 |
 |
 |
| |
Omeprazole Increases Plasma Levels of Raltegravir (RAL) in Healthy Subjects
|
| |
| |
Reported by Jules Levin
ICAAC Wash Dc Oct 2008
Marian Iwamoto,1 Larissa A. Wenning,1 Bach-Yen Nguyen,1 Hedy Teppler,1 Allison R. Moreau,1 Rand R. Rhodes,1 William D. Hanley,1 Bo Jin,1 Charlotte M. Harvey,1 Sheila A. Breidinger,1
Neal Azrolan,1 H. Frank Farmer, Jr.,2 Robin D. Isaacs,1 Jeffery A. Chodakewitz,1 Julie A. Stone,1 John A. Wagner1
1Merck & Co., Inc., Whitehouse Station, NJ; 2Covance Clinical Research Unit, Inc., Daytona Beach, Florida
Copyright 2008 Merck & Co., Inc., Whitehouse Station, New Jersey, USA, All Rights Reserved
AUTHOR CONCLUSIONS
Omeprazole increases plasma concentration of RAL in healthy subjects
Population PK data demonstrate that patients with HIV infection receiving RAL have similar RAL levels whether or not they receive PPI or H2 blockers
--- Reported differences in gastric pH between HIV infected patients vs uninfected subjects may explain this dichotomous finding
RAL codosed with omeprazole or other pH-altering agents is generally well-tolerated and no areas of concern have been identified
Data support use of RAL with gastric pH-altering agents with no dose adjustment
Further investigation is necessary to more fully characterize the PK of RAL given in combination with pH-altering agents in HIV-infected patients
Abstract
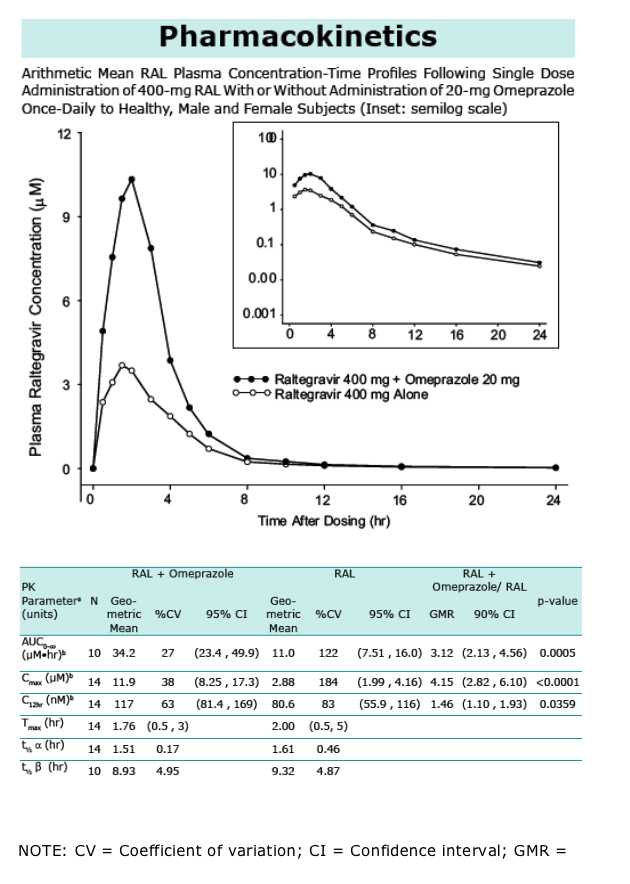
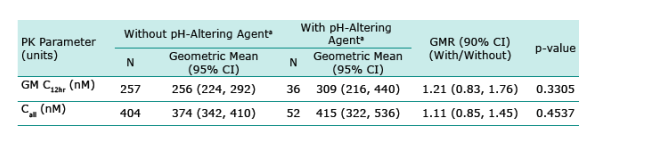
RAL is an HIV-1 integrase strand transfer inhibitor with potent in vitro and in vivo activity. RAL solubility is pH dependent with higher solubility at higher pH. The potential exists for RAL to be administered to patients receiving proton pump inhibitors which may influence RAL absorption and bioavailability. A study was conducted to evaluate co-administration of RAL and the proton pump inhibitor omeprazole in healthy, HIV-1 uninfected subjects. A single-blind, randomized, 2-period crossover study was performed examining single-dose RAL pharmacokinetics with and without administration of omeprazole dosed to steady state (Treatments B and A, respectively). The geometric mean ratio (GMR) (B/A) and 90% confidence interval was 3.12 (2.13, 4.56) for AUC0-∞, 4.15 (2.82, 6.10) for Cmax , and 1.46 (1.10, 1.93) for C12 hr. Tmax and t1/2 differences were not substantial. RAL plasma concentration increases with co-administration of omeprazole in healthy, uninfected subjects. The mechanism is likely increased absorption and bioavailability secondary to an increase in gastric pH. An additional analysis was performed on sparse pharmacokinetic sampling data obtained in the Phase III treatment-experienced HIV-infected patient studies in patients receiving gastric pH altering agents compared to those not on these agents. The GMR (with/ without pH-altering agents) and 90% confidence interval for C12 hr was 1.21 (0.83, 1.76) and Call (geometric mean of all observed concentrations) was 1.11 (0.85, 1.45). The effect of omeprazole on RAL pharmacokinetics in the intended population of use was shown to be diminished relative to healthy subjects.
INTRODUCTION
Raltegravir (RAL) is an HIV-1 integrase strand transfer inhibitor with potent in vitro and clinical activity.
The drug interaction profile of RAL is overall favorable with little indication of clinically meaningful interactions to date.
RAL solubility is pH dependent with higher solubility at higher pH.
The potential exists for RAL to be administered to patients receiving proton pump inhibitors which may influence raltegravir absorption and bioavailability.
A Phase I study was conducted to evaluate co-administration of RAL and the proton pump inhibitor omeprazole in healthy, HIV-1 uninfected subjects.
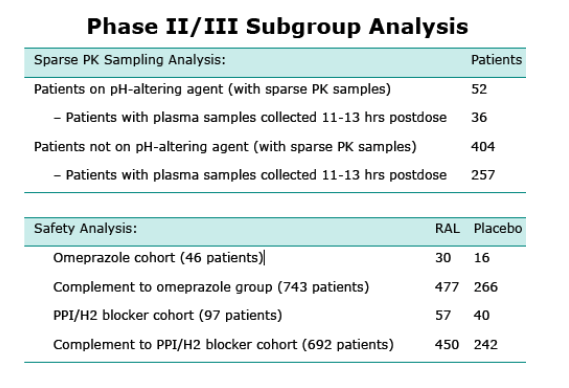
Subgroup analyses were conducted using data obtained from Phase II and III studies of treatment-experienced HIV- infected subjects administered pH-altering agents relative to a complement cohort to evaluate pharmacokinetics and safety of co-administration in the intended patient population.
METHODS



a AUC0-∞, C12hr, and Cmax: model-based geometric mean with between-subject coefficient of variation; Tmax: median and (minimum, maximum); t1/2:
harmonic mean (pseudo standard deviation).
b Geometric mean computed from least squares estimate from an ANOVA performed on the ln-transformed values.
Co-administration of RAL with omeprazole in healthy subjects results in increase in RAL plasma concentration with
-- AUC0-∞ and Cmax increase of approx 3 to 4-fold
-- C12 hr increase of approx 1.5-fold
Similar values for t1/2 and median Tmax of RAL were observed whether RAL administered alone or with omeprazole
Summary of Effects of Various pH-Altering Concomitant Medications on RAL Exposure Measures in HIVInfected Patients in Phase III Studies
Note: Raltegravir (400 mg twice daily) was administered with OBT; CI=Confidence interval; GMR=Geometric mean ratio.
a With pH altering agent: subjects who were on this agent at study entry and had at least one post-randomization dose. Without pH altering agent: otherwise.
The effect of omeprazole on RAL pharmacokinetics in the patient population was smaller than observed in healthy subjects
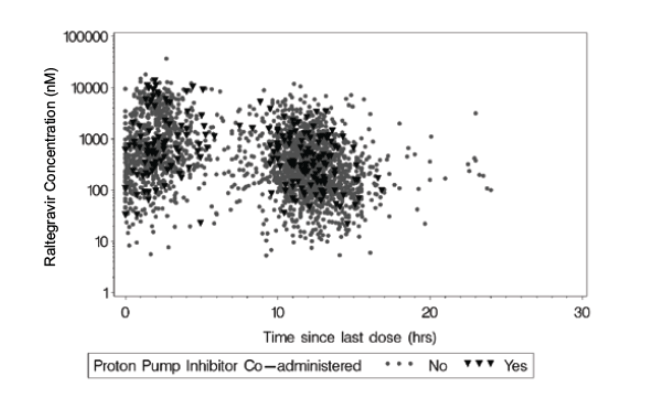
Raltegravir Plasma Concentrations from Sparse Sampling in Phase III Studies for Patients Taking or Not Taking a Proton Pump Inhibitor at the Time of Pharmacokinetic Sample Collection

References
[1] Grinsztejn B, Nguyen BY, Katlama C, et al. Safety and efficacy of the HIV-1 integrase inhibitor raltegravir (MK-0518) in treatment- experienced patients with multidrug-resistant virus: a phase II randomized controlled trial. Lancet 2007; 369:1261-69.
[2] Steigbigel RT, Cooper, DA, Kumar PN, et al. Raltegravir with optimized background therapy for resistant HIV-1 infection. N Engl J Med 2008; 359:339-354.
|
| |
|
 |
 |
|
|