 |
 |
 |
| |
Efficacy and safety of switching from Lopinavir/r to Atazanavir/r in suppressed patients receiving a LPV/r containing HAART: ATAZIP 96 weeks results
|
| |
| |
Reported by Jules Levin
Ninth International Congress on Drug Therapy in HIV Infection
09-13 November 2008 Glasgow, UK
Mallolas J.1, Podzamczer D.2, Domingo P.3, Echevarria P.4, Ribera E.5, Gutierrez F.6, Knobel H.7, Cosin J.8, Ferrer E.2, Arranz J.A.9, Roca V.10, Pich J.11, de Lazzari E.12, Gatell J.M.1, for the ATAZIP study group
Background
ATV is a potent, well-tolerated, once-daily (QD) protease inhibitor (PI) extensively studied in treatment-naive and -experienced patients
The SWAN study (BMS 097)1demonstrated that switching from PI ± RTV-containing regimens to an unboostedATV-containing regimen maintained virologic suppression with improvement in plasma lipids through 48 weeksin patients without previous failures to PI contaningregimens
One year outcomes provided comparable efficacy and safety profiles between study arms with improved lipid parameters in patients switching to ATV/r2
1Gatell et al. Clin Infect Dis 2007
2Mallolas et al. IAS 2007, Sydney
Objective
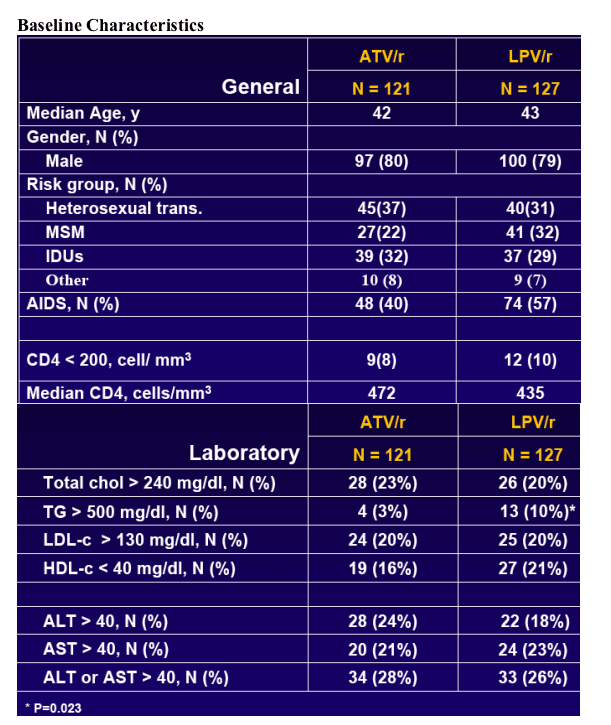
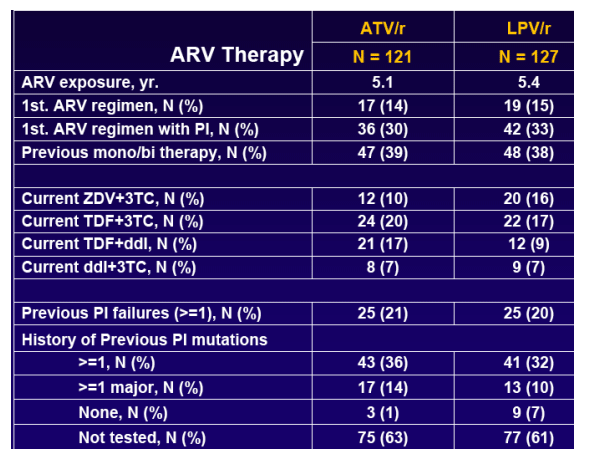
To assess the safety and efficacy of ATV/r based HAART in virologically suppressed patients receiving a LPV/r contaningregimen including patients with previous failures (< 3) to PI regimens or history of having PI associated mutations (< 5) prior to starting the LPV/r-based regimen.
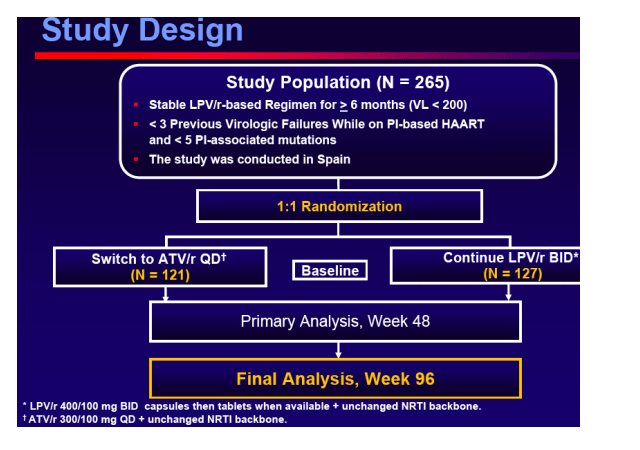
Primary end-point
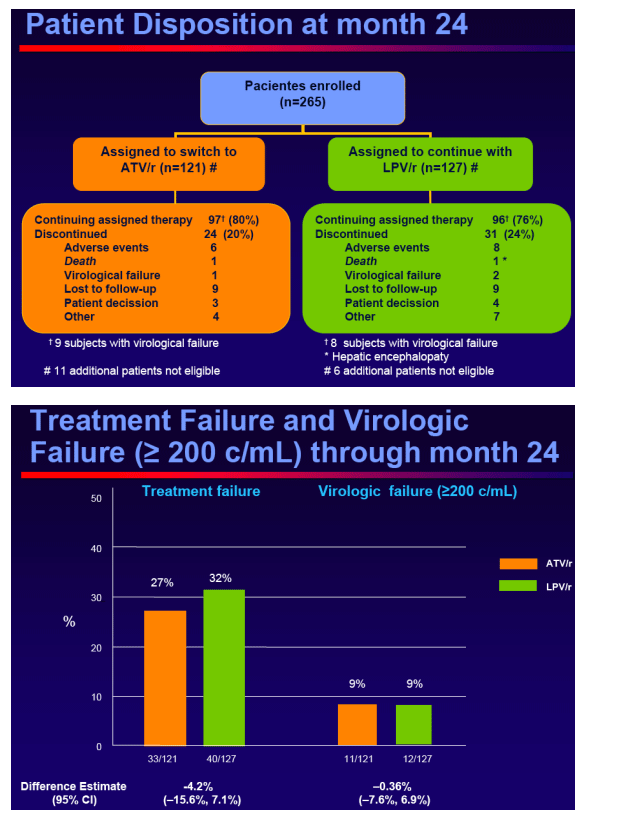
The proportion of patients with treatment failure for any reason through Week 48Includes virologic rebound (> 200 cp/mL), discontinuation of study therapy or lost to follow- up, progression to a new CDC event or death.
Non-inferiority study. Upper limit of 95% CI of estimated difference < 12.5%
Secondary end-points
The proportion of patients with virologic rebound at Week 48 and at Week 96
--Confirmed on-study HIV RNA ≥ 200 copies/mL or last on-study HIV RNA ≥ 200 copies/mL followed by discontinuation
Time to treatment failure and to virologicfailure at Week 96
Safety
Evolution of fasting plasma lipids
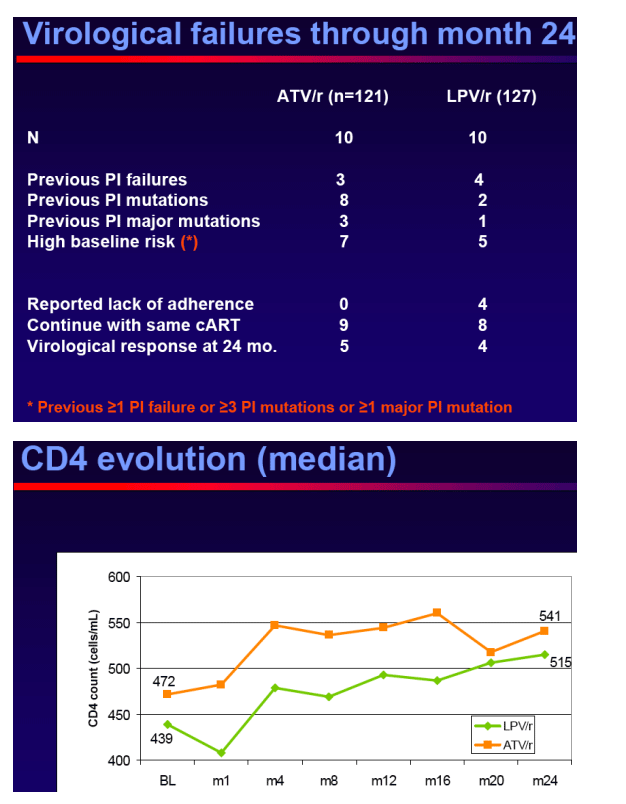
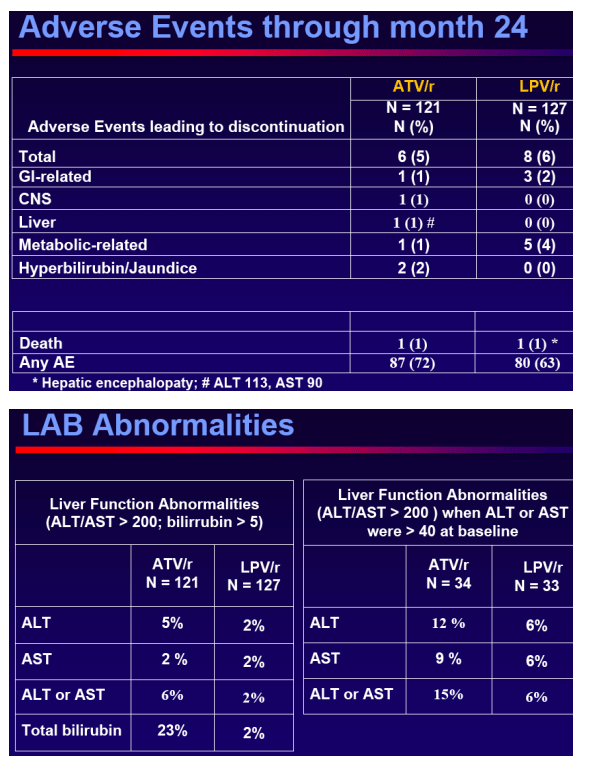
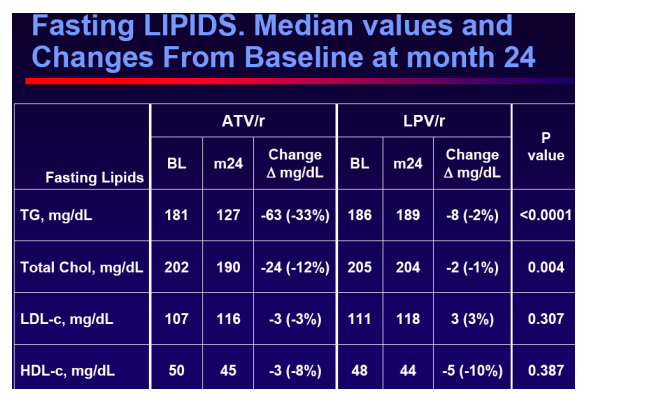
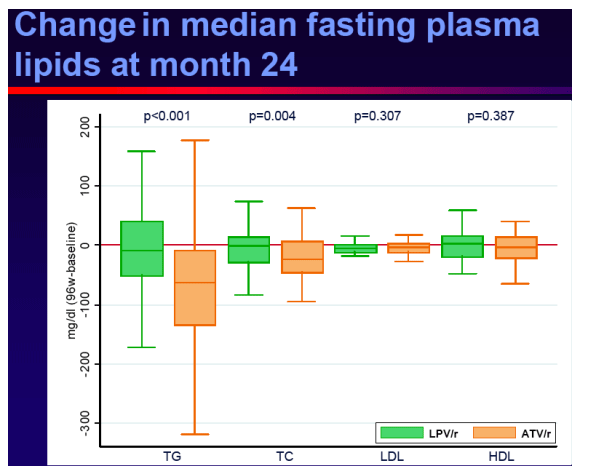
Conclusions
Switching to ATV/r in virologicallysuppressed patients who were receiving a LPV/r containing HAART provided durable and comparable efficacy outcomes, no unexpected safety events and improved lipid parameters
|
| |
|
 |
 |
|
|