 |
 |
 |
| |
Atazanavir Is Safe and Efficacious
in HBV and HCV Coinfected Patients: Results of AI424138 (CASTLE)
|
| |
| |
Reported by Jules Levin
Ninth International Congress on Drug Therapy in HIV Infection, November 9-13, 2008, Glasgow, UK
J. Absalon,1 G. Thal,2 A. Thiry,1 R. Yang,1 M. Mancini,1 D. McGrath1 for the BMS AI424138 Study Group 1Bristol-Myers Squibb, Wallingford, CT, USA; 2Bristol-Myers Squibb, Lawrenceville, NJ, USA
AUTHOR CONCLUSIONS
Virologic and immunologic responses were comparable in patients chronically infected with HBV or HCV infection treated with ATV/RTV or LPV/RTV.
Virologic and immunologic responses were also similar in hepatitis coinfected and hepatitis uninfected patients in both the ATV/RTV and LPV/RTV treatment arms.
ATV/RTV had a more favorable lipid profile and fewer GI AEs among hepatitis coinfected patients than LPV/RTV. Lipid profiles were similar in hepatitis coinfected and hepatitis uninfected patients.
Although the overall rates of elevated transaminases in hepatitis coinfected patients were low in this study compared with those observed in other clinical trials, a small number of patients receiving ATV/RTV and none receiving LPV/RTV had grade 3 to 4 AST elevations. The cause of this higher proportion in the ATV/RTV-treatment arm among this limited number of patients is unclear.
With monitoring of liver function tests, ATV/RTV can be considered as part of HAART among ART-naïve patients with chronic HBV or HCV infection.
Introduction
Chronic hepatitis B (HBV) and hepatitis C (HCV) viral coinfections are 2 of the most common comorbidities in HIVinfected patients, and can complicate antiretroviral therapy (ART).1,2 Therefore, it is important to define the efficacy and safety of ART in HIV patients coinfected with HBV and HCV.
Atazanavir (ATV) is a potent, generally well-tolerated, once-daily HIV-1 protease inhibitor (PI) extensively studied in ART-naïve and ART-experienced patients and is a common component of highly active ART (HAART).3,4
The CASTLE study, an international randomized clinical trial in 134 sites in 29 countries, showed that in combination with tenofovir/emtricitabine (TDF/FTC), ATV/ritonavir (RTV) is noninferior to lopinavir (LPV)/RTV in antiviral efficacy in ART-naïve patients at 48 weeks, with significantly less elevation of lipids and better gastrointestinal (GI) tolerability.5
This large-scale study included HIV-infected, ART-naïve patients who were coinfected with chronic HBV and/or HCV at study entry, providing the opportunity to assess the impact of hepatitis coinfection on the efficacy and safety profiles of the 2 most commonly used boosted PIs in HIV treatment.
Objective
To compare the efficacy and safety of ATV/RTV- and LPV/RTV-based regimens in HIV-infected patients with and without chronic HBV or HCV coinfection using 48-week data from the CASTLE study.
Methods
CASTLE is a randomized, open-label, prospective study comparing once-daily ATV/RTV with twice-daily LPV/RTV, both in combination with fixed-dose TDF/FTC in 883 ART-naïve HIV-infected patients (Figure 1).
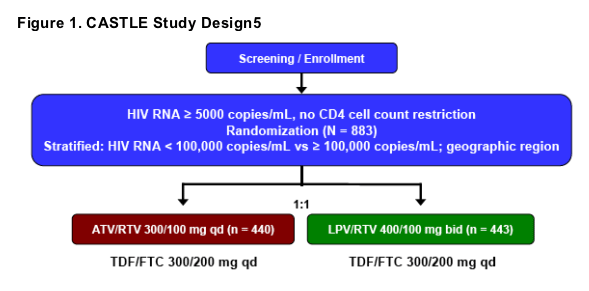
Through the first 48 weeks of the study, the protocol required patients to receive the capsule formulation of LPV/RTV.
The primary end point was the proportion of patients with HIV RNA < 50 copies/mL at Week 48.
Secondary assessments included CD4 cell count change from baseline and safety parameters (adverse events [AEs] and laboratory tests [eg, serum chemistry and hematology, fasting lipid profile]).
Efficacy and safety analyses by HBV and HCV coinfection status for each treatment arm were prespecified.
RESULTS
Baseline Characteristics
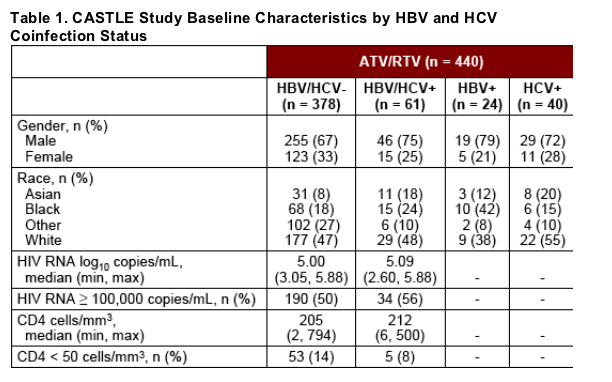
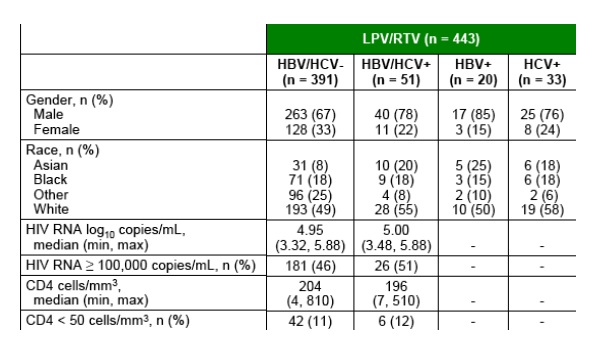
Baseline characteristics among patients with and without chronic hepatitis coinfection are presented in Table 1.
At baseline, 13% of patients in total were infected with HBV or HCV: 5% with HBV infection, 8% with HCV infection, and < 1% with both HBV and HCV infection.
Efficacy: Virologic and Immunologic Response
Overall Population
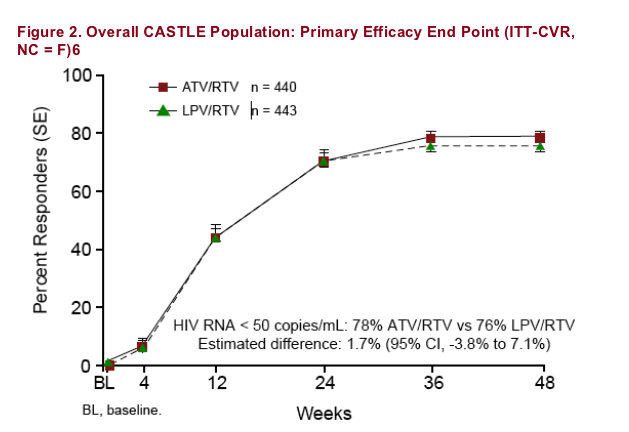
Overall, once-daily ATV/RTV-based HAART demonstrated similar efficacy to twice-daily LPV/RTV-based HAART: 78% of patients on ATV/RTV and 76% on LPV/RTV achieved HIV RNA < 50 copies/mL at Week 48 (difference estimate 1.7% [95% CI, -3.8% to 7.1%]) using an intent-to-treat (ITT) analysis, confirmed virologic response (CVR) noncompleter = failure (NC = F) (Figure 2).
By Hepatitis Coinfection Status
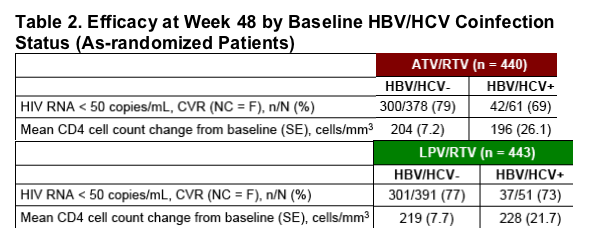
Virologic and immunologic responses were similar in hepatitis uninfected and coinfected patients in both the ATV/RTV and LPV/RTV treatment arms (Table 2).
-- Response was comparable in hepatitis coinfected patients treated with either ATV/RTV or LPV/RTV (Table 2).
Safety
Liver Function
Among hepatitis coinfected patients, incidence of grade 2 to 4 treatment-related hyperbilirubinemia was 15% versus 0% and jaundice was 3% versus 0% in ATV/RTV-treated patients versus LPV/RTV-treated patients, respectively.
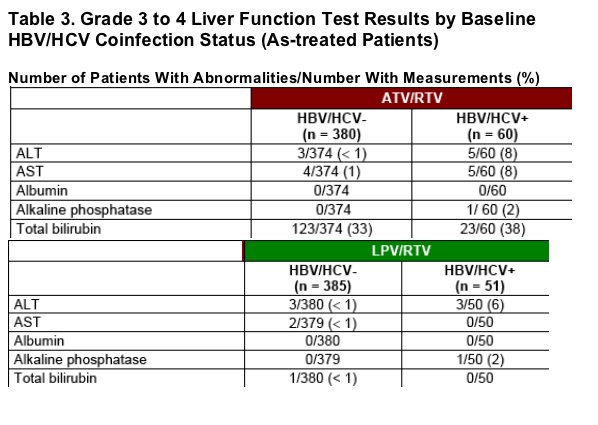
Grade 3 to 4 liver function abnormalities among hepatitis coinfected patients were similar between treatment arms except for aspartate aminotransferase (AST) and bilirubin, which were greater among ATV/RTV-treated patients (Table 3).
Adverse Events
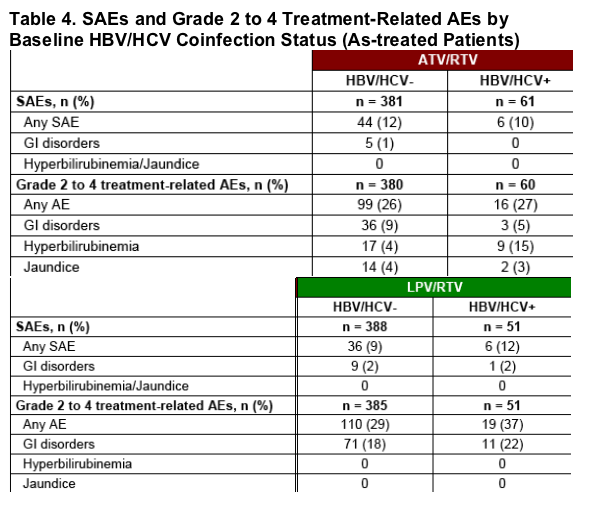
The rates of serious AEs (SAEs) and grade 2 to 4 treatment-related AEs are reported in Table 4.
Few SAEs were reported among hepatitis coinfected patients in either treatment arm.
GI AEs occurred at a higher frequency in both hepatitis uninfected and coinfected patients receiving LPV/RTV compared with those receiving ATV/RTV.
There was a higher rate of grade 2 to 4 jaundice and hyperbilirubinemia observed with ATV/RTV than LPV/RTV treatment, with a higher rate of hyperbilirubinemia observed in hepatitis coinfected versus uninfected patients.
The incidence of severe jaundice and hyperbilirubinemia was low in both treatment groups and among both hepatitis uninfected and coinfected patients.
Lipid Parameters
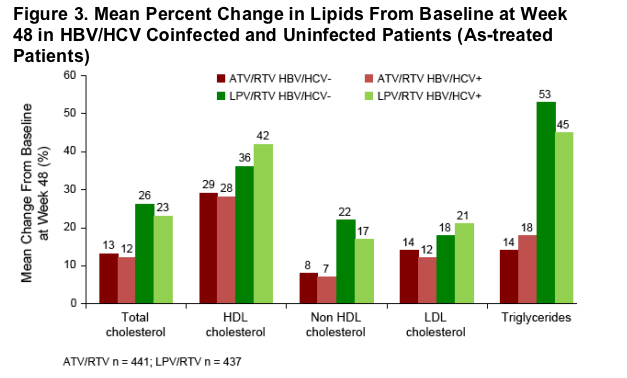
Hepatitis uninfected and coinfected patients treated with ATV/RTV had a more favorable lipid profile compared with LPV/RTV-treated patients (Figure 3). The mean percent change from baseline in total cholesterol, low-density lipoprotein (LDL) cholesterol, non-high-density lipoprotein (HDL) cholesterol, and triglycerides was lower in ATV/RTV-treated patients compared with LPV/RTV-treated patients.
Lipid profiles were similar in hepatitis uninfected and coinfected patients in both the ATV/RTV and LPV/RTV treatment arms (Figure 3).
|
| |
|
 |
 |
|
|