 |
 |
 |
| |
Response to Vicriviroc in HIV-Infected Treatment-Experienced Subjects using an Enhanced Version of the Trofile HIV Co-receptor Tropism Assay:
Reanalysis of ACTG5211 Results. Using Enhanced Tropism Assay Improves Outcomes.
|
| |
| |
Reported by Jules Levin
17th Intl HIV Drug Resistance Workshop
June 10-14, 2008
Sitges, Spain
From Jules: the Enhanced tropism Trofile assay reclassified 25 of 118 patients as D/M who were previously classified as CCR5 tropism. The enhanced assay is able to better identify tropism for patients compared to the standard tropism Trofile assay. To demonstrate the benefit of using the enhanced assay, this study also found greater viral load responses to vicriviroc for patients classified CCR5 than for those D/M using the enhanced assay.
Zhaohui Su1, Jacqueline Reeves2*, Amy Krambrink1, Eoin Coakley2, Michael Hughes1, Charles Flexner3, Timothy Wilkin4, Paul Skolnik5, Wayne Greaves6, Daniel Kuritzkes7, Roy Gulick4, and the ACTG 5211 Team
1Harvard School of Public Health, Boston, USA, 2Monogram Biosciences, South San Francisco, USA, 3Johns Hopkins University, Baltimore, USA, 4Weill-Cornell Medical College, New York, USA, 5Boston University Medical Center, Boston, USA, 6Schering-Plough Research Institute, Kenilworth, USA, 7Harvard Medical School, Boston, USA
AUTHOR CONCLUSION
· Screen tropism results from the enhanced Trofile assay predict early emergence of CXCR4-use in ACTG 5211 VCV recipients
· With increased sensitivity for detection of CXCR4-using minority variants, enhanced Trofile has improved ability to predict antiretroviral responses to VCV compared to the standard Trofile assay
· Enhanced Trofile is an improved screening tool for determining patient eligibility for CCR5 antagonist therapy.
BACKGROUND
· The TrofileTM HIV co-receptor tropism assay (Monogram Biosciences) is useful for the selection of patients for CCR5 antagonist therapy and determines if a patients viral population is CCR5- (R5), CXCR4- (X4) or dual/mixed- (D/M) tropic (1).
· CCR5 antagonists have shown efficacy in suppressing R5 but not X4 or D/M HIV. The positive and negative predictive value of Trofile assay tropism classifications has been demonstrated in clinical trials of CCR5 antagonists, including maraviroc and vicriviroc (VCV).
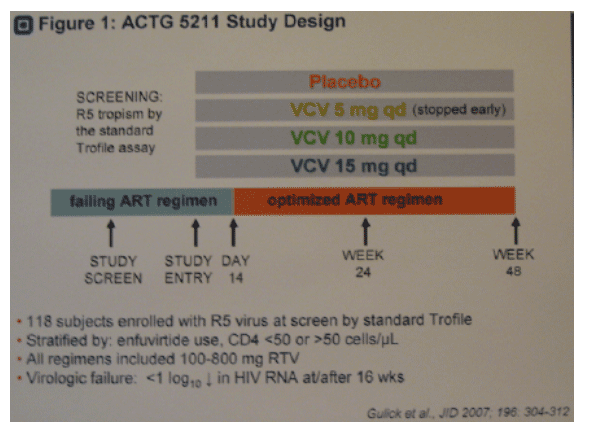
· In ACTG 5211, a placebo controlled dose ranging (5, 10, 15 mg) phase 2b study of VCV in 118 treatment-experienced subjects with R5 virus at study screen by standard Trofile (2, 3) (Figure 1), VCV demonstrated potent virologic suppression in subjects with R5 virus at study screen and entry (1.83 log reduction at week 24, n=71). A reduced virologic response (p=0.007) was observed among 10 VCV recipients with R5 virus at study screen but D/M at entry (0.77 log reduction at week 24) by standard Trofile.
· Low levels of CXCR4-using variants below the detection limit of the standard Trofile assay (5-10% (1)) can sometimes be identified by clonal analysis of Env populations and may be selected following CCR5 antagonist therapy.
· An enhanced sensitivity Trofile assay has been developed that allows improved detection of minor CXCR4-using variants in env clone mixtures (100% sensitivity at detecting X4 Envs as low as 0.3% (4)) and earlier detection of minor CXCR4-using subpopulations in longitudinal samples from PR/RT inhibitor experienced patients (5,6,7).
· We hypothesized that enhanced Trofile might better identify low level CXCR4-using virus in the screening samples from subjects enrolled into ACTG 5211 and
further optimize selection of patients who may benefit from CCR5 antagonists.
METHODS
· Enhanced Trofile was used to determine HIV co-receptor usage at study screen and entry for subjects enrolled in ACTG 5211. Virologic and immunologic responses were examined according to tropism results by enhanced Trofile and treatment arm and compared to responses from previous analyses. All analyses were intent-to-treat.
RESULTS
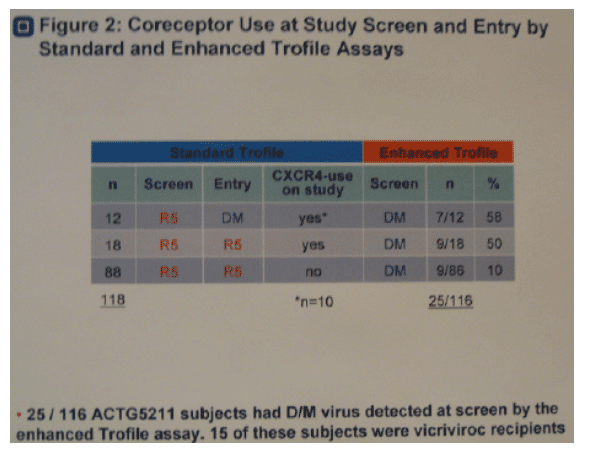
· Figure 2. Of 12 subjects with R5 virus at screen and DM at study entry by the standard Trofile assay, enhanced Trofile reclassified 7 subjects (58%) with DM virus at screen. Of 18 additional subjects with CXCR4-using virus detected on study by standard Trofile, enhanced Trofile reclassified 9 subjects (50%) with DM virus at screen. Of 86/88 subjects tested with R5 virus only at all time points by standard Trofile, enhanced Trofile reclassified 9 subjects (10%) with DM virus at screen.
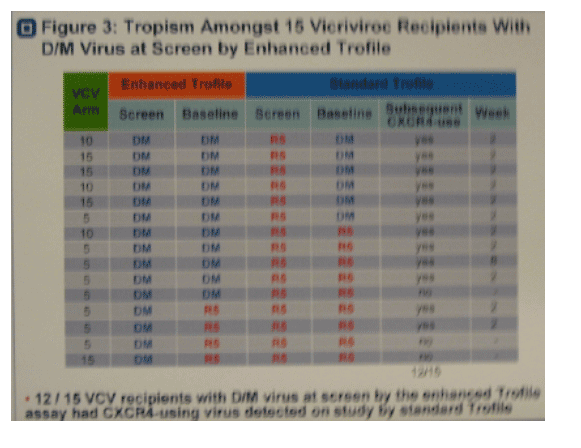
· Figure 3. 15/25 subjects with DM virus at screen by enhanced Trofile received VCV on study. 12/15 had early emergence of CXCR4-using variants detected by standard Trofile.
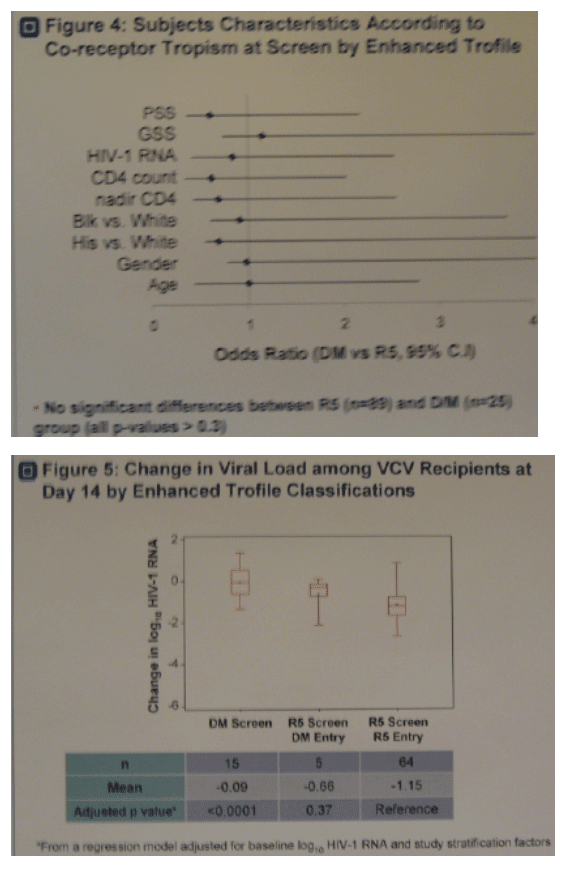
· Figure 4. No significant differences were observed in subjects characteristics according to tropism at screen by enhanced Trofile.
· Figure 5. Subjects with R5 virus at study screen and entry by enhanced Trofile had greater reductions (p=<0.0001) in viral load at day 14 (-1.15 log) compared to subjects with D/M virus at screen (-0.09 log).
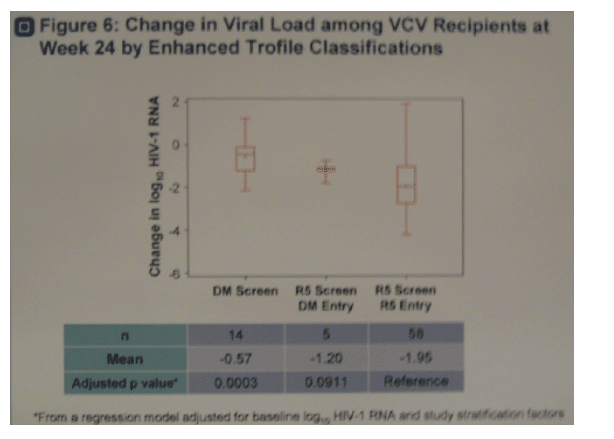
·Figure 6. Subjects with R5 virus at study screen and entry by enhanced Trofile had greater reductions (p=0.0003) in viral load at week 24 (-1.95 log) compared to subjects with D/M virus at screen (-0.57 log).
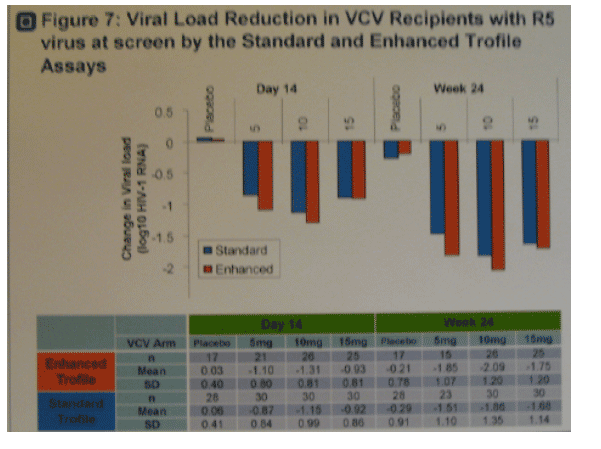
· Figure 7. Trend for improved virologic responses in VCV recipients with
R5 virus at screen by enhanced Trofile compared to standard Trofile.
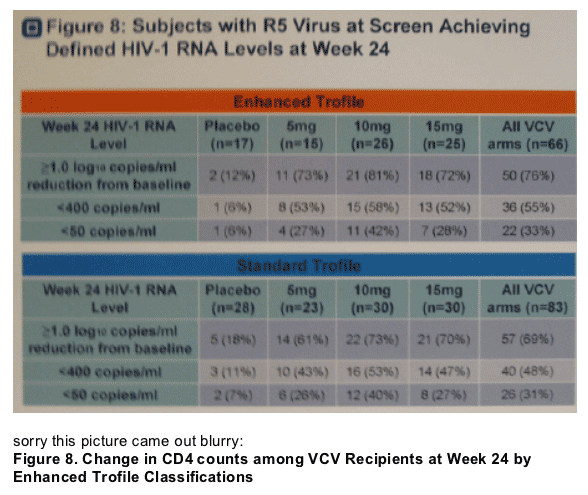
· Figure 8. A greater percent of VCV recipients with R5 virus at screen by enhanced Trofile compared to standard Trofile achieved defined HIV-1 RNA levels (≥1 log reduction from study entry, <400 copies/ml) at week 24.
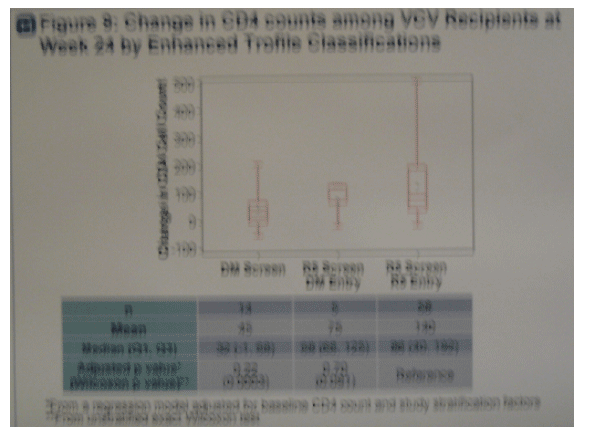
· Figure 9. Subjects with R5 virus at study screen and entry by enhanced Trofile had a trend for greater CD4 cell gains (+140) at week 24 compared to subjects with D/M virus at screen (+45).
These are pictures I took by camera of the poster.
ACKNOWLEDGEMENTS
We thank Dong Han, Neil Parkin, Jeannette Whitcomb and Chris Petropoulos for contributions to this study and are grateful to the Monogram Biosciences Clinical Reference Laboratory for assistance with assays. We also thank participating ACTG sites, ACTG/NIAID/NIH, and the patient volunteers. This study was supported in part by ACTG grant AI-68636.
REFERENCES
(1). Whitcomb et al. AAC Vol. 51, p. 566-575, 2007.
(2). Wilkin et al. CID Vol. 44, p. 591-595, 2007.
(3). Gulick et al. JID Vol. 196, p. 304-312, 2007.
(4). Trinh et al. XVII International Drug Resistance Workshop, Abstract 118, 2008.
(5). Reeves et al. 47th ICAAC, Abstract H-1026/283, 2007.
(6). Reeves et al. 11th European AIDS Conference, Abstract P1.2/03, 2007.
(7). Reeves et al. 3rd International Workshop on Targeting HIV Entry, Abstract 11, 2007.
|
| |
|
 |
 |
|
|