 |
 |
 |
| |
In Mexico City Viva Metabolics!
|
| |
| |
David Alain Wohl, MD - The University of North Carolina
Chaotic, choked with too many people inhabiting too little space and, at times, even lawless, Mexico City was the perfect setting for the 2008 International AIDS Society (IAS) Conference. Like its mega-urban doppelganger, the conference has mutated from its own humble beginnings to become a monstrous mass of humanity. No longer the premier venue for the serious business of addressing an emerging epidemic, IAS has become a huge vortex of a meeting, sucking in people with diverse interests, backgrounds and agendas from across the planet. With something for everyone there was not enough of much of anything. That is because the conference is really a collection of mini conferences all occurring, like parallel universes, in the same time and space. As Lawrence Altman of the New York Times observed, the conference is less a scientific meeting than a convention where people in the business go to meet others in the business. The basic scientist and the sex worker, the prevention counselor and the village hospice director, the politician and the advocate, the lawyer and the physician - all waited in the same line for tacos. Even the potentially volatile mix of science and politics failed to produce any thrills and was largely limited to droll protestors marching against the pharma-de-jour (curiously side stepping the insultingly flashy South African Ministry of Health booth) while delegates snapped photos of their antics for the kids.
As a clinician, the conference I went to felt limited and diluted by the nonclinical. In this circus there were just too many rings. So dodging the anatomically correct HIV prevention hand puppets and narrowly escaping the caped Condom-man and Bupinorphine-Woman, I checked into the scattered sessions dealing with los complicaciones metab—licos. Here is shat I learned.
SMART, D:A:D and ABC
It was not too long ago that metabolic complications of HIV and its therapies were relegated to poor stepsister status at the big HIV meetings. So how ironic (and satisfying to us fat- and lipid-heads) that in 2008 metabolics dominated as a major issue in the management of HIV infection. Big dogs in the HIV treatment world can actually be heard earnestly uttering words like, LDL-HDL ratio, inflammatory markers, myocardial infarction and D-dimers.
Of course, much of our gratitude for this enhanced attention on complications of therapy can be directed to abacavir. At February's CROI meeting, we learned that in the large prospective D:A:D Study this nucleoside analogue and, to a lesser extent, ddI were each associated with an increased risk of myocardial infarction (MI) (1, 2). To call these results surprising would to be to court understatement. Since then we have gone through the traditional stages of denial and bargaining and may now be finally getting to acceptance as the community has moved from casting suspicious looks toward D:A:D to wondering if there just may be something up with abacavir. At IAS we learned, indeed, there is.
The D:A:D investigators who reported the abacavir-MI connection have turned their attention to data from the continuous HIV therapy arm (n=2,752) of the randomized SMART Study, a huge trial of CD4-guided treatment interruption versus maintaining ART (3, 4). (Recall, the SMART Study demonstrated that those discontinuing HIV therapy based on CD4 cell count parameters, suffered higher rates of cardiovascular, renal, hepatic and AIDS-related events, as well as mortality, compared to those maintaining HIV therapy.) In the analysis of the SMART Study data presented at IAS and recently published as a Fast Track article in the journal AIDS, a link between risk of MI and abacavir was again found. Overall, approximately 20% of the SMART Study cohort was receiving abacavir at study entry - typically without didanosine. Patients receiving non-abacavir therapies were more often women and also used anti-hypertensive drugs and lipid-lowering agents less frequently. Yet, the prevalence of major cardiovascular disease (CVD) risk factors was similar across the abacavir, didanosine and other NRTIs groups, ranging from 14% to 18%.
The investigators compared the risk of CVD among those on abacavir to the participants receiving NRTIs other than abacavor or didanosine. CVD was parsed to three increasingly inclusive categories: a) MI (n=19), b) major CVD (MI, stroke, surgery for coronary artery disease, and CVD death) (n=70), and c) expanded CVD (major CVD plus congestive heart failure, peripheral vascular disease, coronary artery disease requiring drug treatment, and un-witnessed deaths) (n=112). For each CVD category abacavir use was associated with an increased risk (adjusted hazard ratio of 4.3 [95% CI: 1.4-13.0], 1.8 [1.0-3.1], and 1.9 [1.3-2.9], respectively). This association between abacavir and CVD persisted even when the analysis was adjusted for CVD risk factors and when those receiving tenofovir were considered the referent group - indicating tenofovir did not share this association. Those who at baseline had five or more CVD risk factors (>45, male sex, smoking, total/HDL cholesterol > 4, BP-lowering treatment, lipid lowering treatment, prior CVD , diabetes, and ischemic changes on ECG) or just ECG abnormalities had significantly increased risk of CVD with abacavir compared to those without these problems (from Jules: in speaking with the study author Jens Lundgren he said these results were driven by the patients with 5 or more risk factors, see the link to the slides he presented at IAS; or it's the slide panel on the far right below: the blue dots are the risk for patients with 5 ot more CDV risk factors and the brown dots are the line for those with less risk factors and the line crosses the perpendicular HR line of 1 so I think that raises a question regarding the actual proof for patients with <5 risk factors). The ECG changes considered abnormal included: Q-wave changes (Minnesota codes 1.1, 1.2, and 1.3), ST-segment depression (codes 4.1, 4.2, and 4.3), T-wave inversions (codes 5.1, 5.2, 5.3, and 5.4), bundle branch block, and QT-interval more than 112%.
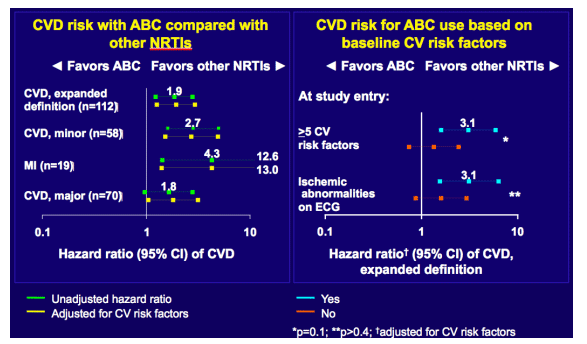
Abacavir and Heart Disease - (08/08/08)
In one of the first attempts at delving into a search for the basis of an abacavir-CVD link, four markers of inflammation - high sensitivity C-reactive protein (hsCRP), interleukin-6 (IL-6), amyloid A and P, as well as the coagulation markers D-dimer and prothrombin fragment 1+2 - were measured in 791 participants including participants with CVD, opportunistic disease, end-stage renal disease, cirrhosis or death and matched controls not experiencing such events and a random sample of participants. The investigators looked at levels of the markers by NRTI exposure and found that hsCRP and IL-6 were significantly elevated in those on abacavir relative to those on other NRTIs even after adjustment for CVD risk factors and other baseline covariates. There were no significant differences in the levels of the other markers between those treated with abacavir or not. In contrast, in the HEAT Study, a trial of abacavir/lamivudine versus tenofovir/emtricitabine in treatment naive patients also receiving lopinavir/ritonavir, IL-6 and hs-CRP levels decreased during treatment in both arms and was not noted to be significantly different across the study groups (5).
The finding in the SMART Study of a potential link between abacavir and these inflammatory markers is provocative and may start to close a loop tying this NRTI to CVD. Previously, it was thought that HIV therapies by controlling viremia reduces inflammation. This line of thinking was borne out by the finding that in a substudy of AIDS Clinical Trials Group (ACTG) trial A5142, all study arms in this treatment naive study improved endothelial function as measured by brachial reactivity. In addition the poorer outcomes (including CVD events) among those stopping HIV therapy in the SMART reinforces these data in a clinically relevant way.(6). However, the possibility that not all antiretroviral exert the same anti-inflammatory effects is starting to be considered and requires further scientific exploration.
Overall, the finding of an association between abacavir and CVD is largely consistent with the results of the D:A:D Study and while there was some overlap in the clinical centers contributing to the D:A:D and SMART studies, according to the authors, the findings persist when such overlap is excluded. The similarity of the results and the observation of elevations of hsCRP and IL-6 among the abacavir receiving participants further raises our concerns regarding abacavir, especially when coupled with preliminary data from ACTG study A5202 finding relatively greater risk of virologic failure with abacavir/lamivudine versus tenofovir/emtricitabine in treatment naive patients with pre-treatment HIV RNA levels above 100,000 copies/mL (7).
However, despite these alarming results there are some points to consider before freaking out and calling all your abacavir-prescribed patients in to the office. In both studies the risk of MI with abacavir was greatest in those with the most CVD risk factors. Such patients require consideration of a change from abacavir or attempts at neutralization of their underlying cardiovascular threats. For those abacavir-treated patients blessed with better lipids and an aversion to tobacco products, any heightened risk of a heart attack is serious, but the potential benefit of a swap in antiretrovirals needs to be balanced against the risks of any substitution - namely, new adverse effects and loss of efficacy (albeit loss of virologic suppression is unlikely).
Additionally, although the risk of CVD with abacavir is increased it is not common either in D:A:D (383 MIs/33,000 patients), SMART (112 CVD-expanded definition events/2,752 subjects) or among those enrolled in 54 ARV clinical trials performed by the makers of the drug (8). These trials included 33 with abacavir as part of the background of a study regimen, 13 in which the drug was studied against a control and eight that did not include abacavir. Over 14,000 subjects were included in these trials but their follow-up averaged less than a year. The rate of MI or other CVD reported during these studies was low (<2.3/1000 person-years and <5.9/100 person-years, respectively) and there was no significant difference between those exposed to abacavir compared to those who were not. These results are not a repudiation of the findings in D:A:D and SMART studies as there are significant limitations to a post-hoc search for clinical events in the context of completed clinical trials rather than prospective studies specifically designed to detect adverse events. But, they do offer a bit of reassurance that this very real problem of abacavir and CVD is not common. Exactly why some people experience a greater risk for CVD with this NRTI and by what mechanism is what we need to determine if we are to use this drug with any confidence.
New Drugs and New Data on Lipids
Increasingly, a measure of a new drug's success extends beyond virologic efficacy to encompass metabolic tolerability. Lipid and fat neutrality is especially important for agents that are gunning for position as an initial regimen. At IAS additional data on lipid parameters during treatment with newer agents were presented.
Raltegravir
In the first randomized trial of raltegravir in treatment naive patients, the integrase inhibitor, studied at four doses ranging from 100 mg BID to 600 mg BID, was compared to efavirenz in patients also receiving tenofovir and lamivudine (9). Previously reported results from the first 48 weeks of this trial of 198 individuals demonstrated similar rates of viral suppression across the raltergravir and efavirenz groups with little change in lipids seen in those receiving raltegravir compared to a significant rise in total and LDL cholesterol with efavirenz. In Mexico City 96-week data continue to show impressively little effect of raltegravir on lipid parameters. Total and LDL cholesterol, as well as triglycerides, remained relatively unchanged from baseline during raltegravir therapy, while these three parameters to varying degrees increased with efavirenz. HDL cholesterol rose in both groups. Why raltegravir does not lead to the typical rebound in lipids that other potent therapies produce in treatment naive patients is unclear as some of this rise, particularly in LDL cholesterol, is thought to be due to reversal of HIV-mediated suppression of cholesterol subsets - another mysterious feature of this mistress of integrase inhibition. Next to nothing is currently known about the effect of raltegravir on body shape and hopefully future conferences will provide some more data in this area.
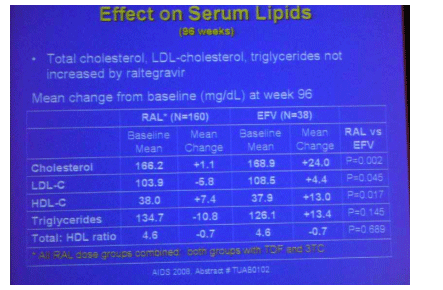
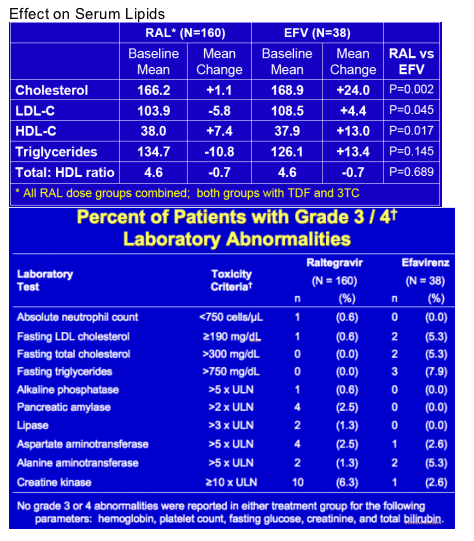
Raltegravir 96 Weeks data from Phase II Study in Treatment-Naive - (08/06/08)
Darunavir
In the PI wars now being waged, accusations regarding artery-clogging lipid increases are lobbed across marketing lines like mortars. While most of the head-to-head data make it clear that there is little, lipid-wise, that separates the ritonavir-boosted protease inhibitors from one another, these small differences are the fulcrums upon which some try to lift their brand. In the ARTMEMIS Trial of treatment naive patients (n=689), darunavir dosed at 800 mg once daily and boosted with 100 mg of ritonavir was compared to lopinavir/ritonavir (either once daily or BID) when both were accompanied by a side order of tenofovir/emtricitabine (10). At 48 weeks, darunavir was found to be non-inferior to lopinavir/ritonavir (HIV RNA <50 copies/mL in 84% of darunavir/ritonavir recipients versus 78% of lopinavir/ritonavir recipients (p <0.001). In a focused presentation at IAS of the relative lipid changes during therapy, there was not much to write home about when it came to any differences between these regimens (11). LDL-cholesterol levels rose similarly and were over 130 mg/dl in only a quarter of those in each study arm. HDL- and total-cholesterol increased a bit more with lopinavir than darunavir in this study; however, triglycerides also increased more with lopinavir. The total cholesterol to HDL cholesterol ratio was remarkably similar between the two arms and only 6% of the darunavir-assigned and 8% of those randomized to lopinavir were on lipid lowering therapies by week 48. These results indicate that when it comes to cholesterol subsets, lopinavir and darunavir appear to be more alike than not. As in most other studies of lopinavir, increases in triglycerides were modestly greater with this protease inhibitor compared to others.
ARTEMIS: lipid tolerability and safety of darunavir and lopinavir in antiretroviral-naive patients - (08/12/08) -
Atazanavir vs. Lopinavir and Race
Another analysis of lipid data derived from a major treatment naive trial explored racial differences in lipid changes during the CASTLE Study (n=883), a trial of therapy with atazanavir 300 mg plus ritonavir 100 mg once daily versus lopinavir/ritonavir, again when tenofovir/emtricitabine formed the nucleotide/side backbone. It is remarkable, given the popularity of atazanavir as a component of early HIV treatment regimens and common perceptions regarding its effect on lipids, that there are actually very few data regarding the use of the drug when boosted with ritonavir and combined with commonly used nucleosides. As was previously reported, the CASTLE study arms were similarly efficacious in driving down viral load and the atazanavir arm experienced a smaller increase in non-HDL cholesterol (the bad stuff) and triglycerides compared to lopinavir (12). In the IAS presentation, the lipid changes among the white (48%), black (16%), Asian (9%) and other race/ethnicity (24%) study participants were examined (13).
The numbers get quite small when comparing lipid changes among racial groups by regimen; however, it is interesting that non-white participants tended to have smaller increases in LDL-cholesterol and non-HDL-cholesterol compared to those who were white regardless of regimen. Blacks in particular seemed to have hardly any increase in these lipid parameters or triglyceride levels on either protease inhibitor. Gender was not included as a factor and it is likely that most of the women in this study, 31% of the cohort, were African-American. A separate analysis of these data examined gender found that women tended to have smaller changes in most lipid parameters compared to men across treatment assignments (14).
The potential that race/ethnicity may impact in the lipid effects of HIV therapies is of great interest and may have a bearing on the tailoring of antiretroviral treatment to patients needs. Further studies exploring such effects are clearly called for and with treatment trials of HIV therapies becoming increasingly diverse, these investigations will be easy to accomplish.
Figure 3. Change From Baseline at Week 48 in LDL Cholesterol (A), Triglycerides (B), Non-HDL Cholesterol (C), HDL Cholesterol (D), and Total Cholesterol (E) (mg/dL) by Treatment Regimen and Race/Ethnicity
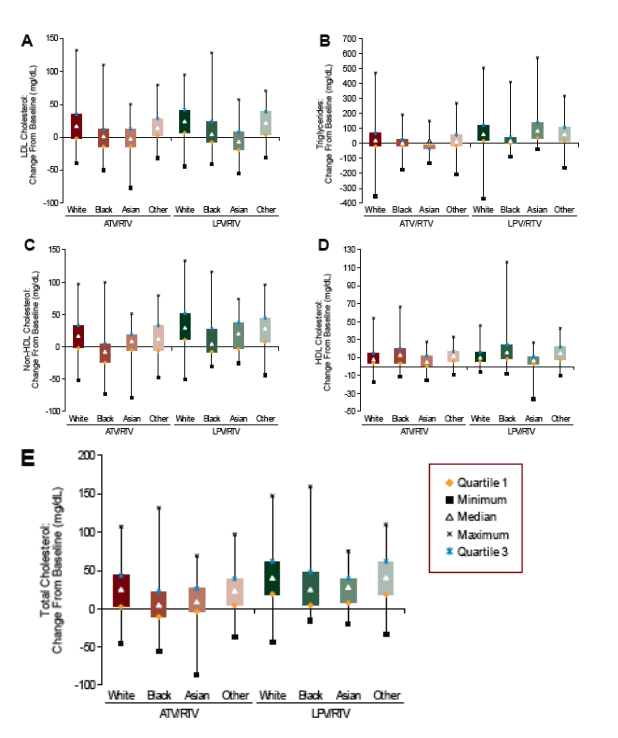
Efficacy and Safety By Racial Group in ARV-Naive Subjects Treated With Atazanavir/Ritonavir or Lopinavir/Ritonavir: 48-Week Results for the CASTLE Study - (08/07/08)
Gender-Based Differences in ART-Naives in CASTLE Study: viral response, lipids, adverse events; and Other Gender-Based Studies at IAC - (08/19/08) -
'Mega'-Analysis of ART and Lipids
Lastly, an analysis of the lipid changes of 4,231 participants in 12 HIV treatment naive clinical trials was also featured at the conference (15). The trials were those that included at least one ritonavir-boosted protease inhibitor combined with one or more nucleoside analogues, was populated with more than 50 participants and had sufficient lipid data out to 48 weeks. Included were some of the more recent chart-toppers including ARTEMIS, GEMINI, ALERT, KLEAN and HEAT, as well as some golden oldies such as SOLO and ABT-863. In a multivariable analysis of the effects of the individual protease inhibitors and the nucleosides on lipid changes there were no significant differences in lipid elevations between boosted saquinavir, atazanavir and darunavir but lopinavir and fos-amprenavir (which induced similar lipid changes) produced greater increases in total cholesterol and triglycerides than these other agents. Turning to the nucleosides, lipid changes with abacavir and stavudine - when combined with lamivudine - were not significantly distinguishable and these agents were associated with greater increases in both LDL- and HDL-cholesterol relative to tenofovir/emtricitabine.
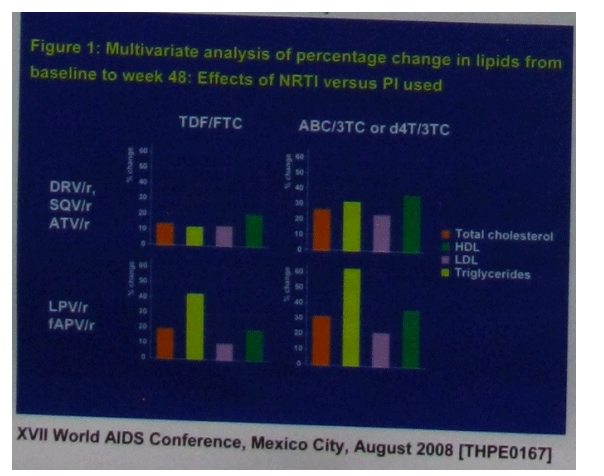
PI Switch Does Not Alter Belly Fat: Pass it on
If I had a quarter for every time I get a call or email about whether I thought a patient with a paunch on HAART should switch their protease inhibitor for another drug, I would be living large. Old beliefs die hard and the idea that protease inhibitors alone cause belly fat increases is so ingrained in our collective consciousness that we become deaf and blind to data that demonstrate the contrary. Case in point: trunk fat in study A5142 increases similarly among those receiving efavirenz + 2 NRTIs, lopinavir/ritonavir + 2 NRTIs and efavirenz + lopinavir/ritonavir (16). Sure, there may be differences in deep belly fat not seen on DEXA scans but at this point we can assume that waist size change is not different across most current initial HIV therapies.
Further support for the more-alike-than-different take on protease inhibitors and belly fat comes from a randomized international study of continuing a ritonavir-boosted PI vs switching to atazanavir/ritonavir in patients with lipohypertrophy and HIV RNA <400 c/mL (n=200) (17). More than 70% of the participants were on lopinavir/ritonavir at the time of study entry. It was hypothesized that the purportedly better glycemic profile of atazanavir/ritonavir may lead to less fat accumulation relative to other drugs of this class that may produce impaired glucose tolerance.
After 48 weeks following randomization there was no significant change in fat distribution measured by DEXA (trunk fat, limb fat, trunk:limb fat ratio), CT (visceral adipose tissue [VAT], subcutaneous adipose tissue [SAT], and total adipose tissue [TAT]), waist circumference or waist:hip ratio. Non-HDL-cholesterol and triglycerides did improve among those changing to atazanavir/ritonavir compared to those remaining on pre-study PIs.
The bottom line here is that switching the protease inhibitors will not melt inches off of the belt line. The same likely holds true for changes to other potent regimens but has not been well studied. Perhaps these and other results will help us to stop thinking only about PIs when looking at our patients' navels.
Tesamorelin and Belly Fat
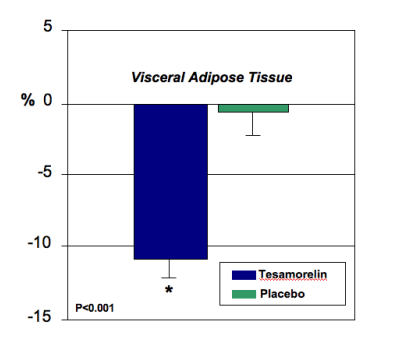
Tesamorelin is growth hormone releasing factor (GHRF), an agent that has previously been found to reduce visceral fat in HIV-infected individuals enrolled in a phase II study published in the New England Journal of Medicine (18). Data from a second, confirmatory trial of 404 HIV-infected patients with elevated waist circumference were presented (19). Subjects were randomized to receive tesamorelin (2 mg SC/d) (n=270) versus placebo (n=126) and at 24 weeks VAT and trunk fat significantly decreased in tesamorelin-treated pts (p<0.001) without significant loss of SAT.
Figure 1. Percent change from baseline in VAT at Week 26 of treatment. Data are mean ± SEM. *P<0.001 vs. placebo.
Results from the 26-week Confirmatory, Phase 3 Trial of Tesamorelin (TH9507), a Growth Hormone-Releasing Factor Analogue, in HIV Patients with Excess Abdominal Fat: A Multicenter, Double-blind, Placebo-controlled Study with 404 Randomized Patients - (08/12/08) -
Long-Term Impact of Tesamorelin (TH9507), a Growth Hormone-Releasing Factor Analogue, on Body Image Parameters in HIV-Infected Patients with Abdominal Fat Accumulation - (08/12/08) -
There were no significant effects of the drug on total-cholesterol/HDL-cholesterol ratio or glucose, although IGF-1 increased, albeit while remaining in the physiologic range. Tesamorelin was well tolerated and most adverse events were related to injections site reactions of this injected agent. Longer term data will assuredly be presented at future conferences and FDA approval of the drug for this indication is being sought. One can only hope (if not advocate for and demand) that if approval is granted that the drug be reasonably priced and/or come with a generous patient support program to assist the many patients living with disfiguring fat accumulation and no financial ability to access surgery or growth hormone.
Conclusions
So after a long week, what did we learn about metabolic complications? The additional data regarding abacavir were provocative and the clinician now has to look at the studies including D:A:D, SMART and the GlaxoSmithKline analyses and decide for themselves what they feel the risk is and for whom. Too often we look for the sound bite that wraps everything up into a neat cognitively easy to digest package. But, in this case, we have to dig deeper to see the limitations of each of these studies but to not dismiss their results.
That HIV therapy can influence the risk of CVD is becoming clearer. However, we are increasingly appreciating that HIV itself can wreak havoc on our cardiac, renal and hepatic systems. While our therapeutic tools are powerful suppressors of viremia they are not metabolically inert. The comparisons of the lipid effects of different regimens, including newer antiretroviral agents helps us to design treatment approaches that maximize benefit but minimize risk. We know that there are differences between antiretrovirals when it comes to lipids and we can now intelligently factor these effects into our calculations of what is the best combination for the patient sitting before us.
For those with abnormal fat accumulation, teasmorelin is a hope. As mentioned above, the drug looks promising, although it is unclear if the effects persist once the agent is discontinued. In addition, the history of growth hormone, which is unobtainable for most people living with HIV and fat accumulation, has led to a very jaded view of whether things will be different with tesamorelin. Hopefully, somewhere, somehow, someone will do the right thing.
In two years, the conference will re-convene in Vienna. There on the threshold of Eastern Europe we will attend a conference that will likely be focused on that region of the planet - where the advances of HIV therapeutics and prevention have been in short supply. That may leave little room for updates on lipids, CVD and body shape, but at the International AIDS Conference surprises can happen and that is why so many of us go.
References
1. Sabin C et al. Do thymidine analogues, abacavir, didanosine and lamivudine contribute to the risk of myocardial infarction? The D:A:D study. Fifteenth Conference on Retroviruses and Opportunistic Infections, Boston. Abstract 957c, 2008.
2. D:A:D Study Group. Use of nucleoside reverse transcriptase inhibitors and risk of myocardial infarction in HIV-infected
patients enrolled in the D:A:D study: a multicohort collaboration. Lancet 2008; 371:1417-1426.
3. SMART Study Group. CD4 count-guided interruption of antiretroviral treatment. N Engl J Med 2006; 355:2283-2296.
4. Lundgren J, Neuhaus J, Babiker A, et al, and the SMART/INSIGHT and D:A:D Study Groups. Use of nucleoside reverse transcriptase inhibitors and risk of myocardial infarction in HIV-infected patients enrolled in the SMART study. In: Program and abstracts of the XVII International AIDS Conference; August 3-8, 2008; Mexico City, Mexico. Abstract THAB0305.
5. Smith KY, Fine DM, Patel P, et al. Similarity in efficacy and safety of abacavir/lamivudine (ABC/3TC) compared to tenofovir/emtricitabine (TDF/FTC) in combination with QD lopinavir/ritonavir (LPV/r) over 96 weeks in the HEAT Study. In: Program and abstracts of the XVII International AIDS Conference; August 3-8, 2008; Mexico City, Mexico. Abstract LBPE1138.
6. Torriani F, Komarow L, Cotter B, et al. Control of HIV viral replication is associated with rapid improvement in endothelial function sustained over twenty-four weeks: A5152s, a substudy of A5142. In: Program and abstracts of the 4th International AIDS Society Conference on HIV Pathogenesis, Treatment and Prevention; July 22-25, 2007; Sydney, Australia. Abstract WEAB302.
7. Sax P, Tierney C, Collier A, et al. ACTG 5202: shorter time to virologic failure (VF) with abacavir/lamivudine (ABC/3TC) than tenofovir/emtricitabine (TDF/FTC) as part of combination therapy in treatment-naive subjects with screening HIV RNA ≥ 100,000 c/mL. In: Program and abstracts of the XVII International AIDS Conference; August 3-8, 2008; Mexico City, Mexico. Abstract THAB0303.
8. Cutrell A, Hernandez J, Yeo J, Brothers C, Burkle W, Spreen W. Is abacavir (ABC)-containing combination antiretroviral therapy (CART) associated with myocardial infarction (MI)? No association identified in pooled summary of 54 clinical trials. In: Program and abstracts of the XVII International AIDS Conference; August 3-8, 2008; Mexico City, Mexico. Abstract WEAB0106.
9. Markowitz M, Nguyen B-Y, Gotuzzo E, et al, and the Protocol 004 Part II Study Team. Sustained antiretroviral efficacy of raltegravir as part of combination ART in treatment-naive HIV-1 infected patients: 96-week data. In: Program and abstracts of the XVII International AIDS Conference; August 3-8, 2008; Mexico City, Mexico. Abstract TUAB0102.
10. Ortiz R, DeJesus E, Khanlou H, et al. Efficacy and safety of once-daily darunavir/ritonavir versus lopinavir/ritonavir in treatment-naive HIV-1-infected patients at week 48. AIDS 2008; 22: 1389-97.
11. Arathoon, E, et al. ARTEMIS: lipid tolerability and safety of darunavir and lopinavir in antiretroviral-naive patients. In: Program and abstracts of the XVII International AIDS Conference; August 3-8, 2008; Mexico City, Mexico. Abstract THPE0151
12. Molina JM et al. Fifteenth Conference on Retroviruses and Opportunistic Infections, Boston; 2008; Abstract 37
13. McGrath D, et al. Efficacy and safety by racial group in ARV-naive subjects treated with atazanavir/ritonavir or lopinavir/ritonavir: 48 week results for the CASTLE study (AI424138). In: Program and abstracts of the XVII International AIDS Conference; August 3-8, 2008; Mexico City, Mexico. Abstract TUPE0058
14. Absalon J, et al. Gender-based differences in ARV-naive patients treated with boosted protease inhibitors (PIs): results from the CASTLE study (AI424138). In: Program and abstracts of the XVII International AIDS Conference; August 3-8, 2008; Mexico City, Mexico. Abstract TUPE0062
15. Hill A, et al. Meta-analysis of first-line HAART with NRTIs plus boosted PIs in 2801 patients: effects on rises in lipid parameters. In: Program and abstracts of the XVII International AIDS Conference; August 3-8, 2008; Mexico City, Mexico. Abstract. THPE0167
16. Haubrich RH, Riddler S, DiRienzo G, et al, and the AIDS Clinical Trials Group 5142 Study Team. Metabolic outcomes of ACTG 5142: a prospective, randomized, phase III trial of NRTI-, PI-, and NNRTI-sparing regimens for initial treatment of HIV-1 infection. In: Program and abstracts of the 14th Conference on Retroviruses and Opportunistic Infections; February 25-28, 2007; Los Angeles, Calif. Abstract 38.
17. Moyle G, Girard J-M, Andrade J, et al, and the ReAL Study Group. Continuation of BID boosted PI vs switch to once-daily ATV/RTV for the management of lipodystrophy: 48-week primary analysis of the 96 week multicenter, open-label, randomized, prospective ReAL study. In: Program and abstracts of the XVII International AIDS Conference; August 3-8, 2008; Mexico City, Mexico. Abstract MOPDB103.
18. Falutz J, Allas S, Blot K, et al. Metabolic effects of a growth hormone-releasing factor in patients with HIV. N Engl J Med. December 6, 2007;357(23):2359-2370.
19. Falutz J, Marsolais C, Allas S, et al. Results from the 26-week confirmatory, phase 3 trial of tesamorelin (TH9507), a growth hormone-releasing factor analogue, in HIV patients with abdominal fat accumulation: a multicenter, double-blind, placebo-controlled study with 404 randomized patients. In: Program and abstracts of the XVII International AIDS Conference; August 3-8, 2008; Mexico City, Mexico. Abstract LBPE1156.
|
| |
|
 |
 |
|
|