 |
 |
 |
| |
Simplification of Antiretroviral Therapy with Efavirenz/Emtricitabine/Tenofovir DF Single Tablet Regimen vs. Continued Antiretroviral Therapy in Suppressed, HIV-1-Infected Patients
|
| |
| |
Reported by Jules Levin
HIV9
9th International Congress on Drug Therapy in HIV Infection
November 9-13, 2008 Glasgow, UK
B Young,1 E DeJesus,2 J Morales-Ramirez,3 J Flaherty,4 R Ebrahimi,4 J-F Maa,5 D McColl,4 D Seekins,5 and A Farajallah5 for the AI266073 Study Team
1Denver Infectious Disease Consultants, Denver, CO; 2Orlando Immunology Center, Orlando, FL; 3Clinical Research Puerto Rico, San Juan, PR; 4Gilead Sciences, Inc., Foster City, CA; 5Bristol-Myers Squibb, Princeton, NJ
BACKGROUND
Co-formulated EFV/FTC/TDF is the first once daily single tablet antiretroviral (ARV) regimen approved in the United States, Canada, and the EU
The components have demonstrated long-term efficacy and safety in treatment naive patients (Arribas JR et al. JAIDS 2008;47:74-78)
This study evaluated whether suppressed patients who simplify therapy to a single tablet regimen of EFV/FTC/TDF have similar effectiveness (efficacy,
safety and tolerability) vs. patients who remain on their unmodified ARV regimen through 48 weeks
AUTHOR CONCLUSIONS
In this study of virologically suppressed patients, through 48 weeks:
High rates of virologic suppression were maintained in both treatment arms
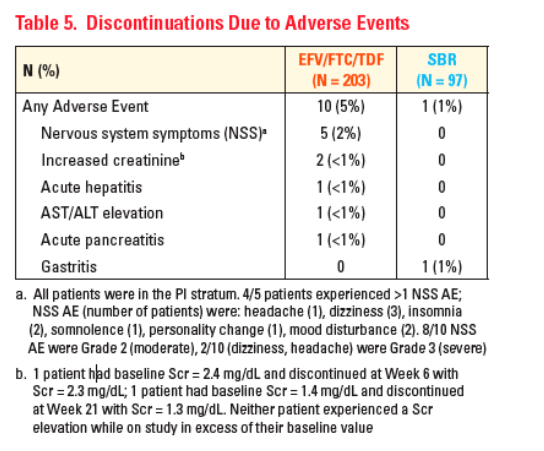
Simplification to EFV/FTC/TDF was well tolerated with low rates of
discontinuations observed in both treatment arms
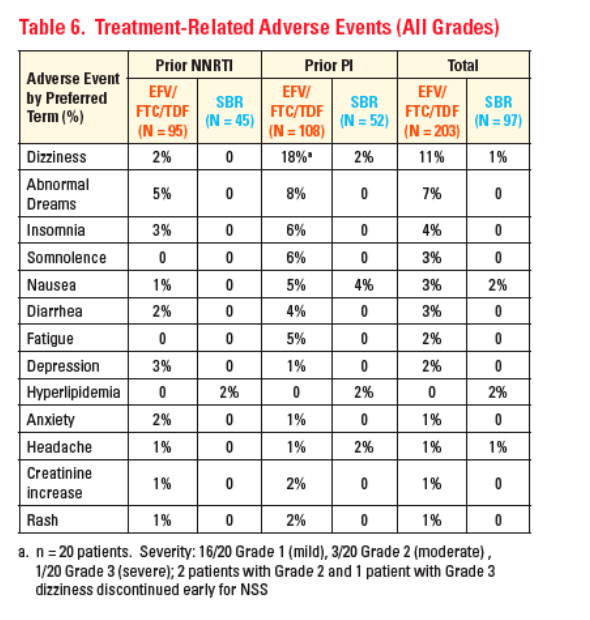
Consistent with previous reports, the most frequently reported AEs in the
EFV/FTC/TDF arm were nervous system symptoms primarily in patients naïve to EFV
-- These were transient, generally mild, and resulted in few treatment discontinuations
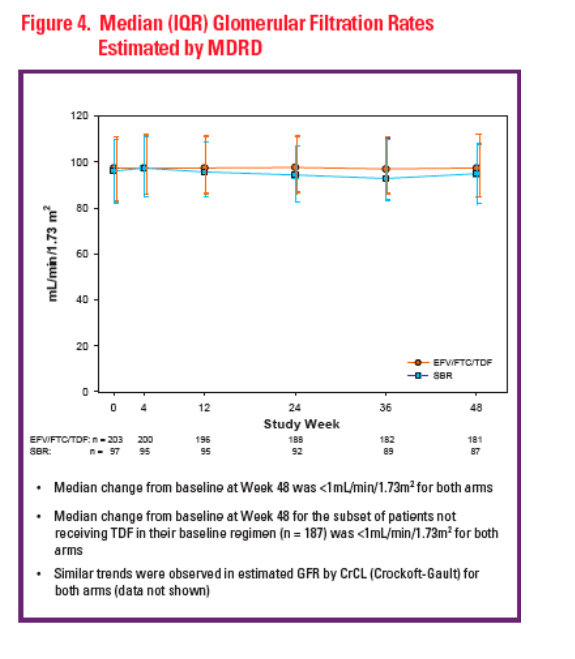
Renal function remained stable through 48 weeks, including patients naïve to TDF at baseline
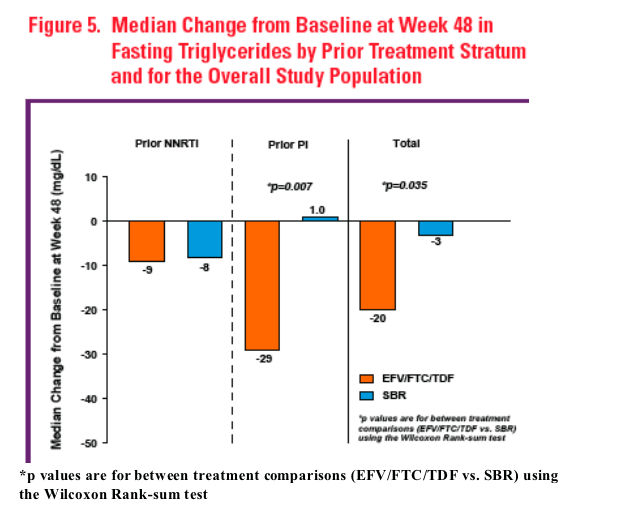
Patients randomized to EFV/FTC/TDF, particularly those on a prior PI containing
regimen, showed significant declines in triglycerides compared to unmodified
therapy
METHODS
Key Inclusion / Exclusion Criteria
HIV-1 RNA < 200 copies/mL for ≥ 3 months on current ARV regimen
Receiving first ARV regimen, or documented suppression on a previous PI-based regimen at time of prior change in therapy
Calculated CrCL ≥ 60 mL/min (by Cockcroft-Gault formula)
Patients with known resistance to study agents and those receiving individual components of the single tablet regimen (EFV+FTC+TDF) were excluded
Study Assessments
HIV-1 RNA, CD4 count, chemistries, CBC performed at baseline and Weeks 4, 12, 24, 36, and 48
GFR estimated by calculated CrCL (Cockcroft-Gault) and Modified Diet in Renal Disease (MDRD)
Patient-reported outcomes:
-- Refer to Poster # P063 (Hodder S, et al.)
Statistical Methods
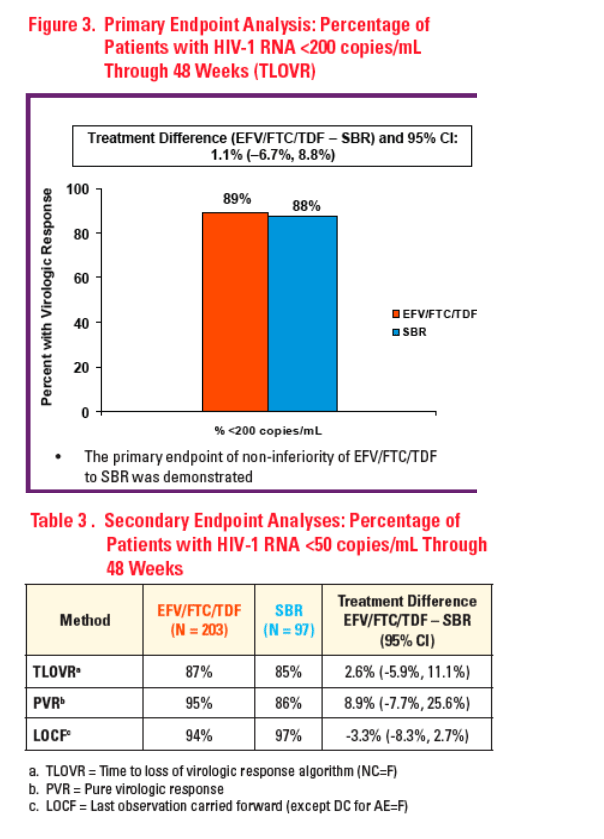
Sample size was estimated using a non-inferiority margin ∇ of 15% at 80% power. EFV/FTC/TDF was to be declared non-inferior to SBR if the lower confi dence boundary of the difference (EFV/FTC/TDF - SBR) for the primary endpoint was greater than -0.15
Assessments were based on intent-to-treat (ITT) analysis*
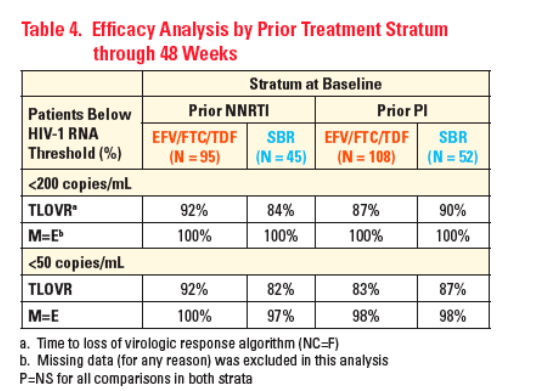
Primary endpoint: HIV-1 RNA < 200 copies/mL through 48 Weeks based on TLOVR, assuming non-completers equal failures (NC=F)
-- Responders were those with HIV-1 RNA < 200 copies/mL at Week 48 without HIV-1 RNA ≥ 200 copies/mL on 2 successive occasions, or without last HIV-1 RNA ≥ 200 copies/mL while on-study followed by DC
Secondary endpoints were the proportion of patients with HIV-1 RNA <50 copies/mL by: TLOVR (NC=F), Pure Virologic Response (PVR) [estimated by Kaplan-Meier
where responders were defi ned as those without 2 consecutive HIV-1 RNA ≥50 copies/mL or without last HIV-1 RNA ≥50 copies/mL followed by DC], and Last Observation Carried Forward (LOCF)** [where those with HIV-1 RNA ≥50 copies/mL or those who DC early due to an adverse event were treated as failures]
*ITT population included all patients randomized and who received at least one dose of study medication
**LOCF endpoint was defi ned post-hoc
CD4 Responses
Median change from baseline in CD4 cell count at Week 48 was 3 cells/μL and 9 cells/μL for EFV/FTC/TDF and SBR, respectively

|
| |
|
 |
 |
|
|