 |
 |
 |
| |
Metabolic Evaluation of Study M05-730 through Week (Wk) 48: Phase 3, Randomized, Open-Label Study of Lopinavir/ritonavir (LPV/r) Tablets Once Daily (QD) versus Twice Daily (BID), Co-Administered with Tenofovir DF (TDF) + Emtricitabine (FTC) in Antiretroviral-Naïve (ARV) HIV-1 Infected Subjects
|
| |
| |
Reported by Jules Levin
BA da Silva, Y-L Chiu, J Li, P Noertersheuser, W Awni, C Klein, T Doan, B Bernstein
Abbott Laboratories, Abbott Park, IL, USA ♦ HIV 9 ♦ 9-13 November 2008 ♦ Glasgow, UK
Author Summary
In this study of antiretroviral-naïve HIV-infected subjects starting a LPV/r-based antiretroviral regimen:
There were increases in all lipid parameters through 48 weeks, including HDL levels. Importantly, however:
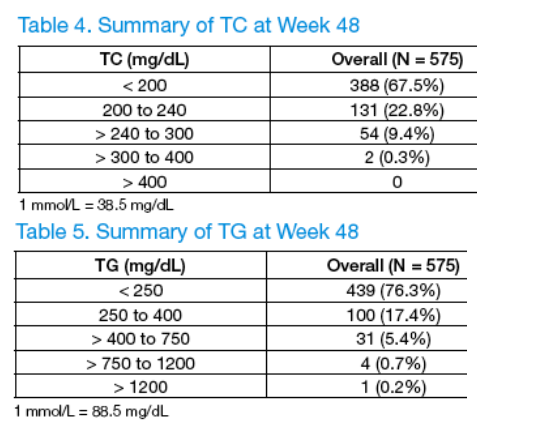
-- Median lipid values for TC, TG, and LDL stayed within the acceptable NCEP or clinically relevant values through 48 weeks. This was reflected by the fact that if subjects came into the study with an acceptable TG level at BL (< 2.825 mmol/L), 80% maintained these levels at Wk 48. Also, if subjects came into the study with an acceptable TC level at BL (< 5.2 mmol/L), 72% maintained these levels at Wk 48. In addition, if subjects came into the study with an acceptable LDL level at BL (< 3.38 mmol/L), 87% maintained these levels at Wk 48.
-- The proportion of subjects with low HDL (<1.04 mmol/L) by NCEP decreased from 60% to 33% from BL to Wk 48.
Although statistically significant differences were noted between QD and BID mean changes from BL to Week 48 for TC and non-HDL parameters, the values were not deemed clinically significant.
As previously stated, lipid ratios are important tools when assessing a patient's overall CV risk. LDL:HDL ratio decreased significantly through 48 weeks, and there was no change in TC:HDL ratio through 48 weeks.
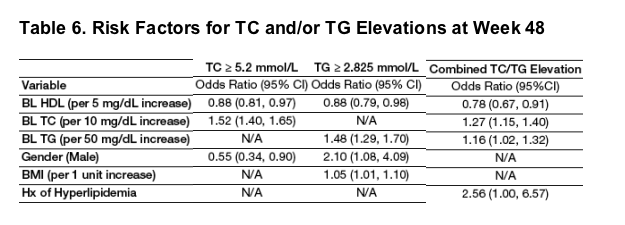
Higher BL TC and TG levels were noted to be risk factors for elevations to >5.2 mmol/L TC and to >2.825 mmol/L TG, respectively.
Interestingly, low BL HDL level was also noted to be a risk factor for elevations in TC, TG and both TC/TG. While gender played a role in TC or TG increases, when both parameters (increased TC and TG) were evaluated, gender was not noted to be a risk factor.
Mean HOMA values did not significantly change from baseline to Week 48 (overall mean change: +0.06, p=0.845), providing additional evidence that when LPV/r is administered long-term, it does not seem to impact insulin sensitivity as determined by HOMA. A similar result was noted in Study M06-613.11
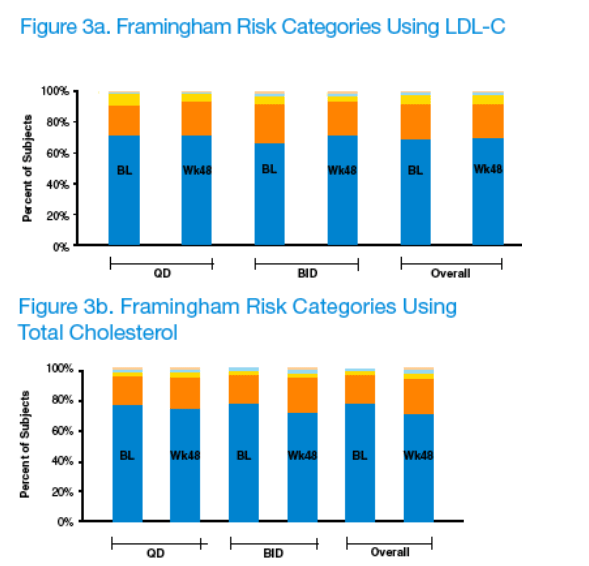
The mean 10-year CV risk, according to Framingham analysis, including the effect of the change in age over 48 weeks, remained unchanged over 48 weeks when the LDL criteria were used (4.63% to 4.60%, p=0.750). When TC was used, the mean 10-year CV risk increased from 4.25% to 5.02% (p<0.001). When the effect of change in age was not included, mean 10-year CV risk using TC increased from 4.25% to 4.77% (p<0.001). While statistically significant, the small mean changes using the TC criteria are not deemed to be clinically significant.
Author Conclusions
We used multiple tools to assess factors that may contribute to CV risk. In this study of antiretroviral-naïve HIV-infected subjects starting a LPV/r-based antiretroviral regimen, we found:
-- In subjects treated with a LPV/r-based regimen through 48 weeks, there was minimal impact on 10-year cardiovascular risk as measured by
TC:HDL and LDL-C:HDL ratios and Framingham risk score.
HOMA did not change through 48 weeks of LPV/r-based treatment, suggesting no increase in insulin resistance.
BACKGROUND
Cardiovascular (CV) risk assessment is an important aspect in the care of HIV-infected patients. This is due to the presence of underlying risk factors for cardiovascular disease in some patients1, the effects of HIV-1 infection and immunosuppression2,3, and the impact of antiretroviral therapy.4
The Framingham Risk Equation is used to assess 10-year risk for coronary heart disease.5 It takes into account multiple factors such as age, gender, total
cholesterol (TC) level, HDL level, smoking status, diabetes status, and blood pressure. Either LDL or TC may be used in calculating the Framingham Risk
Equation.
In addition, increased ratios of both TC:HDL and LDL:HDL have been associated with higher CV risk.6,7
Although not part of the Framingham Equation or the lipid ratios, insulin resistance is also associated with higher CV risk.8
The major impact of LPV/r on lipids is an increase in triglyceride (TG) levels, with smaller increases in TC, LDL, and HDL. Although small, short-term studies have suggested that LPV/r may increase insulin resistance9,10, these findings have not been confirmed in longer-term studies.11
Based on these findings, an evaluation of changes in lipid ratios, tools such as the Framingham risk equation, and an assessment of HOMA (a measure of insulin resistance) may provide a more comprehensive measure of the impact of LPV/r on CV risk.
We therefore evaluated metabolic abnormalities in Study M05-730, the largest LPV/r tablet study in antiretroviral-naïve subjects.
METHODS
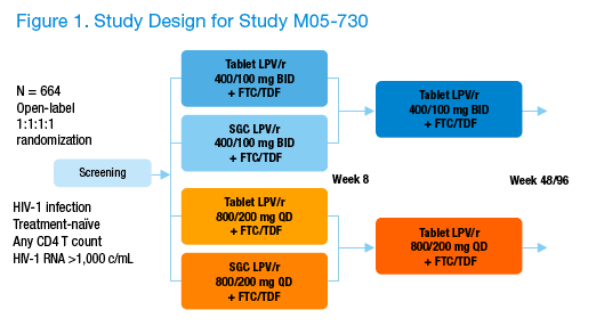
664 ARV-naïve HIV-1-infected subjects were randomized 1:1:1:1 to LPV/r QD SGC, BID SGC, QD tablet, BID tablet for 8 weeks. All subjects received TDF+FTC QD. At Week 8, subjects taking LPV/r SGC were switched to the tablet formulation while maintaining their QD or BID dosing schedule (Figure 1).
Subjects will be followed for up to 96 wks.
Metabolic evaluations at baseline (BL) and Wk 48 are presented.
Baseline demographics and subject disposition through 48 weeks were summarized.
Mean changes in lipid parameters, lipid ratios, HOMA value, and CV risk were tested for significance using paired t-tests and were compared between treatment groups using one-way ANOVA.
Lipid values at BL and Week 48 were summarized with medians and 25-75% interquartile ranges.
Proportions of subjects who met various NCEP criteria and who had various levels of CV risk at Week 48 were calculated.
Multivariate logistic regression was used to identify risk factors for elevations in TC, TG and combined TC/TG at Week 48.
RESULTS
Baseline Demographics
Baseline demographics were comparable between groups (Table 1).
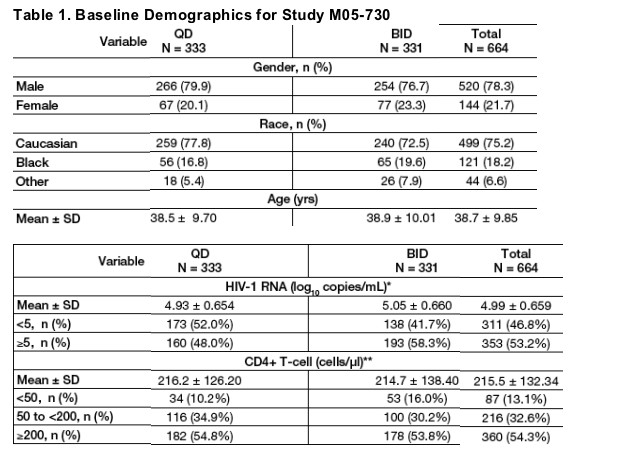
** Statistically significant difference between treatment groups in mean level (p=0.020) and in proportion of subjects with HIV-1 RNA <5 log10 copies/mL (p=0.008).
** One subject (QD group) did not have baseline CD4 determination.
Subject Disposition
Through 48 wks, 15% (QD) and 17% (BID) discontinued prematurely for various reasons. Only two patients discontinued due to either elevated triglyceride (TG) or total cholesterol (TC) levels. One QD subject discontinued with a TG level of 16.45 mmol/L (1456 mg/dL), and one BID subject discontinued with a TC level of 9.56 mmol/L (368 mg/dL).
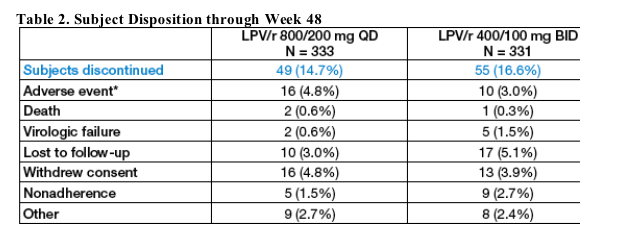
Investigators may have provided more than one reason for a subject's discontinuation. Two subjects discontinued due to either elevated TG or TC.
Changes in Mean Lipid Levels and Lipid Ratios through Week 48
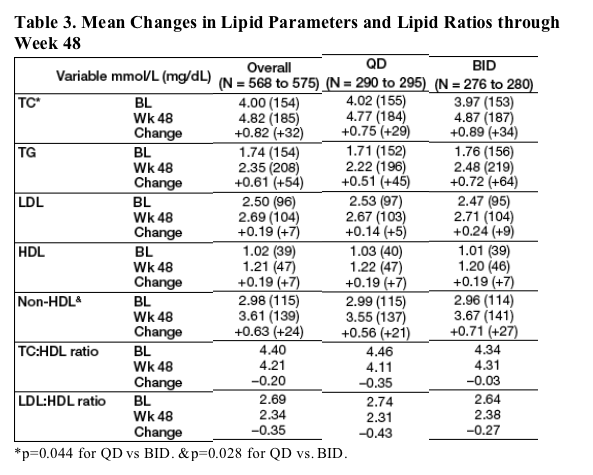
Mean changes from baseline through Week 48 for various lipid parameters are shown in Table 3.
Although statistically significant differences were noted between QD and BID mean changes from BL to Week 48 for TC and non-HDL parameters, the values were not deemed clinically significant.
Notably, among all subjects combined, there was a statistically significant decrease in LDL:HDL ratio (from 2.69 to 2.34, p<0.001) and no change in TC:HDL ratio (from 4.40 to 4.21, p=0.102) through 48 weeks.
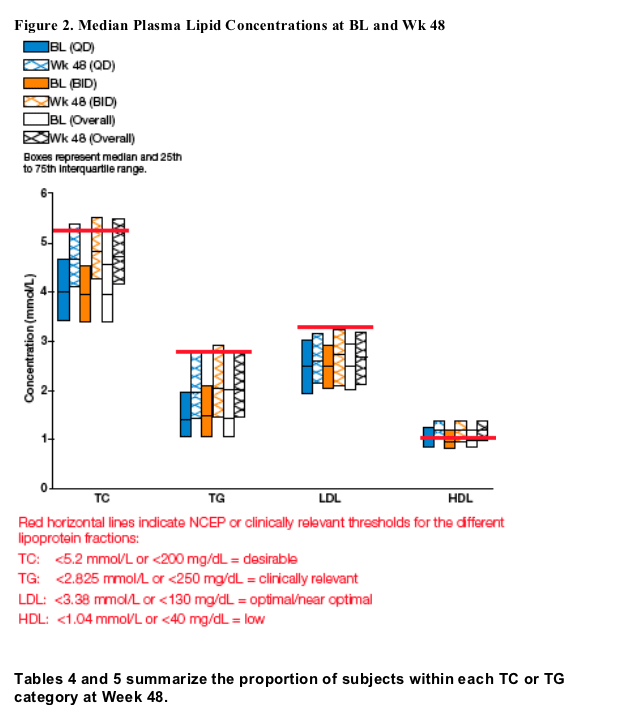
Median Lipid Parameters through Week 48 and Comparison to NCEP Criteria
Median lipid values for TC, TG, and LDL stayed within the acceptable NCEP or clinically relevant values through 48 weeks (Figure 2).
This was reflected by the following:
-- 80% of subjects overall who had TG values < 2.825 mmol/L (250 mg/dL) at BL maintained these levels at Wk 48.
-- Of subjects who had TC in the desirable NCEP range (< 5.2 mmol/L [200 mg/dL]) at BL, 72% maintained these levels at Wk 48.
-- 87% of subjects who had LDL in the optimal/near optimal NCEP range (< 3.38 mmol/L [130 mg/dL]) at BL also had levels in this range at Wk 48.
The proportion of subjects with low HDL (<1.04 mmol/L [40 mg/dL]) by NCEP decreased from 60% to 33% from BL to Wk 48.
Results: Risk Factors for TG and/or TC Increases
Logistic regression analysis was used to identify risk factors for TC ≥ 5.2 mmol/L (200 mg/dL), TG ≥ 2.825 mmol/L (250 mg/dL), and the combination of TC ≥ 5.2 mmol/L and TG ≥ 2.825 mmol/L at Week 48. Results are shown in Table 6.
A 5 mg/dL increase in BL HDL decreased the risk of TC, TG, and combined TC/TG elevations by 12%, 12%, and 22%, respectively.
A 10 mg/dL increase in BL TC increased the risk of TC and combined TC/TG elevations by 52% and 27%, respectively.
A 50 mg/dL increase in BL TG increased the risk of TG and combined TC/TG elevations by 48% and 16%, respectively.
Males had a higher risk of TG elevations and females had a higher risk of TC elevations.
Higher BMI increased the risk of TG elevations, and a history of hyperlipidemia increased the risk of combined TC/TG elevations.
Framingham Risk Equations Estimates at BL and Week 48
The mean 10-year CV risk, including the effect of the change in age over 48 weeks, remained unchanged over 48 weeks when the LDL criteria were used (4.63% to 4.60%, p=0.750). When TC was used instead of LDL, mean 10-year CV risk increased from 4.25% to 5.02% (p<0.001).
When the effect of change in age was not included, the mean 10-year CV risk using TC increased from 4.25% to 4.77% (p<0.001).
Evaluation of HOMA through Week 48
Mean HOMA values did not significantly change from baseline (2.9) to Week 48 (3.0) (overall mean change: +0.06, p=0.845), with no differences between treatment groups.
REFERENCES
1. Saves M, Chene G, Ducimetiere P, et al. Risk factors for coronary heart disease in patients treated for human immunodeficiency virus infection compared with the general population. Clinical Infectious Diseases. 2003;37:292-298.
2. Weber R, Friis-Moller N, Sabin C, et al. HIV and Non-HIV-related Deaths and Their Relationship to Immunodeficiency: The D:A:D Study. 12th Conference on Retroviruses and Opportunistic Infections, Boston, USA 2005. Abstract #595.
3. El-Sadr W and Neaton J. Episodic CD4-Guided Use of ART Is Inferior to Continuous Therapy: Results of the SMART Study. 13th Conference on Retroviruses and Opportunistic Infections, Denver, USA 2006. Abstract #106LB.
4. Post FA and Campbell LJ. Abacavir and increased risk of myocardial infarction. The Lancet. 2008;372:803.
5. Kannel WB, McGee D, Gordon T. A general cardiovascular risk profile: The Framingham Study. Am J Cardiol. 1976;38:46-51.
6. Kinosian B, Glick H, Garland G. Cholesterol and coronary heart disease: Predicting risks by levels and ratios. Ann Int Med. 1994;121(9), 641-7.
7. Criqui MH, Golomb BA. Epidemiologic aspects of lipid abnormalities. Am J Med. 1998;105:48-53.
8. Mondy K, de las Fuentes L, Waggoner A, et al. Insulin resistance predicts endothelial dysfunction and cardiovascular risk in HIV-infected persons on long-term highly active antiretroviral therapy. AIDS. 2008;22:849-856.
9. Noor MA, Flint OP, Maa JF, et al. Effects of atazanavir/ritonavir and lopinavir/ritonavir on glucose uptake and insulin sensitivity: Demonstrable differences in vitro and clinically. AIDS. 2006;20:1813-1821.
10. Lee GA, Seneviratne T, Noor MA, et al. The metabolic effects of lopinavir/ritonavir in HIV-negative men. AIDS. 2004;18:641-649.
11. Cameron DW, da Silva BA, Arribas JR, et al. A 96-week comparison of lopinavir-ritonavir combination therapy followed by lopinavir-ritonavir monotherapy versus efavirenz combination therapy. J Infect Dis. 2008;198:234-240.
|
| |
|
 |
 |
|
|