 |
 |
 |
| |
Pharmacokinetic (PK) Evaluation of Darunavir/Ritonavir
(DRV/r) and Raltegravir (RAL) in Healthy Subjects
|
| |
| |
Reported by Jules Levin
ICAAC/IDSA Wash DC Oct 2008
MS Anderson,1 V Sekar,2 F Tomaka,2 J Mabalot,1 R Mack,2 L Lionti,1 S Zajic,1 L Wenning,1 C Vanden Abeele,3 M Zinny,4 NM Lunde,5 B Jin,1 JA Wagner,1 M Iwamoto1
1Merck & Co., Inc., Whitehouse Station, NJ, 2Tibotec, Inc., Yardley, PA, 3Tibotec BVBA, Mechelen, Belgium, 4Promedica Clinical Research Center, Inc., Boston, MA, 5Prism Research, S
Conclusions
Multiple oral doses of 400-mg RAL given in combination with 600-mg DRV plus 100-mg RTV in healthy subjects resulted in a common adverse experience of rash.
Based on limited PK data, coadministration of DRV/r and RAL resulted in a modest effect on RAL with no clinically important changes in DRV pharmacokinetics.
Abstract
Background: Both DRV/r, a next generation PI, and RAL, an InSTI, have separately demonstrated potent clinical activity in HIV-infected patients. This study evaluated the effect of co-administration of DRV/r and RAL on PK and safety in healthy subjects.
Methods: Eighteen healthy subjects were enrolled in an open-label, sequential 2-period study. In Period 1, all subjects were administered 400 mg RAL q12hr for 4 days. In Period 2, subjects were administered 400 mg RAL + 600/100 mg DRV/r q12hr for 12 days. Period 2 immediately followed Period 1. Doses were administered with a meal.
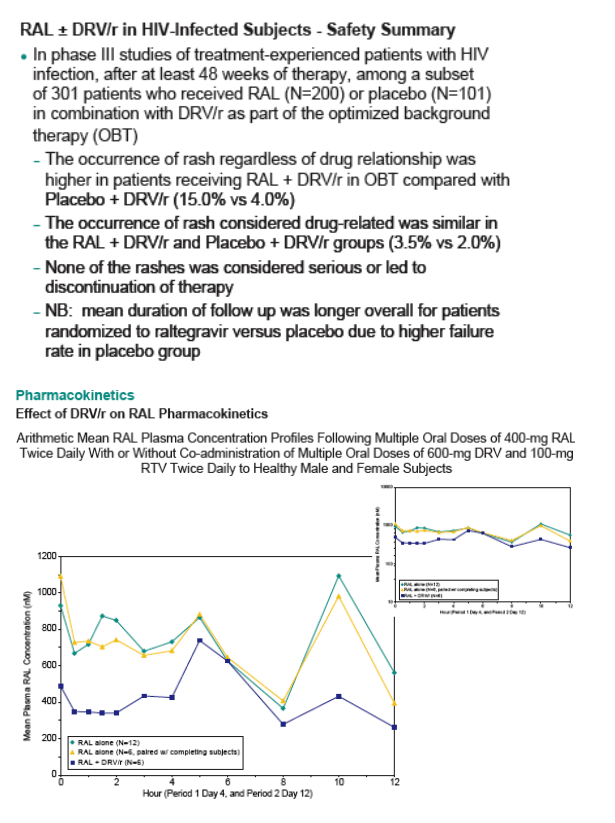
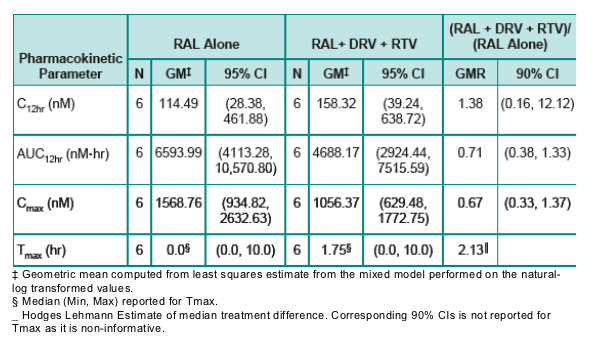
Results: Eight subjects presented with a clinical adverse experience (AE) of rash. Seven were rated mild to moderate in intensity; one progressed to a serious adverse event of maculopapular rash. Onset occurred between Days 8 and 12 of Period 2 of concomitant dosing. Six subjects out of 18 completed the study, but constituted an insufficient data set from which to draw PK conclusions. However, based on limited data, DRV/r had only modest effect on mean RAL PK parameters. The geometric mean ratio (GMR) for RAL co-administered with DRV/r relative to RAL alone was 0.71 (90% CI: 0.38 to 1.33) for AUC12hr, 0.67 (0.33 to 1.37) for Cmax, and 1.38 (0.16 to 12.12), for C12hr, respectively. Summary DRV PK parameters of AUC12hr, Cmax, C12hr in the presence of
RAL aligned closely to historical data reported in healthy subjects given DRV/r alone.
Conclusions: Contrary to clinical experience with the combination in HIV-infected patients, co-administration of RAL with DRV/r in healthy subjects resulted in a common adverse experience of rash. Based on limited PK data, a
co-administration of DRV/r and RAL resulted in a modest effect on RAL with no clinically important changes in DRV pharmacokinetics. MN.
Introduction
Raltegravir (RAL) is a novel HIV-1 integrase inhibitor with potent in vitro and clinical activity against HIV-1.
RAL is primarily metabolized by glucuronidation and has no inhibitory or inductive potential on CYP enzymes.
Darunavir (DRV) is a novel PI with potent in vitro and clinical activity against HIV-1.
DRV and ritonavir (RTV) are each metabolized primarily by CYP3A. DRV in combination with RTV is a net inhibitor of CYP3A.
An evaluation for a drug interaction between RAL and DRV/r was conducted as
HIV-infected patients are likely to benefit from combination administration.
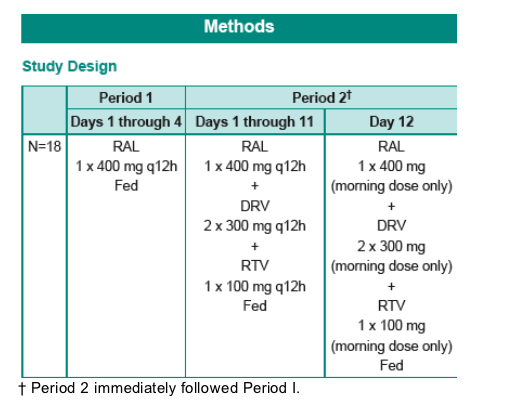


Due to the incidence of rash, the study was not further enrolled. These subjects were not replaced.
Due to an insufficient number of evaluable subjects and greater than anticipated variability, no pharmacokinetic conclusions for the effect of DRV/r on RAL can be drawn from this study.
However, based on limited pharmacokinetic data, co-administration of DRV/r and RAL appears to result in a modest effect on RAL.
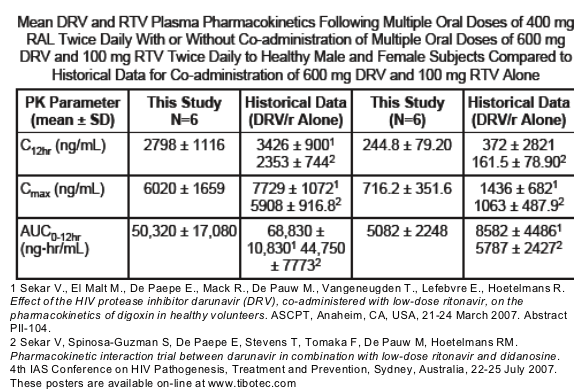
In the presence of RAL, mean DRV C12hr, Cmax and AUC12hr aligned closely to historical data reported in healthy subjects co-administered 600 mg DRV and 100 mg ritonavir b.i.d. alone.
These results are consistent with a report from the French expanded access program that includes an arm of 400 mg RAL b.i.d. co-administered with 600 mg DRV and 100 mg RTV b.i.d. (n=83) and which concludes that Despite a large interpatient variability, DRV/r combination had no deleterious effect on RAL Cmin.1
1 Long K., Soulie C., Schneider L., Ghosn J., Piketty C., Boue F., Reynes J., Raffi F., Calvez V., Katlama C., Peytavin G. Therapeutic Drug Monitoring of Raltegravir (MK-0518) in Experienced HIV-infected Patients. 11th European Aids Conference, October 24-27, 2007, Madrid, Spain.
|
| |
|
 |
 |
|
|