 |
 |
 |
| |
Hexadecyloxpropyl Tenofovir (CMX157) Has Enhanced Potency In Vitro Against NRTI Resistant HIV Relative to Tenofovir and a Favorable Preclinical Profile
|
| |
| |
Reported by Jules Levin
17th Intl HIV Drug Resistance Wkp
June 10-14, 2008
Sitges, Spain
ER Lanier B Lampert, Trost, G Painter and M Almond, Chimerix Inc, Research Triangle Park, NC, USA
AUTHOR CONCLUSIONS/DISCUSSION
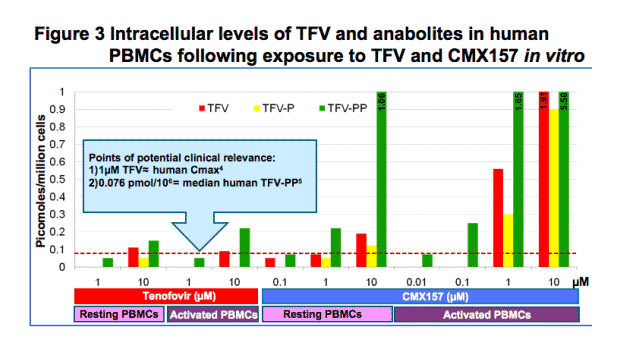
CMX157 is designed to improve bioavailability and cellular penetration of tenofovir while decreasing peripheral exposure to TFV per se. The greatly diminished IC50s for CMX157 versus TFV suggested that higher intracellular levels of the active anabolite, TFV-PP, were being made following exposure to similar molar equivalents. We tested this in human PBMCs using dilutions of both drugs which bracketed the Cmax of TFV in humans following standard dosing of TDF4. The TFV Cmax is approximately 1 μM (1167 nM) following 300 mg dosing of TDF. PHA/IL-2 activated human PBMCs exposed to 10 nM CMX157 contained more TFV-PP (70 fmol/106) than those exposed to 1000 nM TFV (50 fmol/106). Notably, these levels were similar to those seen in patients taking TDF (median 76 fmol/106). These data in combination with a favorable toxicology profile support further development of CMX157 for HIV.
1) CMX157 is >300 fold more potent than TFV in vitro against wt HIV and against clinically relevant HIV mutants
2) The amount of active drug produced in human PBMCs is much higher following equimolar exposure to CMX157 vs TFV
3) Toxicology and TK data reveal high exposure to CMX157 in plasma with minimal toxicity (rodent and non-rodent)
INTRODUCTION
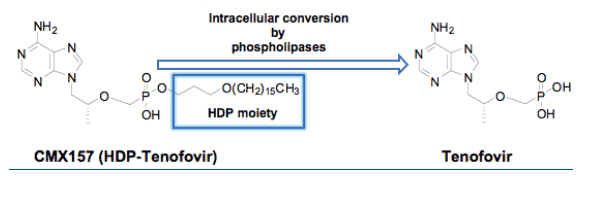
CMX157 is a lipid (1-0-hexadecyloxypropyl, HDP) conjugate of the acyclic nucleotide analogue tenofovir. Tenofovir disoproxil fumarate (TDF), a prodrug of tenofovir (TFV) is one of the most widely used nucleoside/tide reverse transcriptase inhibitors (NRTIs). However, it loses activity against specific HIV mutants, including those with K65R, multiple thymidine analog mutations (TAMs) or multi-NRTI resistant (MNR) mutations and has been associated with nephrotoxicity. Our goal is to increase efficacy and decrease toxicity of TFV. The approach we have taken is to create a prodrug that is more stable in plasma, but effectively penetrates cells and yields high intracellular levels of the active anabolite, TFV-diphosphate (TFV-PP). Unlike tenofovir-DF and most prodrugs, CMX157 is not efficiently cleaved to free tenofovir in vivo. This should both increase the levels of active TFV-PP in target cells for HIV and decrease the levels of free TFV in blood, potentially lowering the apparent IC50 and reducing the rate of secretion into the kidney.
Chimerix's lipid conjugate technology employs covalent coupling of a synthetic lipid to drugs, in this case via a phosphonate moiety2. The goal of lipid conjugation is to increase oral bioavailability, increase potency and target delivery to specific tissues. The novel molecules are designed to resemble natural lipids that are readily absorbed from the small intestine and distributed to tissues via lymph and/or plasma. Hydrophobic lipid conjugates may be incorporated into chylomicrons released into the lymphatic system and ultimately venous blood while hydrophilic conjugates may be absorbed and pass directly into portal blood. Once in the cell the drug is cleaved from the lipid carrier by hydrolytic action of phospholipases on the phosphorous-O-alkyl bond3.
METHODS
CMX157 IC50s were determined for a panel of 30 NRTI isolates with major NRTI mutations, including K65R +/- M184V, multiple TAM combinations +/-M184V, K70E in various combinations and MNR complexes including T69SXX and Q151M (PhenoSense). A separate panel of 14 NRTI resistant clinical isolates and wild-type isolates from subtypes A-G, O and HIV-2 were examined in PBMCs. Additional studies determined activity in monocytic cells, cytotoxicity in dividing and non-dividing cells, tenofovir-diphosphate levels in PBMCs, serum effects ,and toxicity/toxicokinetics in rats and monkeys. For tenofovir anabolite analysis, PBMC samples were lysed with 70% ice cold methanol and centrifuged; supernatants were analyzed in triplicate using LC/MS/MS. TFV ,TFV mono- and diphosphate were separated by gradient, reverse phase, ion-paring chromatography and detected by positive ion electrospray. RSDs between 2.4 and 14.2% were obtained for data shown.
RESULTS
Activity against Wild-Type HIV:
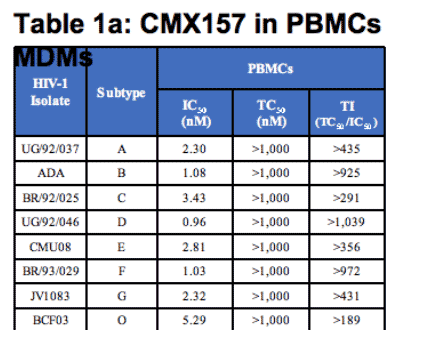
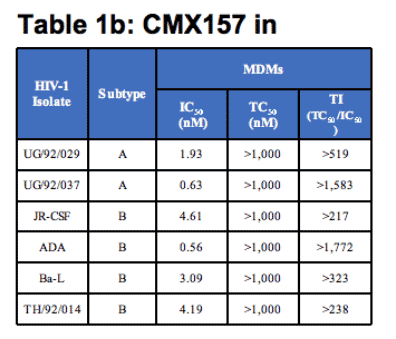
The in vitro antiviral activity profile for CMX157 was evaluated extensively for cell-type effects and HIV strain effects. As shown below (Table 1a), it is active against all major subtypes of HIV-1 in PBMCs with IC50 values ranging between 0.20 and 7.18 nanomolar (nM). Five isolates were tested for subtype B and three isolates were tested for all other subtypes; values shown are for the median isolate in each group. IC50s against three isolates of HIV-2 in PBMCs were <5 nM. CMX157 was also active against HIV in monocyte derived macrophages (MDMs) with IC50s between 0.56 and 4.61 nM as shown below in Table 1b. By comparison, TFV IC50s against HIV-1 subtypes A-G, and O ranged from 1600-4900 nM4.
Effect of human serum on in vitro efficacy:
Extrapolating from the IC90 values obtained in 0% to 40% human serum results in an estimated IC90 value of 144 nM for 100% human serum (assuming a linear relationship). Similarly, extrapolating from IC50 values results in an estimated IC50 value of 52.5 nM for 100% human serum.
In Vitro Cytotoxocity:
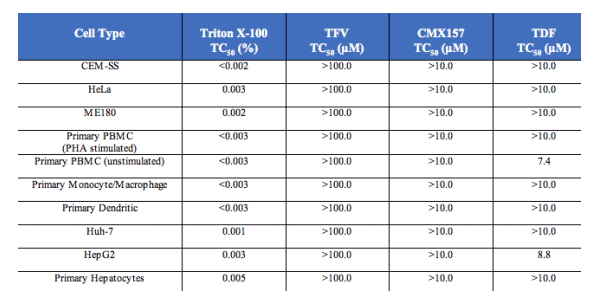
Cellular cytoxicity was evaluated in vitro using dividing and non-dividing cells relevant for HIV antivirals and multiple other human cell types. No toxicity was observed. Evaluation of CMX157 in bone marrow progenitor cells in colony formation assays is in progress.
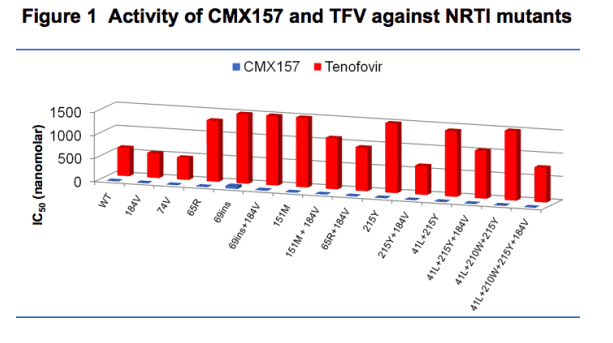
Activity of CMX157 and TFV against NRTI resistant HIV:
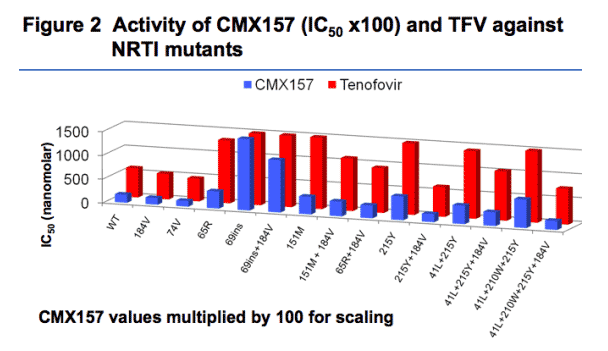
IC50s for CMX157 ranged from 0.66 nM for L74V/M184V to 57 nM for A62V/T69SVG/V75I/215I in the PhenoSense assay; corresponding IC50s for tenofovir were 227 nM and 16,959 nM. CMX157 IC50s for M41L/L210W/ T215Y averaged 6.3 nM without M184V and 2.2 nM with M184V (2,240 and 770 nM for tenofovir respectively). Similar data were obtained in PBMCs. Data for key mutants with actual IC50s are presented in Figure 1. To visualize comparisons, CMX157 IC50s were multiplied 100 times and presented against actual TFV IC50s in Figure 2.
Toxicology: No toxicity was observed in rats administered CMX157 up to 100 mg/kg/d for 7 days; the CMX157 Cmax was 686 ng/ml and the TFV Cmax was 322 ng/ml on day 7. GeneTox studies were negative (no mutagenicity/clastogenicity). There were no important effects in Safety Pharmacology studies at doses of 40 and 200 mg/kg. Twenty-eight day Tox/TK Studies in rats and monkeys showed doses of 50 and 200 mg/kg/day were well tolerated with good systemic exposure. The in-life portion of these studies is complete, but pathology and reports are pending. The expected NOAEL is 50 or 200 mg/kg/day.
Levels of active anabolite in human PBMCs:
Following 24 hour incubations with equimolar TFV or CMX157, human PBMCs exposed to CMX157 had higher intracellular levels of active drug (TFV-PP). For example, 10nM CMX157 yielded higher levels of TFV-PP than 1000nM TFV.
References
1. G.R. Painter, L.C. Trost, et al. (2008) Drugs of the Future. submitted
2. G.R. Painter and K.Y. Hostetler (2004) Trends in Biotechnology 22:423-427.
3. N. Gummad and A.K. Menon (2002) J. Biol. Chem. 277:25337-25343.
4. Viread package insert (2007)
5. J.J. Kiser, C.L. Aquilante, et al. (2008) JAIDS 47(3): 298-302
|
| |
|
 |
 |
|
|