 |
 |
 |
| |
Single-Dose Pharmacokinetics of Enteric-Coated Didanosine in Subjects with Hepatic Impairment Compared to Healthy Adult Subjects
|
| |
| |
Reported by Jules Levin
9th International Workshop on Clinical Pharmacology of HIV Therapy
April 7-9, 2008
New Orleans
C. Chien, A. Persson, H. Sevinsky, J. Dudley, M. Lu, S. Kaul, and R. Nettles
Bristol-Myers Squibb Research and Development, Princeton, NJ
AUTHOR CONCLUSIONS
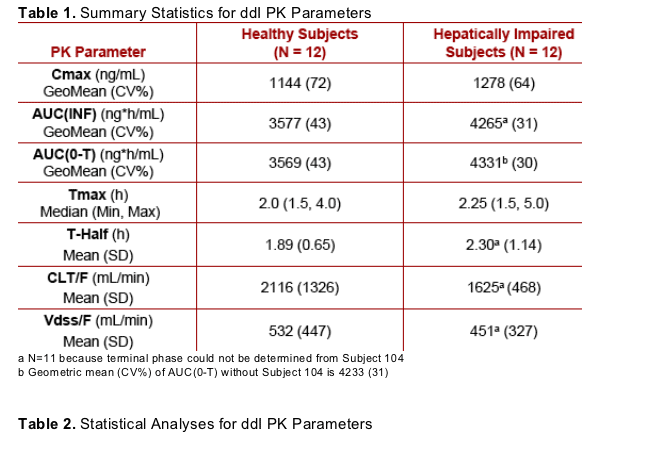
After administration of ddI-EC 400 mg, increases in Cmax, AUC(INF) and AUC(0-T) in subjects with Child-Pugh Class B or C hepatic impairment of 13%, 19% and 21%, respectively, relative to healthy control subjects were observed
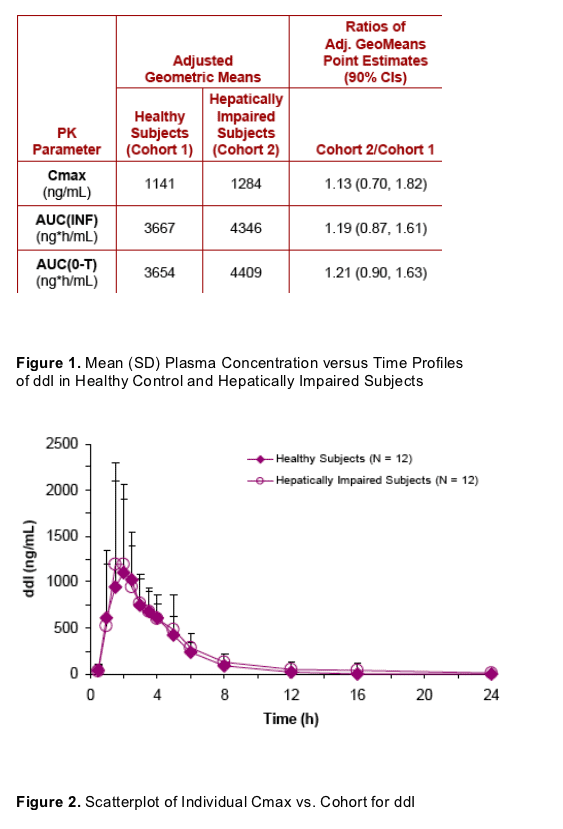
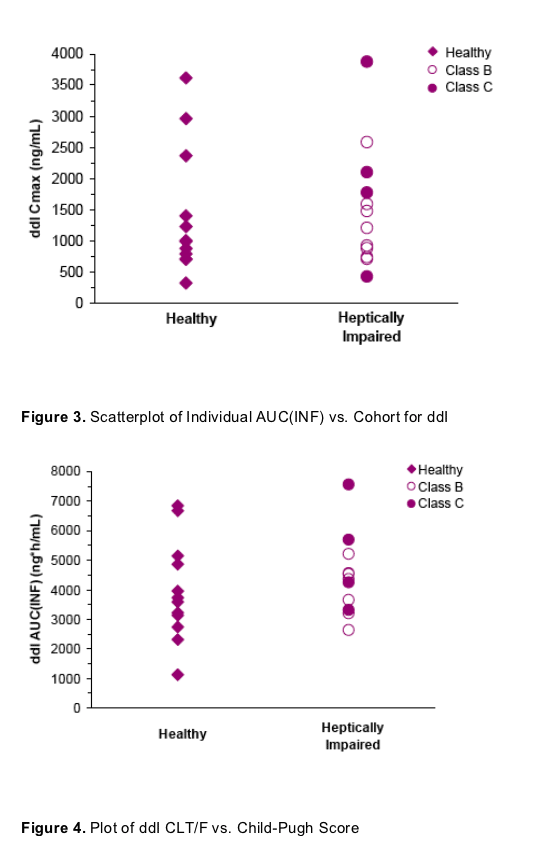
Scatterplots of individual ddI PK parameter versus cohort illustrate the overlap between healthy and hepatically impaired subjects
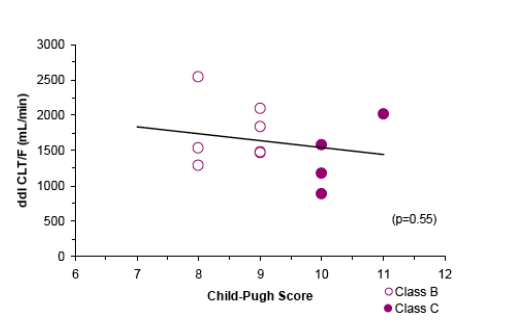
Based on exploratory analyses of these limited data, CLT/F of ddI did not demonstrate any dependence on Child-Pugh scores
The slightly elevated ddI exposures observed in hepatically impaired subjects are likely not of clinical relevance
BACKGROUND
Liver impairment is of increasing concern in HIV-infected patients:1, 2
- With normal liver function prior to HAART
- With chronic liver disease treated with HAART
- With HBV/HCV co-infection
Didanosine (ddI) is a Nucleoside Reverse Transcriptase Inhibitor (NRTI) that is a common component of HAART in the treatment of HIV3
HAART administration to hepatically impaired patients is increasingly common whereas pharmacokinetic data in such subjects are often unavailable1
OBJECTIVES
Primary:
- To compare the pharmacokinetics (PK) of a single 400 mg oral dose of ddI entericcoated beads in capsules (ddI-EC) in subjects with hepatic impairment and in healthy control subjects
Secondary:
- To asses the safety and tolerability of a single oral dose of ddI-EC in subjects with hepatic impairment or in healthy subjects
-- To explore the relationship between Child-Pugh classification and clearance (CLT/F) of ddI
METHODS
Single dose, non-randomized, open-label study with 25 HIV-negative subjects
- 12 subjects with hepatic impairment
· Child-Pugh Class B = 8
· Child-Pugh Class C = 4
- 13 healthy matching controls
- Subjects were matched (1:1) with regard to
· Age (+/- 5 years)
· Weight (+/- 15%)
· Gender
· Smoking status
» Light, < 10 cigarettes/day
» Moderate, 10 - 20 cigarettes/day
» Heavy, > 1 pack/day
- Each subject received a single 400 mg oral dose of ddI-EC in the fasted state
- Serial blood samples were collected up to 24 h post dose
Bioanalytical Methods
- LC-API/MS/MS
- LLOQ = 0.5 ng/mL for ddI
- QC deviations within +/- 10% for ddI
PK Analysis
- PK parameters were calculated by non-compartmental analysis using Kinetica
Safety and Tolerability
- Assessed by adverse event reports, vital signs, ECGs, physical examinations, and clinical labs
Statistical Analysis
- Analyses of variance on log AUC(INF) and log Cmax
- Point estimates and 90% CIs for the ratios of geometric means
-- The relationship between Child-Pugh Score and CLT/F of ddI in hepatically impaired subjects was evaluated graphically and by linear regression
RESULTS
Demographics
25 subjects completed study: 13 healthy, 12 hepatically impaired
- Gender (%):
· Healthy: 9 males (69%), 4 females (31%)
· Hepatically Impaired: 8 males (67%), 4 females (33%)
- Mean Age (range):
· Healthy: 53 (45 - 69 years)
· Hepatically Impaired: 54 (46 - 66 years)
- Race (%):
· Healthy: 11 White (85%), 2 African American (15%)
· Hepatically Impaired: 11 White (92%), 1 African American (8%)
One healthy subject did not satisfy the protocol-defined conditions for matching and was replaced and excluded from PK analysis
Pharmacokinetics
Safety Summary
There were no deaths, serious adverse events or discontinuations due to adverse events (AEs)
6 total AEs, none occurred more than once:
- 4 AEs in hepatically impaired [gastritis, nausea, vomiting (19 h post-dose), anorexia]; all mild and deemed possible or probably related to study drug
- 2 AEs in healthy (fatigue, headache); all moderate and deemed not related to study drug
Marked laboratory abnormalities were reported in 75% of hepatically impaired subjects
- Majority were consistent with the degree of hepatic impairment in Child-Pugh Class B and C hepatic disease
Single oral doses of ddI-EC (400 mg) were generally safe and well-tolerated in healthy and hepatically impaired subjects
REFERENCES
1. Dieterich DT. Human immunodeficiency virus and the liver: lessons learned and still to be learned. Semin Liver Dis. 2003 May;23(2):107-14.
2. Benhamou Y, et al. Liver fibrosis progression in human immunodeficiency virus and hepatitis C virus coinfected patients. Multivirc Group Hepatol 1999;30(4):1054-8.
3. Videx EC Package Insert, Bristol-Myers Squibb Company, Princeton, NJ, 2007.
|
| |
|
 |
 |
|
|