 |
 |
 |
| |
Multidrug Resistance in Antiretroviral Therapy-Experienced Patients in North America: 2000-2005
|
| |
| |
Reported by Jules Levin
17th HIV Drug Resistance Workshop
Sitges, Spain
June 10-14, 2008
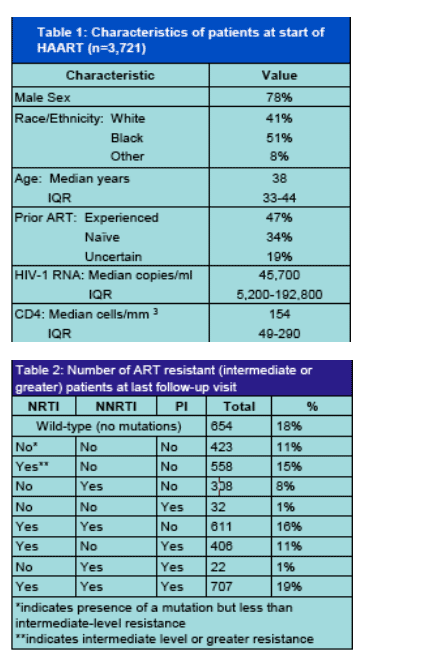
The study authors report 9% of patients are resistant to all 3 classes of ART drugs in 2000 and 8.5% in 2005. And, patients with resistance to PIs and NNRTIS or all 3 classes have the highest death rates. The percent of patients with resistance to all drugs in 2 classes is reported as 28% in 2000 and 16% in 2005. In 2005 patients resistant to any drugs within 3 classes: 16%, declined from 24%; patients resistant to any drugs within 2 classes in 2005: 33%. See Tables and Figure below.
Steven G. Deeks, M.D.1 and the North American AIDS Cohort Collaboration on Research and Design (NA-ACCORD)
1University of California - San Francisco, CA, USA
AUTHOR CONCLUSIONS
The prevalence of limited class resistance declined over time while the prevalence of high level multi-drug resistance remained stable, at least through 2005.
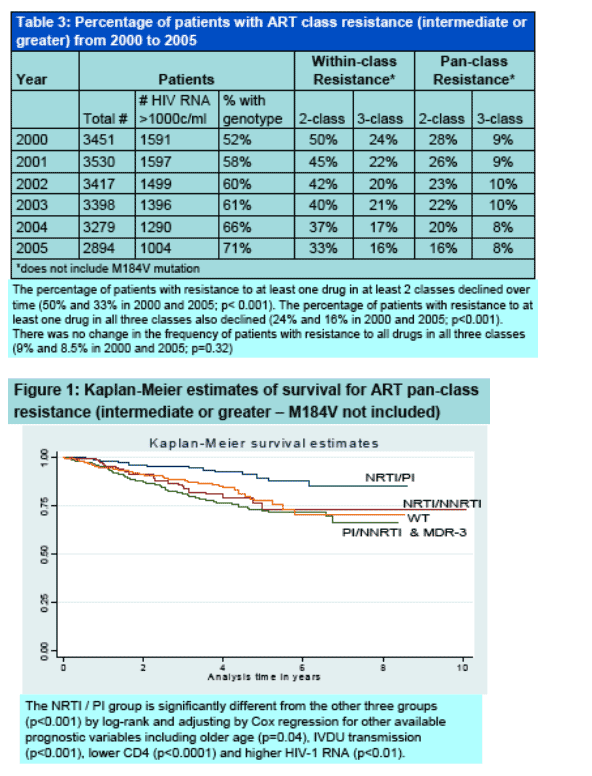
The overall mortality rates in those with resistance was high; this was particularly true for those with NNRTI resistance.
Individuals who had no evidence of resistance at the time of genotypic testing also had high mortality rates; presumably as this was a marker of non-adherence.
BACKGROUND
HAART has been available for over a decade, and a large but undefined number of patients have had virologic failure and developed multi-drug resistance. The continuing evolution of available antiretroviral drugs and classes has clearly altered the epidemiology of drug resistance, but the impact of these drugs also remains poorly defined.
No studies have had adequate sample size to determine broadly the calendar trends in multidrug resistant HIV-1 RNA in North America. In a large multisite collaboration of U.S. and Canadian HIV cohorts, we investigated the prevalence of MDR over time in patients who received HAART. We also investigated the impact of different resistance patterns on subsequent mortality.
METHODS
Study Sample: North American AIDS Cohort Collaboration on Research and Design (NA-ACCORD) is a consortium of 22 clinical and interval HIV cohorts from Canada and the U.S. It is a region of the International Epidemiologic Databases to Evaluate AIDS (IeDEA), consortium of 7 global cohorts in Africa (4 regions), Asia/Australia, Latin America and North America. The goal of the consortium is to address research questions in HIV/AIDS that cannot be answered with individual cohorts. The NA-ACCORD and IeDEA are supported by the National Institutes of Health.
Analysis: Eight clinical practice-based HIV cohorts contributed data on patients who had initiated HAART, and had a clinic visit during 2000-2005.
We identified all those who had had 2-class (NRTI,PI, or NRTI,NNRTI or PI/NNRTI) or 3-class (NRTI,NNRTI,PI) resistance after starting HAART. Resistance was based on the Stanford Genotypic Resistance Interpretation Algorithm (Version 4.3.3) and was defined by intermediate level or greater scores. Rates of resistance were computed annually from 2000-2005. To be included, a patient had to have at least one HIV-1 RNA in that calendar year.
We used two definitions of ART class resistance:
1) "Pan-class" - resistance to all available drugs in a class
2) "Within-class" - resistance to any available drugs in a class
Survival after development of ART resistance was also assessed using Kaplan-Meier plots and Cox proportional hazards regression.
RESULTS
NA-ACCORD Participating Cohorts:
AIDS Link to the IntraVenous Experience (ALIVE; Gregory Kirk)
Adult AIDS Clinical Trials Group Longitudinal Linked Randomized Trials
(ALLRT; Constance Benson, Ron Bosch, Ann Collier)
HIV Research Network (HIVRN; Kelly Gebo)
HAART Observational Medical Evaluation and Research (HOMER; Robert
Hogg, Richard Harrigan, Julio Montaner)
HIV Outpatient Study (HOPS; John T. Brooks)
Johns Hopkins HIV Clinical Cohort (JHHCC; Richard Moore)
Kaiser Permanente Northern California (Michael Horberg, Michael Silverberg)
Multicenter Hemophilia Cohort Study-II (MHCS-II;James Goedert )
Multicenter AIDS Cohort Study (MACS; Lisa Jacobson)
Montreal Chest Institute Immunodeficiency Service Cohort (Marina Klein)
Ontario HIV Treatment Network Cohort Study (OHTN; Sean Rourke, Anita
Rachlis)
John T. Carey Special Immunology Unit Patient Care and Research Database,
Case Western Reserve University (PCRD; Benigno Rodriguez)
Polaris HIV Seroconversion Study (Liviana Calzavara)
Research in Access to Care for the Homeless Cohort (REACH; David
Bangsberg)
Southern Alberta Clinic Cohort (John Gill)
Studies of the Consequences of the Protease Inhibitor Era (SCOPE; Steven
Deeks, Jeff Martin)
University of Alabama at Birmingham 1917 Clinic Cohort (Michael Saag)
University of North Carolina, Chapel Hill HIV Clinic Cohort (UCHCC; Joseph
Eron, Sonia Napravnik)
University of Washington HIV Cohort (Mari Kitahata)
Veterans Aging Cohort Study (VACS; Amy Justice)
Vanderbilt-Meharry CFAR Cohort (Timothy Sterling)
Women's Interagency HIV Study (WIHS; Stephen Gange)
Support: National Institute of Allergy & Infectious Diseases, with supplemental
funding from the National Cancer Institute.
Executive Committee: Richard Moore, Michael Saag, Stephen Gange, Mari
Kitahata, Rosemary McKaig (NIH), Aimee Freeman
Epidemiology/Biostatistics Core: Barry Merriman, Alison Abraham
Data Management Core: Stephen Van Rompaey, Eric Webster, Brenda Simon
Administrative Core: Carol Lent
|
| |
|
 |
 |
|
|