 |
 |
 |
| |
Effect of Atazanavir with and without Ritonavir on the Pharmacokinetics of the CYP2C8 Probe Rosiglitazone in Healthy Subjects
|
| |
| |
Reported by Jules Levin
9th International Workshop on Clinical Pharmacology of HIV Therapy
April 7-9, 2008
New Orleans
H. Sevinsky, T. Eley, C. Yones, A. Persson, T. Li, X. Xu, L. Mahnke, J. Zhang and R. Bertz
Bristol-Myers Squibb Research and Development, Princeton, NJ
AUTHOR CONCLUSIONS
ATV is a weak inhibitor of CYP2C8
- Exposures of CYP2C8 substrates may be modestly increased in the presence of ATV without RTV
RTV induction offsets CYP2C8 inhibition by ATV
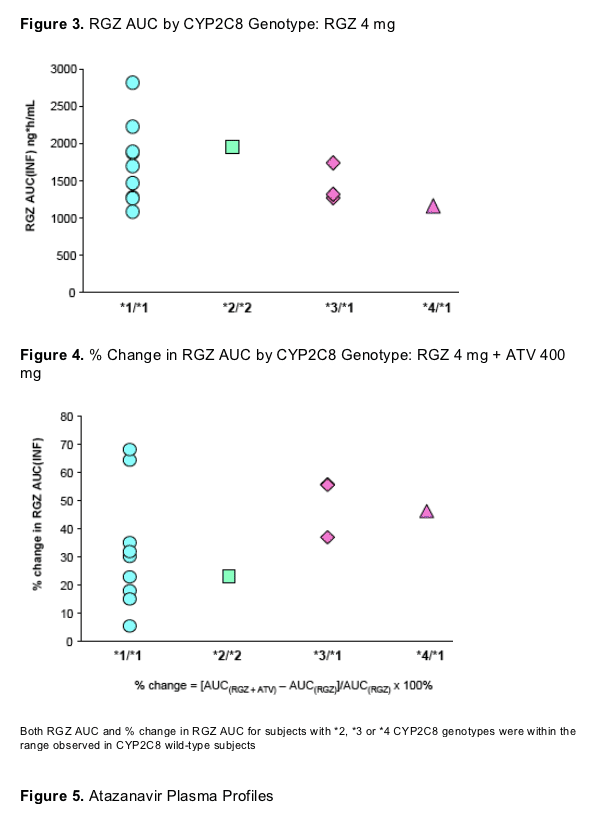
Although data in subjects with rare alleles are limited, there does not appear to be an in vivo relationship between CYP2C8 genotype and RGZ exposure or magnitude of ATV effect
BACKGROUND
Atazanavir (ATV) is a once-daily protease inhibitor indicated for the treatment of HIV infection as part of combination therapy
- Dosing regimens include ATV 400 QD in treatment-naïve patients and ATV/RTV 300/100 QD in treatment-experienced patients
- ATV is a known inhibitor of CYP3A4 and UGT1A1 in vivo
- ATV directly inhibits CYP2C8 in vitro (Ki #&126; 1.7 _g/mL)
ATV concentrations following ATV/RTV 300/100 mg
#&183; average: #&126;0.4 _g/mL (free), #&126;2.4 _g/mL (total)
#&183; Cmax: #&126;0.9 _g/mL (free), #&126;6.1 _g/mL (total)
- RTV has been shown to be both an inhibitor and an inducer of CYP2C8 in vitro and is an inducer of CYP2C9 in vivo1,2,3
- RTV increases ATV exposure, therefore the inhibitory effect of ATV on CYP2C8 could be enhanced in the presence of RTV
Rosiglitazone (RGZ) was chosen as a CYP2C8 probe substrate
- Bioavailability is 99%
-- Metabolized by CYP2C8 (major) and CYP2C9 (minor)
CYP2C8 Genotypes
CYP2C8 is polymorphic
- CYP2C8*1 is wild-type

In vitro studies indicate that CYP2C8*2 and CYP2C8*3 genotypes have reduced
CYP2C8 activity5,6,7
- CYP2C8*2: decline of #&126;50%
- CYP2C8*3: decline of #&126;85%
The relationship between in vivo CYP2C8 activity and genotype is not well characterized
- CYP2C8*1/*3 carriers demonstrated a mean repaglinide AUC that was 45% less than CYP2C8*1/*1 carriers8
- CYP2C8*3/*3 carriers had mean RGZ AUC 36% less than CYP2C8*1/*1
#&183; Mean RGZ AUC in CYP2C8*1/*3 was not significantly different from CYP2C8*1/*19
OBJECTIVES
Primary:
- To assess the effect of ATV 400 mg QD and ATV/RTV 300/100 mg QD at steady state on the single dose PK of the CYP2C8 probe RGZ in healthy subjects
Secondary:
- To explore the relationship between ATV exposure, CYP2C8 genotype and RGZ PK
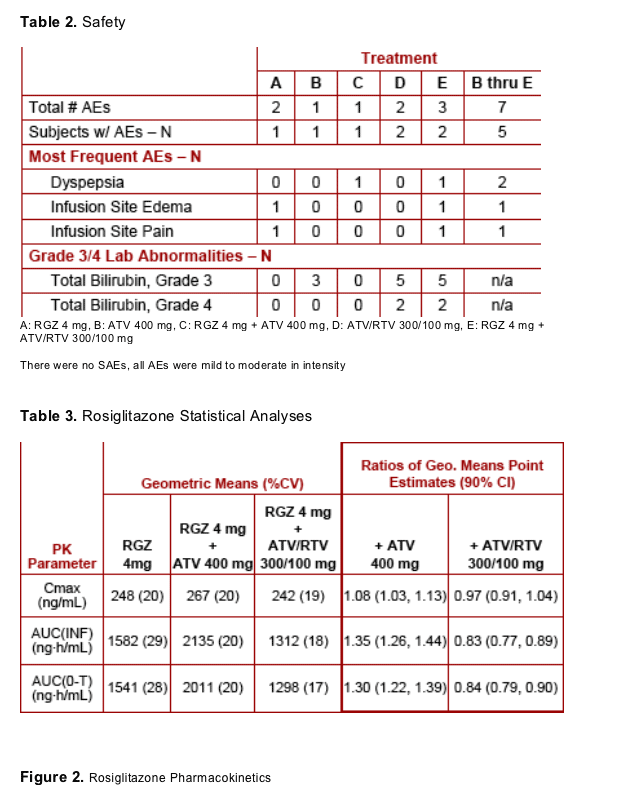
- To assess the safety and tolerability of RGZ and ATV coadministration with and without RTV
METHODS
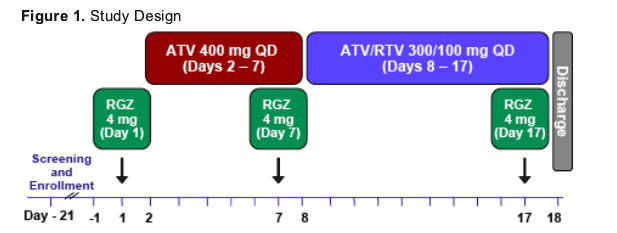
- Open-label, single-sequence crossover study in 14 healthy subjects
- Study drugs administered in the morning with a light meal
- PK analyses on Days 1, 7 and 17 for RGZ
- Sparse PK sampling on Days 7 and 17 for ATV and Day 17 for RTV
Bioanalytical Methods: LC-MS/MS
- RGZ LLOQ = 1 ng/mL, ATV LLOQ = 10 ng/mL, RTV LLOQ = 5 ng/mL
- QC deviations within +/- 8.67% for all analytes
Noncompartmental PK analyses
Statistical Analysis
- Estimates were generated using general linear models fitted to log-transformed data, with treatment as a fixed effect and measurements within each subject as repeated measurements
- Point estimates and 90% CIs for the ratios of geometric means for RGZ Cmax, AUC(0-t) and AUC(INF)
- Day 1 RGZ used as reference in all comparisons
-- The relationship between CYP2C8 genotype and ATV exposures as it relates to RGZ PK was explored using scatter plots
RESULTS
Demographics
14 subjects completed study, 12 men (86%), 2 women (14%)
- Mean age (range) = 37 years (23 - 47 years)
- 4 African American subjects (29%), 10 Caucasian subjects (71%)
#&183; CYP2C8*1/*1, N = 9
#&183; CYP2C8*2/*2, N = 1
#&183; CYP2C8*3/*1, N = 3
#&183; CYP2C8*4/*1, N = 1
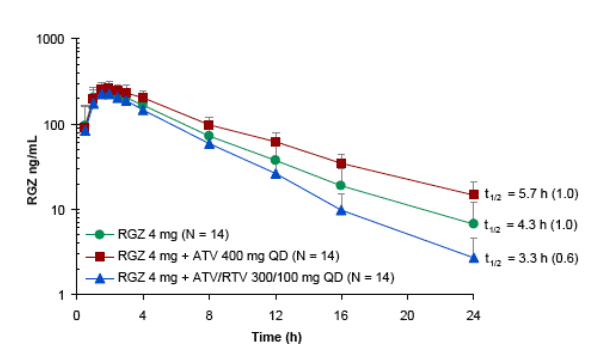
Due to high bioavailability, RGZ Cmax was not affected by the coadministration of ATV or ATV/RTV
Differential effects in AUC observed:
- RGZ exposures increased when coadministered with ATV 400 mg
-- RGZ exposures decreased when coadministered with ATV/RTV 300/100 mg
Inhibitor classification (per FDA):
- strong: ≥ 5-fold increased AUC, moderate: ≥ 2-fold but < 5-fold increased AUC, weak: ≥ 1.25-fold but < 2-fold increased AUC
ATV appears to be a weak CYP2C8 inhibitor in vivo
RTV appears to be a CYP2C8 inducer, strong enough to overcome inhibition by ATV
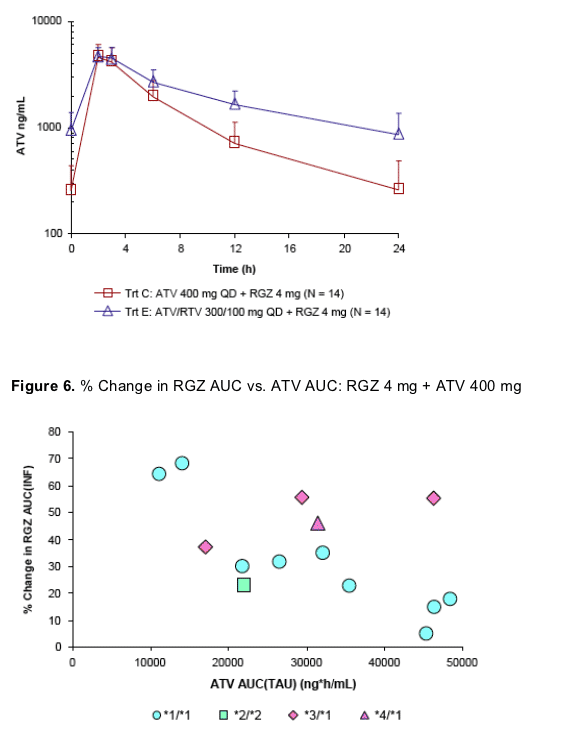
ATV (± RTV) exposures in the presence of RGZ were consistent with historical ATV (± RTV) data when administered alone
There does not appear to be a clear relationship between ATV AUC and increase in RGZ AUC based on these limited data, despite concentration-dependent inhibition of CYP2C8 observed in vitro
The relationship between RGZ AUC and ATV AUC when RGZ is coadministered with ATV/RTV is confounded by the addition of RTV and it apparent effects on
CYP metabolism
REFERENCES
1 Walsky RL. J Clin Pharmacol 2005: 45; 68-78.
2 Norvir US package insert, Abbot Laboratories 2006.
3 Dixit V. Drug Metab Disp 2007: 35(10); 1853-1859.
4 Totah RA. Clin Pharm Ther, 2005: 77(5); 341-352.
5 Dai D. Pharmacogen 2001: 11: 597-607.
6 Soyama A. Biol Pharm Bull 2001; 24: 1427-1430.
7 Soyama A. Pharmacol Toxicol 2002; 91: 174-178.
8 Niemi M. Clin Pharm Ther 2003; 74(4): 380-387.
9 Kirchheiner J. Clin Pharm Ther 2006; 80(6): 657-667.
|
| |
|
 |
 |
|
|