 |
 |
 |
| |
Week-96 end of trial analysis of antiretroviral-naive patients randomized to the lopinavir/ritonavir single drug arm in the MONARK trial
|
| |
| |
Reported by Jules Levin
IAC Mexico City Aug 3-8 2008
J. Ghosn1-3, P. Flandre2, C. Delaugerre3, M.-L. Chaix3, P. Dellamonica4, R.A. Rode5,
Y. Wang5, M. Norton5, I. Cohen-Codar6, P. NgoVan6, C. Rouzioux3,
and JF. Delfraissy1 for the MONARK Study Group
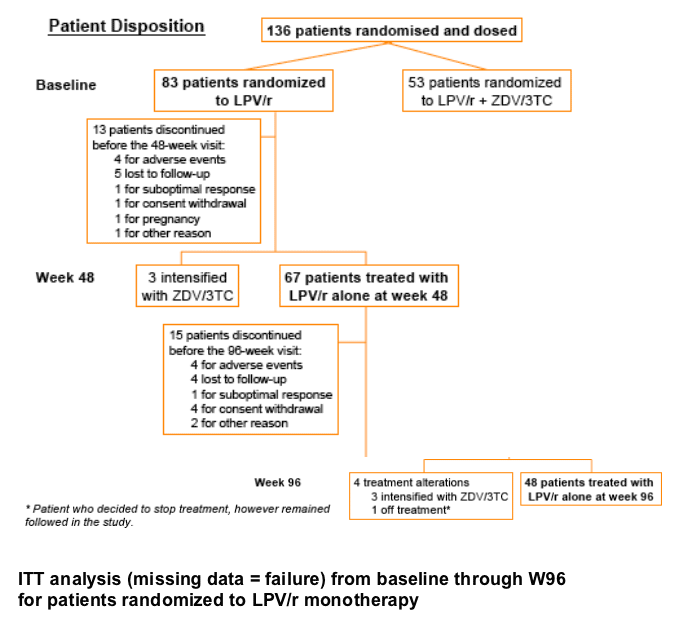
The MONARK trial enrolled 136 antiretroviral-naive patients who were
randomized and dosed with either lopinavir/ritonavir (LPV/r) monotherapy
(n=83) or LPV/r + AZT/3TC (n=53). We focus here on patients in the LPV/r
monotherapy arm followed through Week 96. The on-treatment (OT) analysis
considers only patients who had an HIVRNA< 50 copies/mL at Week 48 (n=56).
The intent-to-treat (ITT) analysis involves all patients randomized to LPV/r monotherapy (n=83).
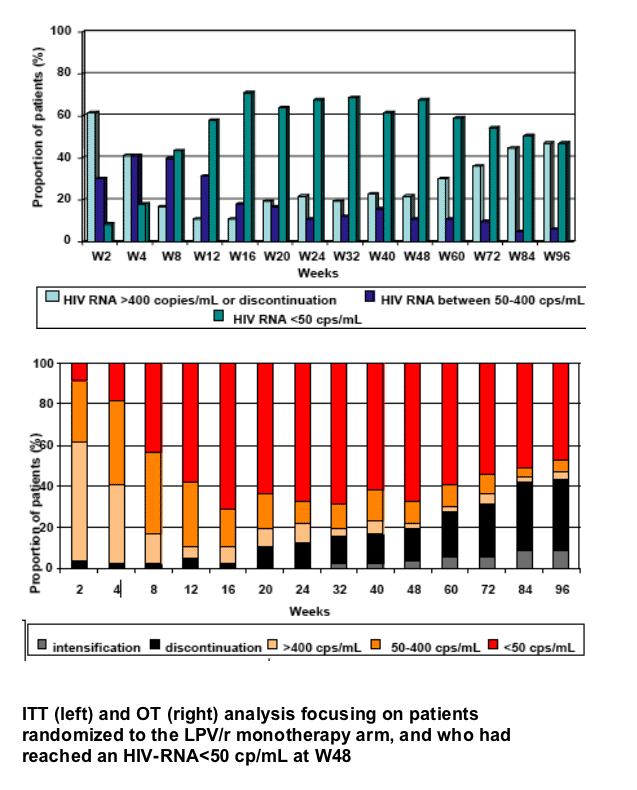
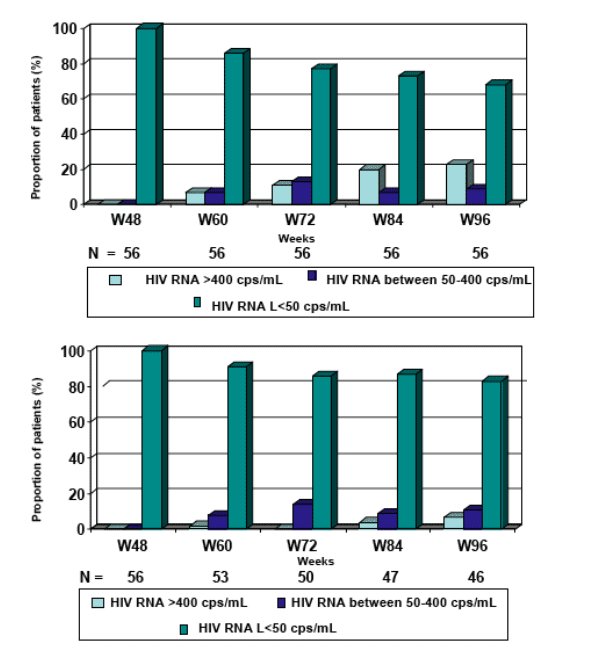
The follow-up of the 56 patients, virologically controlled at Week 48, indicate that 46 of them remained on LPV/r monotherapy at Week 96, with 38/46 (83%) having HIV RNA <50 copies/mL.
Factors associated with sustained undetectable HIV-RNA through W96 were baseline HIV-RNA and an HIV-RNA<400 copies/mL at W2.
PI-associated resistance mutations were evidenced in 5/83 patients in the monotherapy arm from baseline to W96 (46I+63P at W40, 76V at W44, 13V+46I+76V at W62, 10F+82A at W76 and 76V at W90). Among these 5 patients with selected PI resistance mutations, 3/5 intensified with NRTIs and re-suppressed to <50 copies/mL, 1/5 was switched to nevirapine + AZT/ 3TC, 1/5 continued on LPV/r monotherapy (investigatorŐs decision).
CONCLUSIONS
LPV/r monotherapy was able to maintain sustained viral suppression up to W96 in patients who already achieved an HIV-RNA<50 cp/ml at W48: 83% of patients with HIV-RNA<50 cp/mL at W48 maintained an HIV-RNA<50 cp/mL at W96.
The occurrence in some patients of low-level viremia (50-500 copies/mL) may increase the risk for drug resistance.
Major PI-associated resistance mutations were evidenced in 5 out of 83 (6%) patients treated with LPV/r monotherapy.between baseline and W96. However, these mutations did not jeopardize future therapeutic options in any of the 5 patients.
|
| |
|
 |
 |
|
|