 |
 |
 |
| |
Pharmacokinetic/Pharmacodynamic (PK/PD) Analyses for Raltegravir (RAL) in Phase III Studies in Treatment Experienced HIV- Infected Patients Following 48 Weeks of Treatment
|
| |
| |
Reported by Jules Levin
ICAAC/IDSA Meeting
Oct 2008 Washington, DC
Larissa A Wenning, Eunhee Hwang, Bach-Yen Nguyen, Hedy Teppler, Robert Danovich, Marian Iwamoto, John A Wagner, Deborah Panebianco, Julie A Stone
Merck & Co., Inc., Whitehouse Station, NJ;
Copyright 2008 Merck & Co., Inc., Whitehouse Station, New Jersey, USA, All Rights Reserved
AUTHOR CONCLUSIONS
Some relationships have been identified between RAL Call and (to a lesser extent) Cmin , but not GM C12hr, and treatment outcome at Week 48 in treatment experienced patients in BENCHMRK 1 and 2
The identified relationships are generally shallow for patients with additional active agents in OBT
PK appears to have less influence on treatment outcome than other covariates (e.g. baseline HIV RNA; use of other active agents in OBT)
The current PK/PD analyses do not suggest a threshold RAL concentration associated with reduced efficacy
ABSTRACT
Background: RAL is an HIV-1 integrase strand transfer inhibitor with potent in vitro and in vivo activity. PK/PD analyses were performed to examine potential relationships between RAL PK and viral response in treatment experienced patients.
Methods: Plasma samples were collected during 2 Phase 3 studies (RAL dose
400 mg twice daily plus optimized background therapy (OBT)) during visits at Weeks 4 through 32 (1 sample per visit; median 6 samples per patient). A population PK model could not be developed due to interoccasion variability in absorption, and so 3 measures of observed concentrations were reported for each patient: Geometric Mean (GM) C12hr (GM of samples collected between 11 and 13 hrs postdose); Call (GM of all samples) and Cmin (minimum of all samples). Efficacy parameters HIV RNA <400 c/mL, HIV RNA <50 c/mL, virologic failure, and resistance mutations at amino acid 148 and/or 155 following 48 weeks of treatment were used for the PK/PD analyses. Statistical analyses of potential relationships between RAL PK and efficacy outcomes were performed using logistic regression models.
Results: GM C12hr showed little or no association across the efficacy responses. Call and to a lesser extent Cmin were associated with efficacy responses (HIV RNA <400 and <50 c/mL; occurrence of virologic failure; development of mutations at amino acid 155; and change from baseline in log10 HIV RNA) after adjustment for other covariates. Some covariates, e.g., baseline HIV RNA and naïve use of darunavir, were far more influential on efficacy outcome than PK.
Conclusions: Some relationships have been identified between RAL Call and Cmin, but not GM C12hr, versus treatment outcome. The relationships are generally shallow for patients with additional active agents in OBT. The current PK/PD analyses do not suggest a threshold RAL concentration associated with reduced efficacy.
Introduction / Background
RAL is a novel HIV-1 integrase strand transfer inhibitor with potent in vitro and in vivo activity against HIV-1.
RAL is primarily metabolized by glucuronidation via UGT1A1.
RAL pharmacokinetics similar across
-- Gender, race, age (adults), HIV infection status, hepatic function, renal function, and body mass index
RAL PK demonstrates a relatively large degree of interoccasion variability in absorption
--- Large degree of dose-to-dose variation in Tmax and Cmax
--- No covariates (e.g., gender, race, age, food status) identified which adequately explain variability
Development of a population PK model was problematic
Study Design
Plasma samples collected during 2 Phase III studies in treatment experienced patients (BENCHMRK 1 and BENCHMRK 2)
--- RAL dose 400 mg twice daily plus optimized background therapy (OBT)
Samples collected at Weeks 4, 8,16, and 32 regardless of time of dose and at Weeks 12 and 24 just prior to AM dose (1 sample per visit)

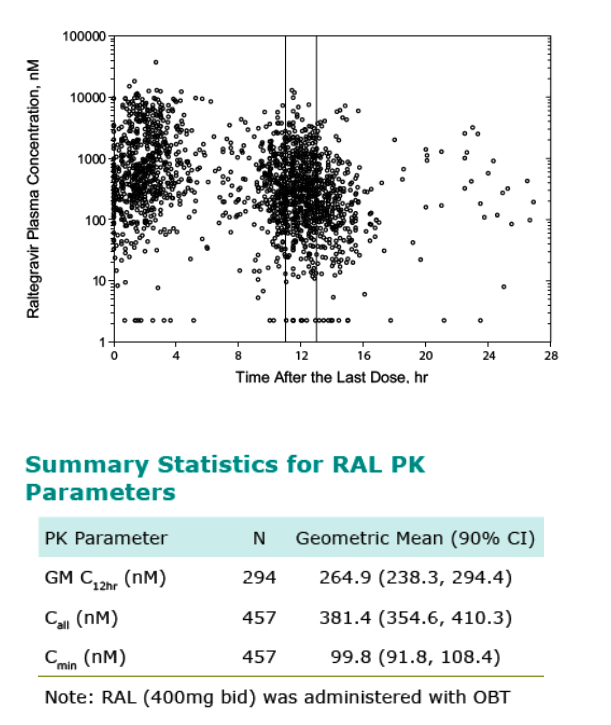
GM C12hr measurement reflects median of 2 samples/ patient; ~60% of patients have value
Call measurement reflects median of 6 samples/patient; 457 of 462 patients have value
Little to no relationship identified between GM C12hr and any PD endpoint
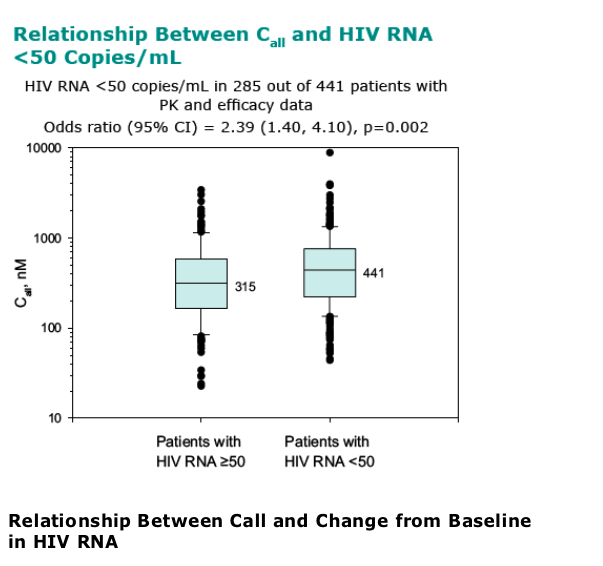
Statistically significant relationships identified between many PD endpoints and Call; Call appears to be the PK parameter best correlated with efficacy
outcomesHIV RNA <400 copies/mL
-- HIV RNA <50 copies/mL
-- Virologic failure
-- Mutation at AA 155
-- Change from baseline in HIV RNA
PK appears less influential than other covariates (e.g. baseline HIV RNA; use of other active agents in OBT)
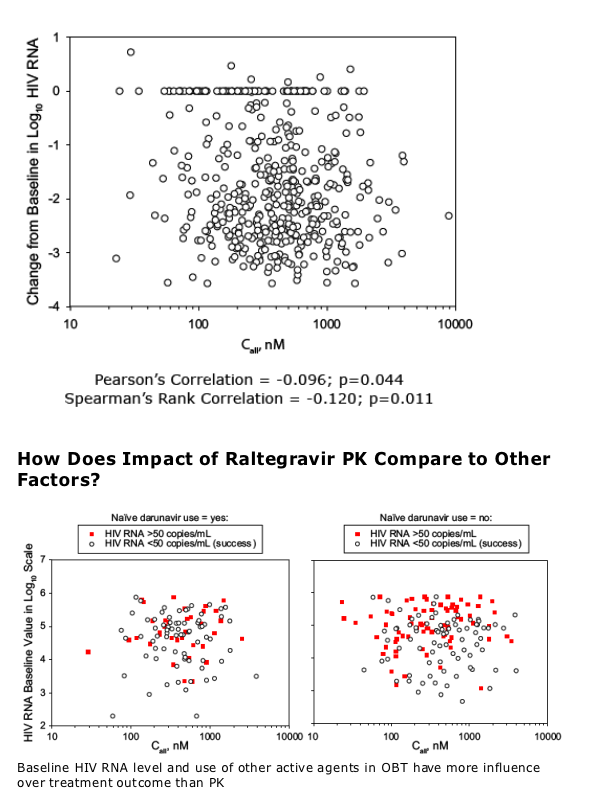
Naïve Darunavir: No
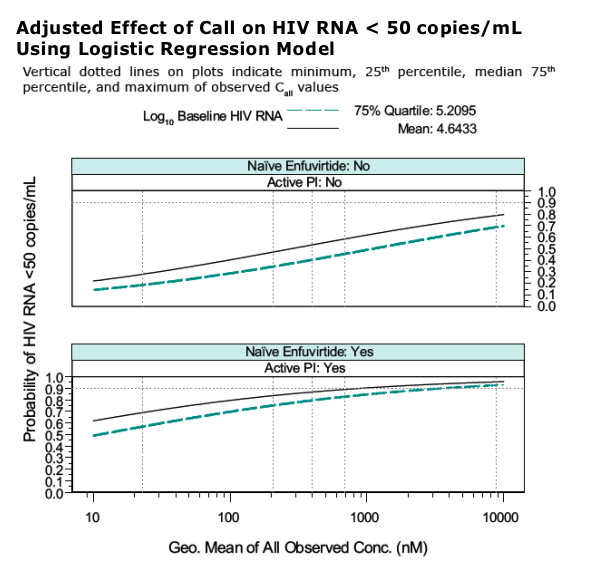
Influence of PK on outcome is different for different subpopulations
|
| |
|
 |
 |
|
|