECTIVE
To develop regression models assessing the relationship (if any) of exposure of MVC to change from baseline (cfb) in transaminases (ALT and AST) and CK at weeks 4 and 24, in the context of other potential prognostic factors.
AUTHOR CONCLUSIONS
Hepatic safety biomarkers:
-- No MVC concentration-related relationships were identified for ALT.
-- For AST at week 24, higher MVC exposure was associated with a small reduction from baseline.
For CK the only prognostic factor identified was baseline CK.
Baseline values appeared to be the strongest predictors of change from baseline for AST, ALT and CK at weeks 4 and 24. Higher baselines values were associated with decreases from baseline at 4 and 24 weeks in study.
ABSTRACT
Background: Maraviroc (MVC), a CCR5 antagonist, has been approved for combination treatment of HIV-1-infected subjects with exclusively CCR5-tropic virus. The studies MOTIVATE 1 and 2 compared MVC 300mg (adjusted to 150mg for interactions) once daily, twice daily and placebo (2:2:1) with optimised background therapy (OBT). Although MVC has an acceptable safety and tolerability profile relative to placebo there were concerns about the hepatic safety of CCR5 antagonists as a class. Therefore, analyses of the influence of MVC exposure and other prognostic factors on change from baseline (cfb) in hepatic transaminases (ALT, AST) were conducted. Also creatine kinase (CK) was similarly assessed because of reports of myalgia and a higher median change seen in MVC groups compared with placebo.
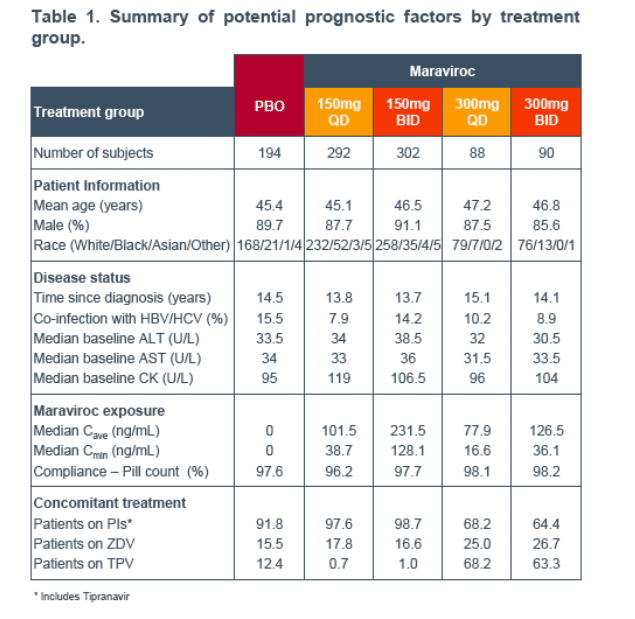
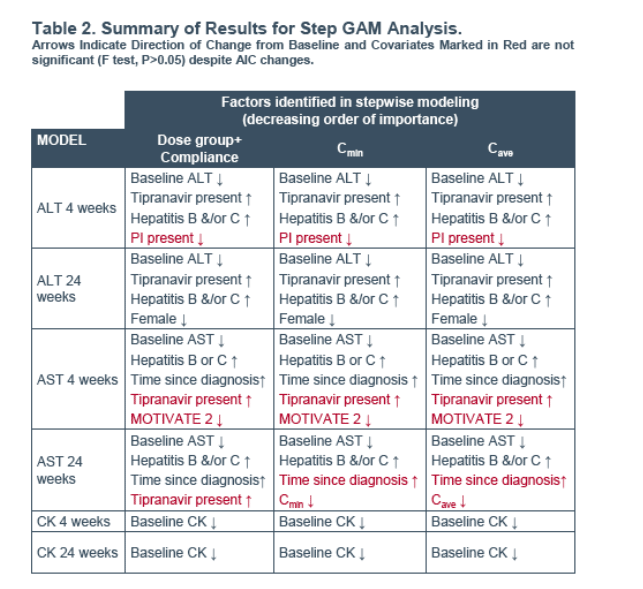
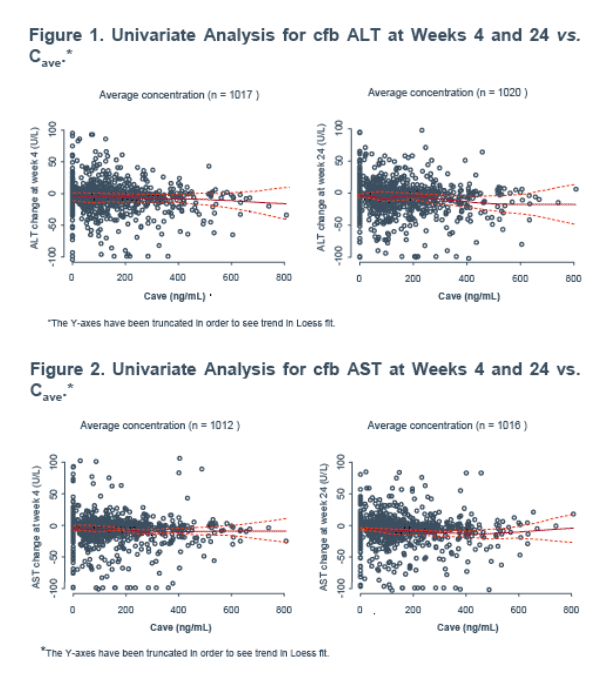
Methods: Graphical analysis and step-wise generalised additive modeling (GAM) were used to investigate prognostic factor influences on cfb in ALT, AST and CK at weeks 4 and 24 (LOCF). Prognostic factors included exposure (dose group, Cave or Cmin), demographics (age, gender, race), disease (time since diagnosis, hepatitis B or C) and presence of particular drugs/drug classes. Exposure variables were derived from population modeling of sparse outpatient PK sampled up to 24 weeks or discontinuation. In the pooled studies, 966 subjects had on-treatment laboratory and covariate information.
Results: No exposure-related relationships were identified for ALT. For AST, higher MVC exposure was associated with a reduction from baseline at week 24. For CPK no exposure-related changes were identified. For all three safety biomarkers a higher baseline was predictive of changes from baseline.
Conclusions: For the safety biomarkers tested no significant exposure-related increases from baseline were identified at weeks 4 and 24.
STUDY DESIGN
MOTIVATE 1 and 2 are identical Phase 3 studies comparing MVC (once and twice daily) plus OBT with OBT+PBO in treatment experienced, R5 HIV-1 infected subjects. The studies differed only in the regions in which they were conducted.
The studies were designed to allow patients with limited treatment options to enroll and therefore the exclusion criteria for hepatic enzymes were set at >5 x ULN for transaminases and >2.5 x ULN for bilirubin. Patients co-infected with hepatitis B and/or C virus could be enrolled.
Maraviroc doses were adjusted down from 300mg to 150mg in subjects taking potent CYP3A4/Pgp inhibitors in their OBT regimens to maintain Cmax values not exceeding the range observed with 300mg MVC in the absence of inhibitors. However, this did not fully correct for area under the curve with more potent inhibitors, therefore a wide range of exposures was observed [5].
Two samples for pharmacokinetic (PK) analysis were taken at weeks 2 and 24 with single samples at all visits from 4 to 20 weeks (up to 9 samples/patient).
METHODS
Individual PK parameters were obtained from population pharmacokinetic modeling of the sparse PK concentration versus time after dose data [5]. MVC exposure variables (Average Concentration [Cave; AUC/dose interval] and Cmin) were calculated from post hoc PK parameters for each subject.
Data analysis:
An exploratory univariate analysis (biomarker cfb versus Cave or Cmin) was performed using a Loess fit in S-PLUS (GAM function) for each biomarker at weeks 4 and 24.
Thereafter a step-wise generalized additive modeling procedure (GAM in S-PLUS) was used to investigate potential prognostic factors on cfb on ALT, AST and CK at weeks 4 and 24.
-- Three exposure models (dose group plus compliance or Cmin or Cave) were run for each biomarker at 4 and 24 weeks.
-- A decrease in the Akaike Information Criterion (AIC) was used for identifying significant factors.
Potential prognostic factors tested in addition to exposure/dose group in the GAM analysis:
-- Study (MOTIVATE 1 or 2)
-- Age, sex, race, hepatitis B and/or C (for ALT and AST), time since diagnosis of HIV, time since first treatment of HIV
-- Baseline value of biomarker
-- Presence of specific ARVs or ARV classes: non-nucleosides, nucleosides/tides, protease inhibitors (PI), enfuvirtide, tipranavir (TPV) (for ALT and AST only), zidovudine (ZDV) (for CK only)
Data:
Full data including all potential prognostic factors were available in 966 patients from the full analysis, intent-to-treat population. Patients without any MVC PK data or missing covariates were excluded from this analysis.
For patients who did not have safety biomarker data for weeks 4 or 24, a last-observation-carried-forward procedure was used.
RESULTS
Univariate analysis
No trends with exposure (Cmin or Cave) were identified for any of the endpoints despite the wide MVC exposure range (Figures 1, 2 and 3).
Stepwise modeling
Exposure (Cmin or Cave) was identified by a small change in AIC in the
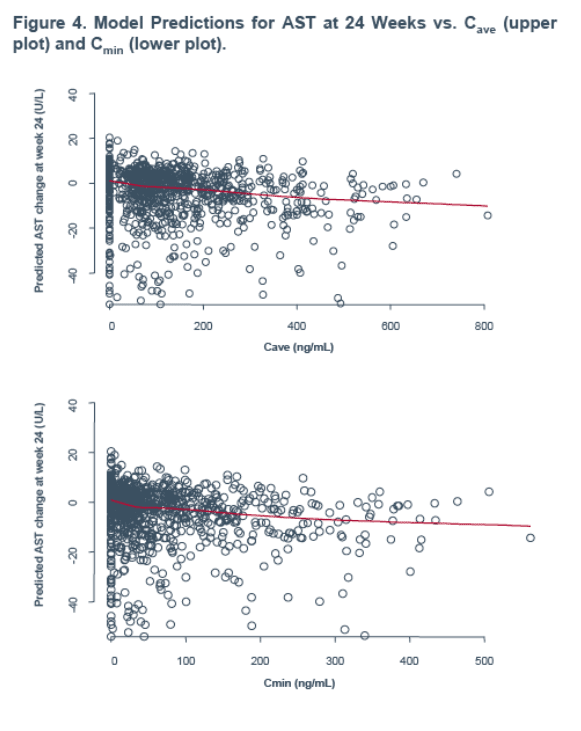
model for AST at 24 weeks where increased exposure was associated with a small decrease from baseline in AST values (Figure 4).
All endpoints (AST, ALT and CK at weeks 4 and 24) showed a strong relationship (decrease) with baseline values at both 4 and 24 weeks.
For transaminases, co-infection with hepatitis B and/or C and the presence of tipranavir were identified as independent predictive factors for increases from baseline across treatment groups in most models (Table 2).
|
| |
|
 |
 |
|
|