 |
 |
 |
| |
Effect of extrinsic and intrinsic factors on the pharmacokinetics of darunavir/ritonavir in HIV-1-infected patients: results of a randomized, controlled, Phase III study (ARTEMIS)
|
| |
| |
Reported by Jules Levin
9th International Workshop on Clinical Pharmacology of HIV Therapy
April 7-9, 2008
New Orleans
Vanitha Sekar,1 Carline Vanden Abeele,2 Ben Van Baelen,2 Peter Vis,3 Ludo Lavreys,2 Martine De Pauw,2 Melissa Haley,1 Tony Vangeneugden,2 Sabrina Spinosa-Guzman,2
Richard Hoetelmans2
1Tibotec Inc., Yardley, PA, USA; 2Tibotec BVBA, Mechelen, Belgium; 3Exprimo NV, Mechelen, Belgium
Introduction
· As part of combination antiretroviral treatment, the protease inhibitor darunavir (with low-dose ritonavir; DRV/r) has demonstrated clinical activity in a broad range of HIV-infected patients).1-3
· As an initial treatment for HIV infection, DRV/r-based therapy was shown to be noninferior to lopinavir/ritonavir (LPV/r)-based therapy in the 48-week primary efficacy analyses of the ARTEMIS (AntiRetroviral Therapy with TMC114 ExaMined In naive Subjects; TMC114-C211) trial3
- 84% of DRV/r and 78% of LPV/r patients achieved the primary efficacy endpoint of HIV-1 RNA <50 copies/mL (intent-to-treat/time-to-loss of virologic response)
- DRV/r therapy was also associated with a lower incidence of common gastrointestinal (GI) toxicities and lipid abnormalities than LPV/r therapy.
· The current analysis assessed the pharmacokinetics of DRV and determined the effect of extrinsic and intrinsic factors on DRV pharmacokinetic (PK) parameters during 48 weeks of therapy in ARTEMIS.
Author Conclusions
This PK analysis showed that DRV C0h was consistently above the protein binding-corrected EC50 value for wild-type virus (55ng/mL) for all patients. Additionally, the median DRV C0h was 37-fold greater than this EC50 value.
Exposure to DRV was not influenced by age, body weight or hepatitis coinfection status. The effects of race and sex were not considered clinically relevant
-- PK results in this study confirm previous findings in treatment-experienced patients.1,2
Ongoing studies will further evaluate the potential impact of sex and race on DRV/r (GRACE) and compare once-daily versus twice-daily DRV/r in treatment-experienced patients with zero DRV resistance-associated mutations (TMC114-C229).
Results
Patient characteristics
· A total of 343 patients were randomized and treated in the DRV/r arm (Table 1). Women and non-Caucasians were well represented (30% and 60%, respectively).
· Consistent with the treatment-naïve status of patients, few patients (8%) in the DRV/r arm had CDC category C HIV infection. At baseline, 34% of patients in the DRV/r arm had 3100,000 copies/mL and median CD4 cell count was 228 cells/mm3.
Pharmacokinetics
Of the 343 patients randomized and treated with DRV/r, 335 patients with sparse sampling data were included in the population PK analysis for DRV.
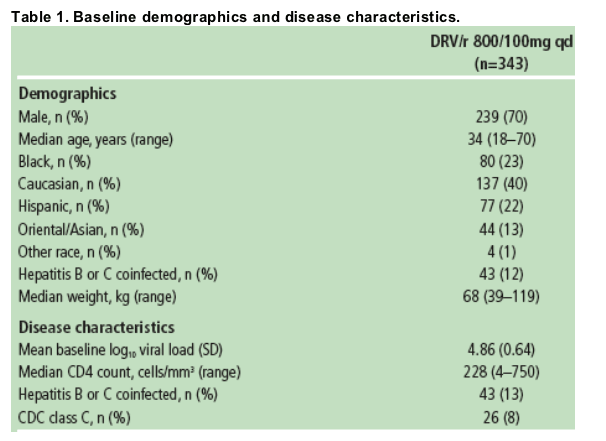
Overall median AUC24h was 87,854ng·h/mL (range 45,000-219,240ng·h/mL) and DRV C0h was 2041ng/mL (range 368-7242ng/mL).
For all DRV/r-treated patients, median DRV C0h was consistently above 55ng/mL, which is the protein binding-corrected EC50 value for wild-type virus.
The median DRV C0h was 37-fold greater than this EC50 value (Figure 1).
Subgroup analysis
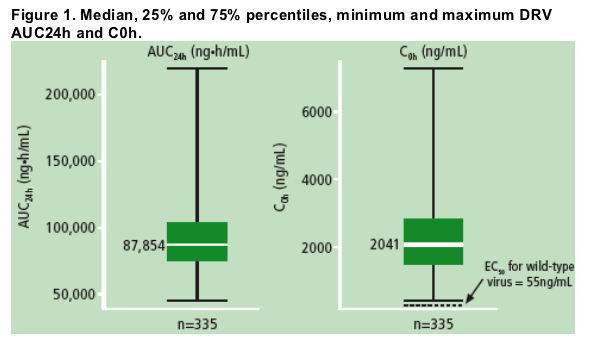
Graphical analysis by sex showed that female patients had a slightly higher exposure compared with male patients (Figure 2a) as was seen before.4
Comparison of mean AUC24h values showed a trend towards slightly lower exposure in Asian patients compared with the overall population (Figure 2b).
The differences in exposure relative to sex and race were not considered clinically relevant.
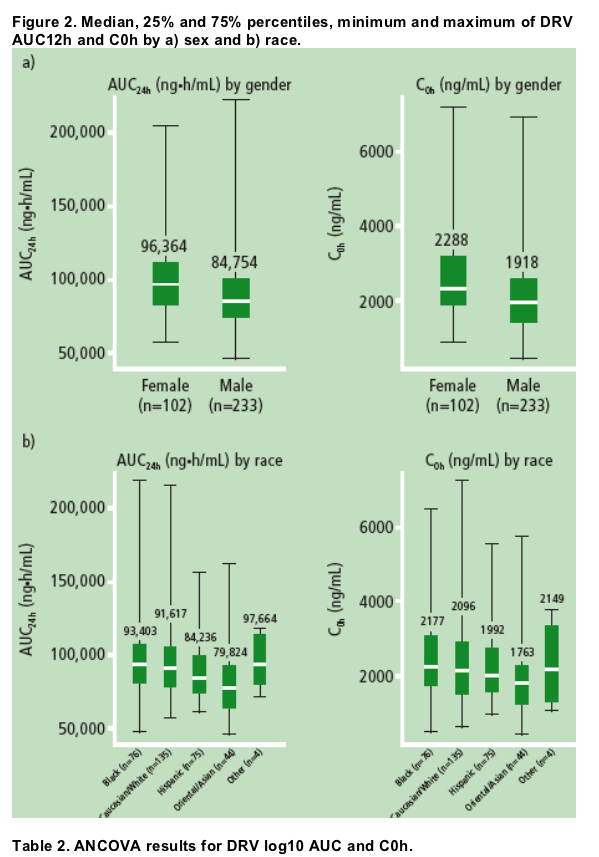
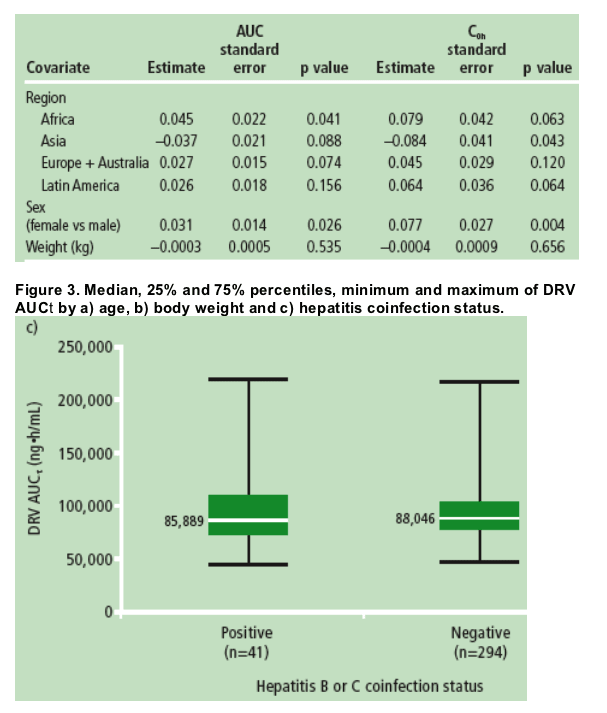
Age, body weight and hepatitis coinfection status did not influence exposure to DRV (Figure 3a-c).
These findings were supported by the ANCOVA results (Table 2) and were consistent with observations in the POWER and TITAN studies,5,6 where DRV/r was administered at 600/100mg bid.
Methods
Patients and study design
· ARTEMIS is a Phase III, randomized, controlled, open-label trial.
· In this study, treatment-naïve, HIV-1-infected, adult patients were randomized to receive DRV/r 800/100mg qd or LPV/r 800/200mg total daily dose, plus a standard daily dose of tenofovir disoproxil fumarate/emtricitabine.
· The study protocol was reviewed and approved by the appropriate institutional ethics committee(s) and health authorities, and the study was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all patients.
PK analysis
· Sparse sampling for the determination of plasma concentrations of DRV and ritonavir was performed in all patients
-- blood samples were drawn at Weeks 4, 8, 24, 48, 72 and 96, with two samples taken at Weeks 4 and 24 (the first sample immediately before DRV/r intake, and the second at least 1 hour after the first was drawn).
· Plasma concentrations of DRV and ritonavir were determined by a validated liquid chromatography-mass spectrometry/mass spectrometry method. The lower limit of quantification was 10.0ng/mL for DRV and 5.00ng/mL for ritonavir.
· Estimates of exposure (area under the curve [AUC24h]) and trough concentrations (C0h) of DRV were calculated
- a population PK model was developed for DRV on the basis of data in HIV-1- infected patients and healthy volunteers.4 The final population PK model to describe DRV pharmacokinetics was a two-compartmental model with first-order absorption, where apparent clearance is dependent on the AAG concentration and the dose. This model was then applied to the samples drawn in this trial to derive empirical Bayesian estimates of DRV exposure at steady-state.
· Descriptive statistics were calculated for the population PK parameters, overall and by subgroups for alpha-1 acid glycoprotein (AAG) levels at baseline, hepatitis coinfection status, sex, age, race, region and body weight.
· An analysis of covariance (ANCOVA) was used to assess the effect of region, sex and weight on DRV pharmacokinetics.
References
1. Clotet B, et al. Lancet 2007;369:1169-78.
2. Madruga JV, et al. Lancet 2007;370:49-58.
3. DeJesus E, et al. 47th Interscience Conference on Antimicrobial Agents and Chemotherapy, Chicago, IL, USA, 17-20 September 2007. Abstract H-718b.
4. Vis P, et al. 15th Meeting of the Population Approach Group in Europe, Bruges, Belgium, 14-16 June 2006. Abstract 964.
5. Sekar V, et al. 13th Conference on Retroviruses and Opportunistic Infections, Denver, CO, USA, 5-8 February 2006. Abstract J-121.
6. Sekar V, et al. 11th European AIDS Conference, Madrid, Spain, 24-27 October 2007. Abstract P4.1/10.
|
| |
|
 |
 |
|
|