 |
 |
 |
| |
Black Race and Drug Interactions Affect Nonnucleoside Levels in TDM Analysis
|
| |
| |
9th International Workshop on Clinical Pharmacology of HIV Therapy
April 7-9, 2008
New Orleans
Mark Mascolini
Black race independently raised efavirenz or nevirapine concentrations in people with drug levels determined by the British therapeutic drug monitoring (TDM) service in Liverpool [1]. Protease inhibitors (PIs) and tenofovir boosted nevirapine levels, while zidovudine and higher weight lowered efavirenz levels. As anticipated, the anti-TB drug rifampin lowered concentrations of both nonnucleosides.
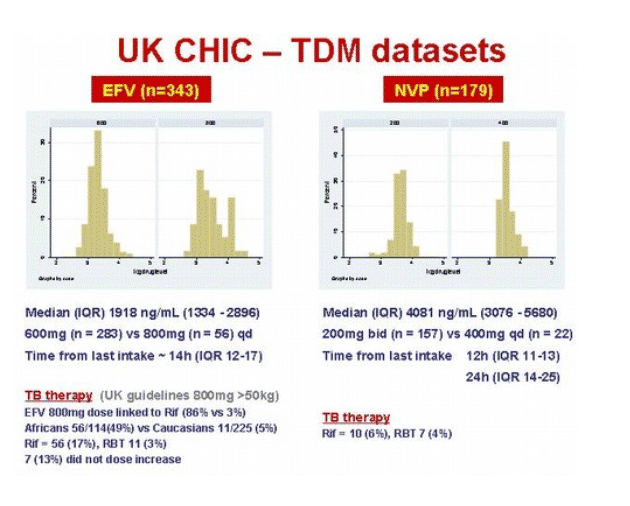
Saye Khoo and Liverpool TDM colleagues scrutinized nonnucleoside levels in 339 people taking efavirenz and 179 taking nevirapine, all of whom had samples sent for drug-level analysis. Among those taking efavirenz, 114 (34%) were black, while 48 nevirapine users (27%) were black. People taking nevirapine had a longer nonnucleoside treatment history than those taking efavirenz (median 91 versus 42 weeks) and a longer time on their current regimen (median 40 versus 23 weeks).
In the nevirapine group 22 people (12%) were taking 400 mg once daily, and 56 people (16.5%) were taking 800 mg of efavirenz daily instead of 600 mg. (UK guidelines call for 800 mg of efavirenz in people weighing more than 50 kg.) In the efavirenz group 56 people (17%) were taking rifampin, while 10 (6%) in the nevirapine group were taking that anti-TB drug.
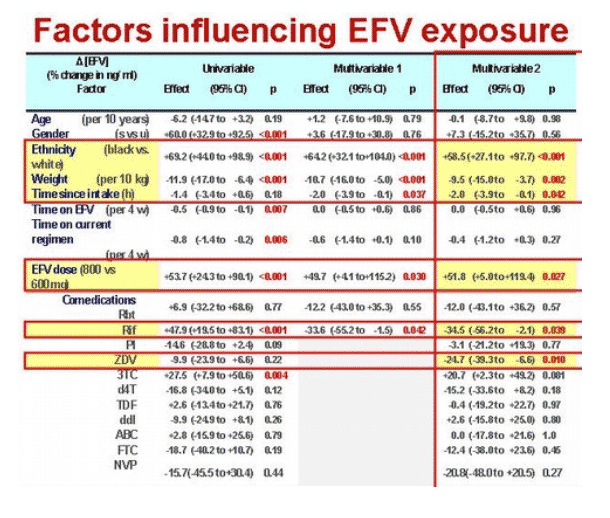
Median efavirenz concentration measured approximately 14 hours after dosing stood at 1918 ng/mL (interquartile range [IQR] 1134 to 2896 ng/mL). Multivariate analysis sorted out six factors that affected efavirenz levels (calculated as the percent change in ng/mL):
· Black versus white race: +58.5% (95% confidence interval [CI] +27.1 to +97.7, P < 0.001)
· Every extra 10 kg of weight: -9.5% (95% CI -15.0 to -3.7, P = 0.002)
· Every extra hour between dosing and drug measurement: -2.0% (95% CI -3.9 to -0.1, P = 0.042)
· 800 versus 600 mg of efavirenz: +51.8% (95% CI +5.0 to +119.4, P = 0.027)
· Rifampin: -34.5% (95% CI -56.2 to -2.1, P = 0.039)
· Zidovudine: -24.7% (95% CI -39.3 to -6.6, P = 0.010)
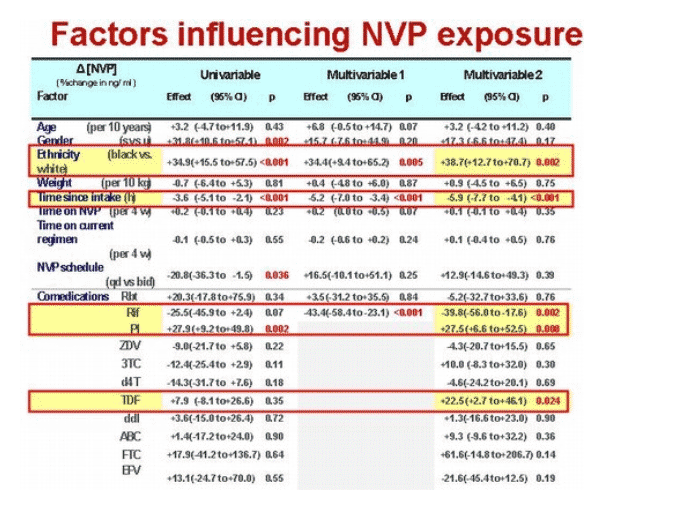
Median nevirapine levels reckoned 12 or 24 hours after dosing measured 4081 ng/mL (IQR 3076 to 5680). Five factors independently influenced nevirapine concentrations:
· Black versus white race: +38.7% (95% CI +12.7 to +70.7, P = 0.002)
· Every extra hour between dosing and drug measurement: -5.9% (95% CI -7.7 to -4.1, P < 0.001)
· Rifampin: -39.8% (95% CI -56.0 to -17.6, P = 0.002)
· PI cotreatment: +27.5% (95% CI 6.6 to 52.5, P = 0.008)
· Tenofovir: +22.5% (95% CI +2.7 to +46.1, P = 0.024)
Khoo noted that the correlation between black race and higher efavirenz and nevirapine concentrations is consistent with results of the 2NN study [2] and with findings that shifts in the gene that encode the CYP 2B6 metabolizing enzyme raise nonnucleoside levels. The nevirapine correlations with weight, rifampin, PIs, and tenofovir also reflect consistent earlier reports. Prior research yielded conflicting results on whether weight lowers efavirenz levels. Khoo could find no earlier work linking zidovudine with lower efavirenz concentrations, and he said "no obvious mechanism" explains that correlation.
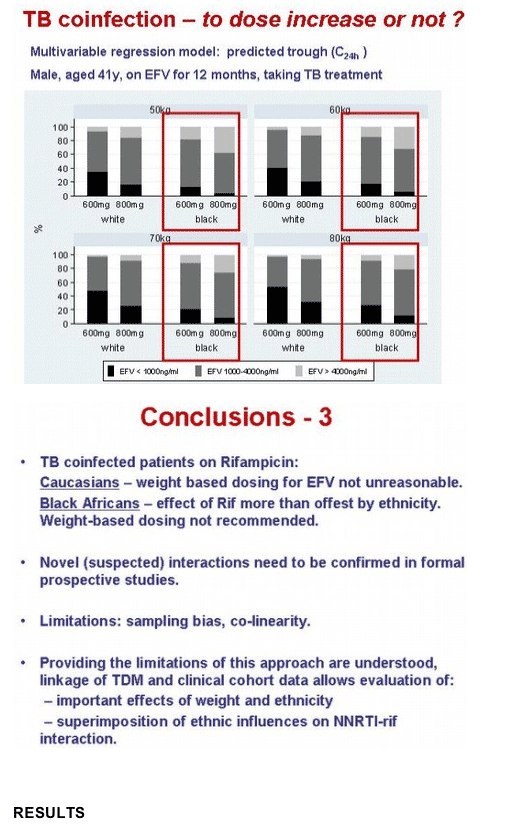
The Liverpool team proposed that weight-based dosing for whites taking rifampin with a nonnucleoside is "not unreasonable." For blacks, though, the impact of race on nonnucleoside levels apparently offsets the impact of rifampin. Khoo cautioned that drug-level correlations derived from a TDM database must be interpreted with caution because of several possible limitations: selection bias (samples are sent for drug-level testing when something is going wrong), inaccurate dose timing, lack of clinical data or inaccurate clinical data, unreported cotreatment with herbal remedies, and lack of adherence data.
References
1. Stoehr W, Back D, Dunn D, et al. Factors influencing efavirenz and nevirapine plasma concentration: Effect of ethnicity, weight, and co-medication. 9th International Workshop on Clinical Pharmacology of HIV Therapy. April 7-9, 2008. New Orleans. Abstract O13.
2. Kappelhoff BS, van Leth F, MacGregor TR, et al. Nevirapine and efavirenz pharmacokinetics and covariate analysis in the 2NN study. Antivir Ther. 2005;10:145-155.


|
| |
|
 |
 |
|
|