 |
 |
 |
| |
A Pharmacokinetic Study to Evaluate an Interaction between Maraviroc and Raltegravir in Healthy Adults
|
| |
| |
Reported by Jules Levin
ICAAC/IDSA Oct 28 2008 Wash DC
E Andrews, P Glue, J Fang, P Crownover, R Tressler, and B Damle
Pfizer Inc, New York, USA
AUTHOR CONCLUSION
When MVC and RAL were coadministered in healthy adults, MVC Cmax, MVC AUCt, RAL C12 and RAL AUCt were reduced compared to monotherapy.
These changes are not likely to be clinically relevant as exposure to both drugs appeared to be above pharmacokinetic-pharmacodynamic targets.
Thus, no dose adjustment may be required when MVC and RAL are coadministered.
MVC and RAL when administered alone and concomitantly were generally safe and well tolerated.
BACKGROUND
Maraviroc (marketed as Celsentri/Selzentry) and raltegravir (marketed as Isentress) are two new antiretroviral drugs approved by the FDA and EMEA for use in treatment-experienced patients with HIV-1 infection. Maraviroc is the first approved CCR5 antagonist and raltegravir is the first approved integrase inhibitor.
Maraviroc is a substrate for CYP3A4 and P-gp, and is not an inducer or inhibitor of cytochrome P450 enzymes.1
Raltegravir is a substrate for UGT 1A1 and P-gp.2,3 It is not a substrate for cytochrome P450 enzymes and is neither an inducer or inhibitor of these enzymes.2
As there is interest in using these two agents as part of a combination antiretroviral drug regimen, we evaluated the two-way pharmacokinetic (PK) interaction between maraviroc and raltegravir.
OBJECTIVES
To estimate the effect of coadministration of maraviroc (MVC) and raltegravir (RAL) on the pharmacokinetics of MVC and RAL.
To evaluate the short-term safety and tolerability of MVC and RAL when administered alone and concomitantly.
METHODS
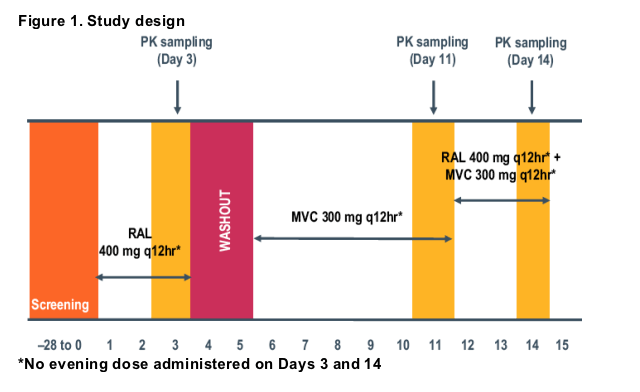
This was a Phase 4 open-label, multiple dose, fixed-sequence study (see Figure 1) performed in healthy adult volunteers at a single US site.
MVC 300 mg and/or RAL 400 mg was administered q12hr orally (Figure 1) on an empty stomach (no food for 1 hour before and after dosing).
PK evaluation was performed on Days 3 (RAL), 11 (MVC), and 14 (RAL + MVC).
On PK sampling days (Days 3, 11, and 14) blood samples for PK analysis were collected prior to the morning dose (immediately pre-dose) and at 0.5, 1, 1.5, 2, 3, 4, 6, 8, and 12 hours postdose.
MVC and RAL plasma levels were assayed using validated LC/MS/MS assays.4 Calibration standard responses were linear over the range of 0.500 to 500 ng/mL for MVC and 2.00 to 1,000 ng/mL for RAL.
PK was determined by non-compartmental analyses.
Point estimates of the geometric mean ratios (GMRs) and 90% confidence intervals (CIs) were determined for PK parameters (Cmax, AUCt, and C12).
Safety was assessed by monitoring adverse events (AEs), safety laboratory tests, vital signs, and ECGs.
RESULTS
Eighteen subjects (16 male, 2 female) were enrolled in this study, with an age range of 25- 54 years. Race was recorded as black for nine subjects, white for six, and 'other' for three. BMI ranged from 19.9 to 30.4 kg/m2, with a mean of 26.2 kg/m2.
Seventeen subjects completed the study. One subject withdrew consent during the study for personal reasons and was discontinued on Day 10 prior to the full PK day (Day 11) while receiving MVC.
Pharmacokinetics
Median plasma concentration-time plots are presented in Figure 2 (MVC) and Figure 3 (RAL).
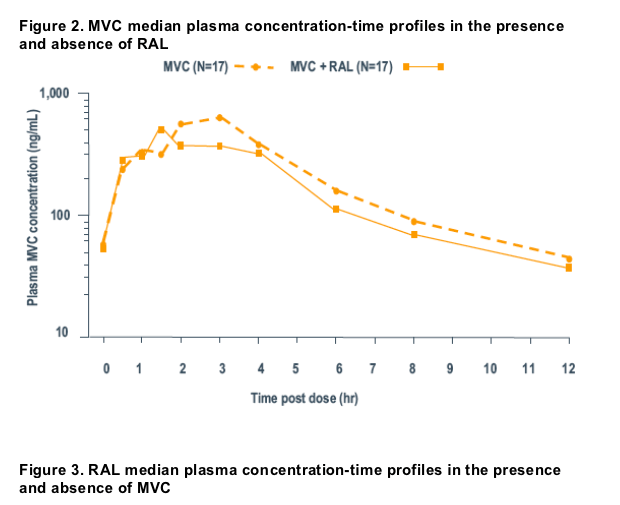
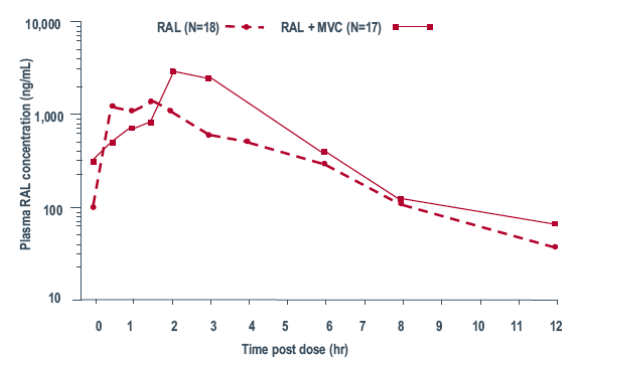
Geometric mean values for plasma PK parameters are presented in Tables 1 and 3 and statistical analyses are presented in Tables 2 and 4 for MVC and RAL, respectively.
When coadministered with RAL, MVC AUCt was reduced by an average of 14% relative to monotherapy. However, MVC average concentration (AUCt divided by 12) was >100 ng/mL in all subjects. RAL coadministration reduced MVC Cmax by an average of 21% relative to MVC monotherapy. Median Tmax was similar for MVC given together with RAL compared to MVC given alone.
When coadministered with MVC, RAL AUCt was reduced by an average of 37% relative to monotherapy. MVC coadministration reduced RAL C12 values by an average of 28% relative to RAL monotherapy. Median Tmax was 1.5 hours shorter for RAL given together with MVC compared to RAL given alone.
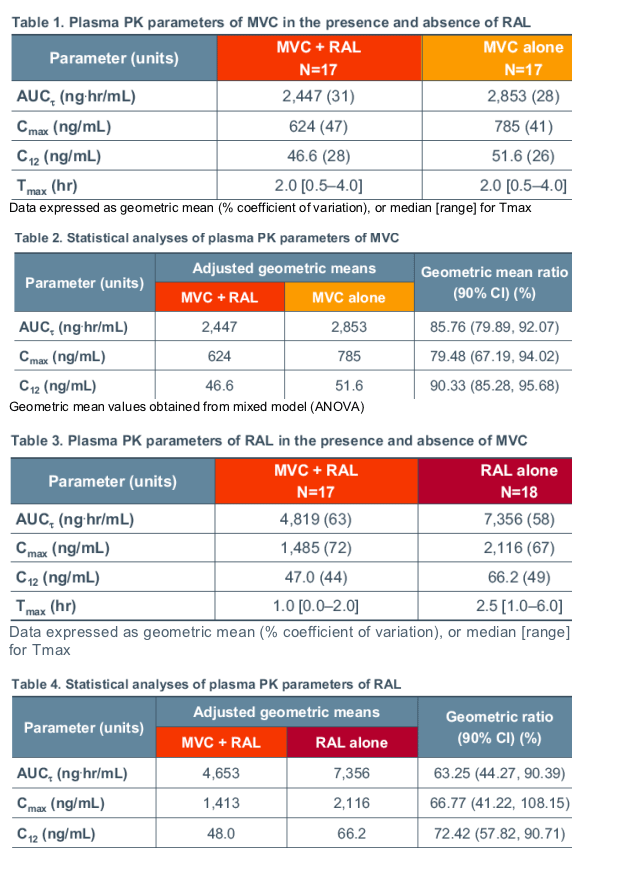
Geometric mean values obtained from mixed model (ANOVA)
Safety results
There were no serious adverse events, discontinuations, or dose reductions due to AEs.
At least one treatment-emergent AE was experienced by three subjects (16.7%) during treatment with raltegravir, four subjects (22.2%) during treatment with maraviroc, and four subjects (23.5%) during treatment with maraviroc plus raltegravir. All events were mild and treatment related, and included constipation, dyspepsia, folliculitis, back pain, headache, peripheral neuropathy, and somnolence.
DISCUSSION
In healthy volunteers, relative to monotherapy, coadministration of MVC and RAL was associated with reductions in MVC Cmax and RAL C12, as well as a reduction in AUCt for both agents.
Of all PK parameters investigated in a recent analyses of the exposure-response (efficacy) relationship for MVC in treatment-naive5 and treatment-experienced6 patients, MVC exposure (C-ave) appeared to correlate most closely with virologic success.
The analysis in treatment-experienced patients suggested a greater than 80% net probability of success at an MVC average concentration (AUCt divided by 12) of >100 ng/mL, in subjects likely to respond to MVC.6
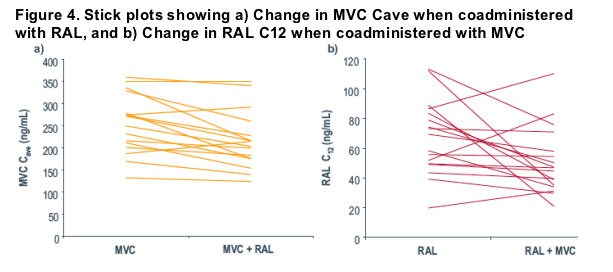
In the present study, this MVC C-ave threshold value was achieved in all subjects when MVC was coadministered with RAL, which suggests that coadministration is unlikely to affect the antiretroviral activity of MVC (Figure 4a).
Historically, considerable inter- and intra-subject variability has been observed in the pharmacokinetics of RAL. For observed C12 in pivotal Phase 3 studies (BENCHMRK 1 and 2), the coefficient of variation for inter-subject variability was 212% and for intra-subject variability was 122%.2 Based on available clinical experience from Phase 1 and 2 studies, a drug-drug interaction-associated increase in exposure of up to 2-fold is considered to be not clinically relevant.3
In a recent evaluation of the correlation of a range of RAL PK parameters with antiretroviral activity in Phase 2 and 3 studies in treatment-experienced patients, no statistically significant relationship was observed between RAL C12 and virologic response.7 Based on available clinical experience from Phase 1 and 2 studies, a drug-drug interaction-associated decrease in RAL C12 of less than 60% is considered to be not clinically relevant.3
When given with MVC, RAL C12 values were reduced by an average of 28% (90% CI: 9, 42). Two of 17 subjects had a decrease in C12 >60% (76% for one subject and 69% for the other; Figure 4b). However, given the variable PK associated with RAL, we believe this is unlikely to be clinically significant.
Safety and efficacy data from studies of patients receiving regimens that include both MVC and RAL will be helpful in understanding the role of this combination in the treatment of HIV-1 infection.
CONCLUSION
When MVC and RAL were coadministered in healthy adults, MVC Cmax, MVC AUCt, RAL C12 and RAL AUCt were reduced compared to monotherapy.
These changes are not likely to be clinically relevant as exposure to both drugs appeared to be above pharmacokinetic-pharmacodynamic targets.
Thus, no dose adjustment may be required when MVC and RAL are coadministered.
MVC and RAL when administered alone and concomitantly were generally safe and well tolerated.
REFERENCES
1. Pfizer Inc. Selzentry prescribing information. 2007.
2. Merck & Co, Inc. Isentress prescribing information. 2008.
3. Merck. Isentress (raltegravir) 400 mg For Treatment of HIV. (NDA 22-145). Briefing Document. Presented as part of the FDA Antiviral Drugs Advisory Committee meeting, held September 5, 2007. Available at:
www.fda.gov/ohrms/dockets/ac/07/briefing/2007-4314b1-01-Merck.pdf.
4. Merschman SA, et al. J Chromatogr B Analyt Technol Biomed Life Sci 2007; 857:15-24.
5. McFadyen L, et al. XVII International AIDS Conference. Mexico City, Mexico, 3-8 August, 2008; Poster TUPE0053.
6. McFadyen L, et al. 11th European AIDS Conference. Madrid, Spain, 24-27 October, 2007; Poster P4.1/06.
7. Wenning L, et al. 9th International Workshop on Clinical Pharmacology of HIV Therapy. New Orleans, USA, 7-9 April 2008.
|
| |
|
 |
 |
|
|