 |
 |
 |
| |
DELPHI: Safety, tolerability and efficacy of darunavir/ritonavir over 48 weeks
in ARV treatment-experienced children and adolescents
Final analysis
|
| |
| |
Reported by Jules Levin
ICAAC/IDSA, Oct 27, 2008 Wash DC
S Blanche, R Bologna, P Cahn, S Rugina,
P Flynn, C Fortuny, P Vis, V Sekar,
B Van Baelen, S Spinosa-Guzman
DELPHI: Author Conclusions
Final Week 48 analysis
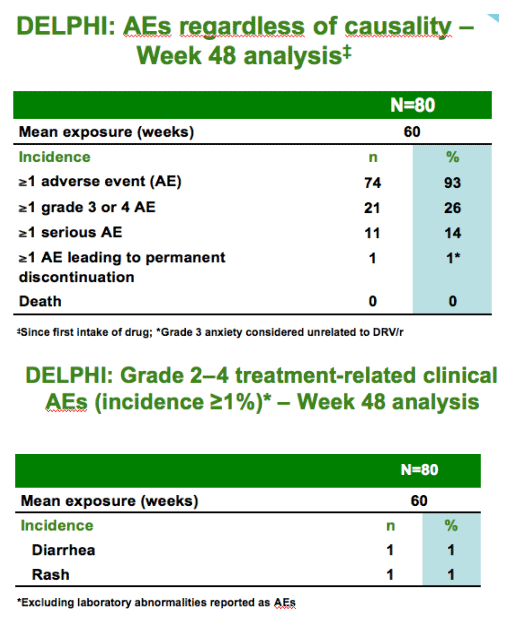
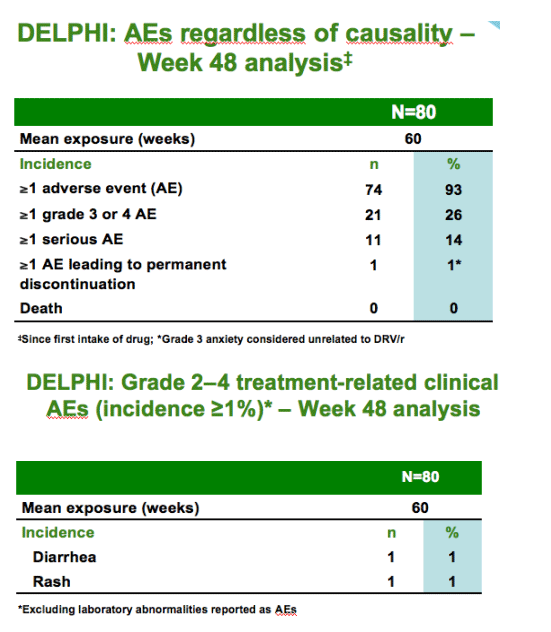
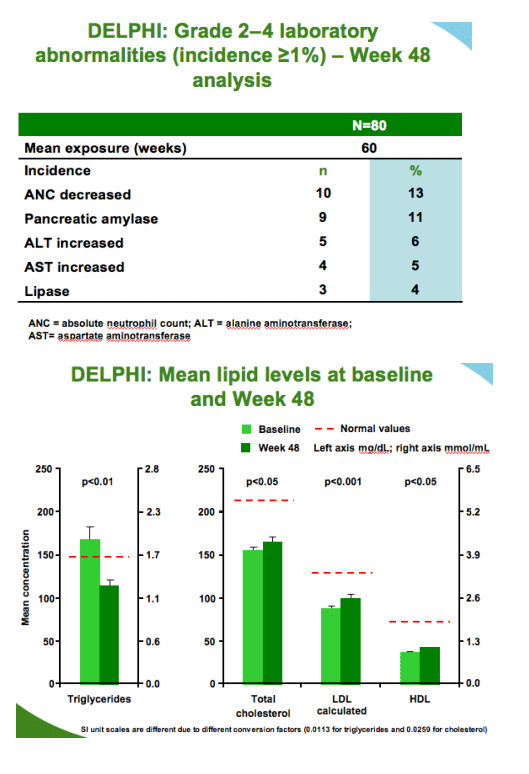
In treatment-experienced, HIV-1-infected children and adolescents aged 6-17
years, DRV/r showed
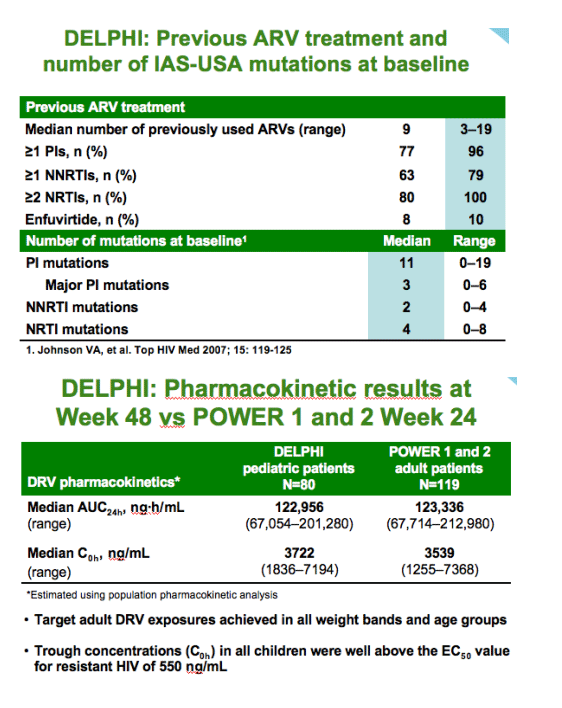
--comparable exposure to adults with appropriate dose selection
--favorable safety and tolerability
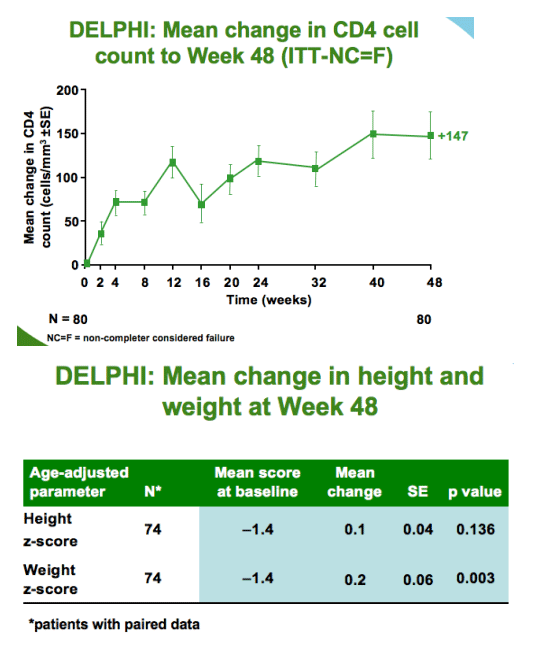
--positive changes in body weight
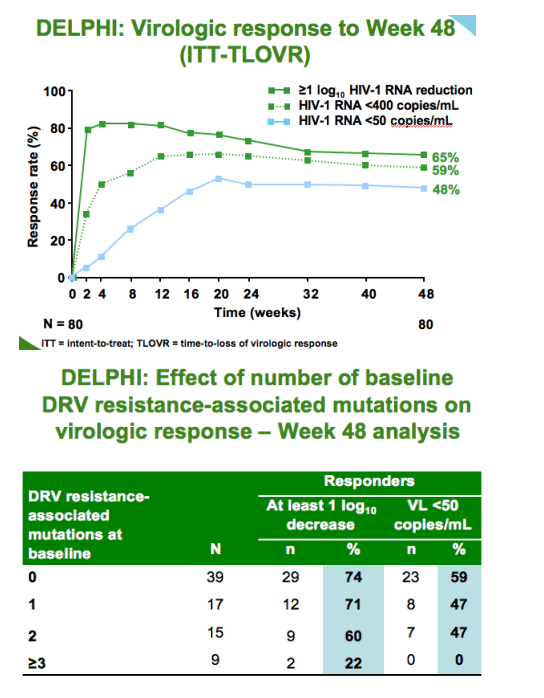
--good virologic response rates in the presence of <3 DRV resistance-associated mutations
DRV/r is a valuable therapeutic option for treatment-experienced, HIV-1-infected pediatric patients
|
| |
|
 |
 |
|
|