 |
 |
 |
| |
Merck's Longitudinal Analysis of Resistance to the HIV-1 Integrase Inhibitor Raltegravir: Results from P005 a Phase 2 Study in Treatment Experienced Patients
|
| |
| |
Reported by Jules Levin
XVII International HIV Drug Resistance Workshop
10-14JUN2008, Sitges, Spain
Michael D. Miller, Robert M. Danovich, Yuxiong Ke, Marc Witmer, Jing Zhao, Charlotte Harvey, Bach-Yen Nguyen, and Daria Hazuda for the P005 Study Team. Merck and Co, West Point PA 19486

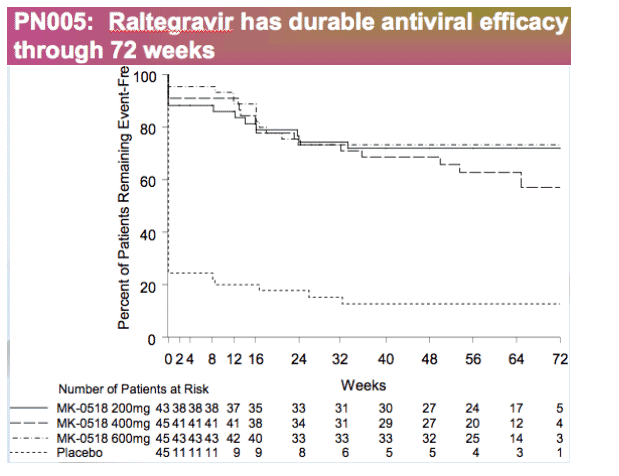
Phase 2 Protocol 005 (treatment-experienced)
Patients with 3-class-resistance received Optimized Background Therapy (OBT) + raltegravir (200, 400, or 600 mg b.i.d.) or OBT + placebo
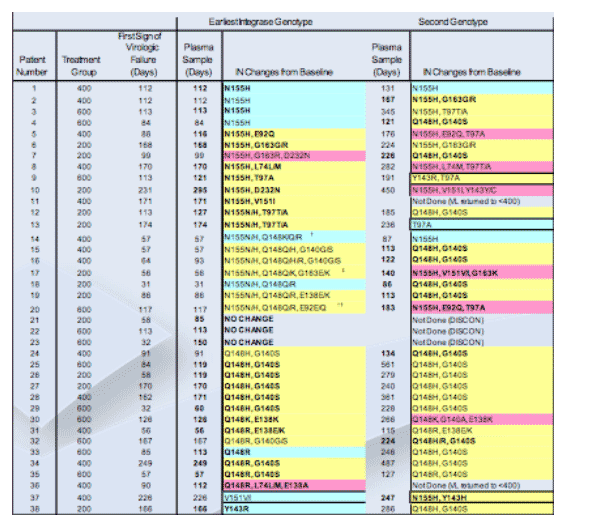
48 week efficacy: Virologic failure observed in 38/133 (28.6%) patients on RAL
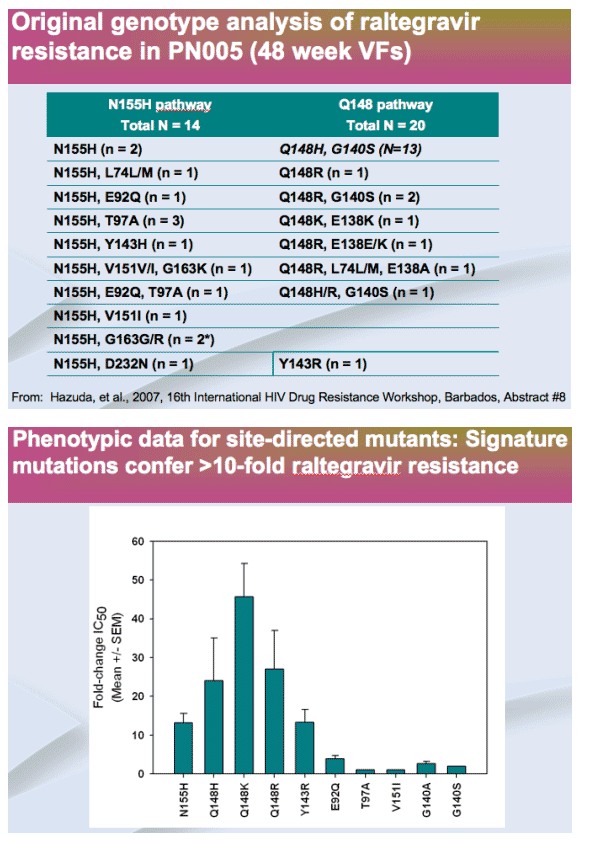
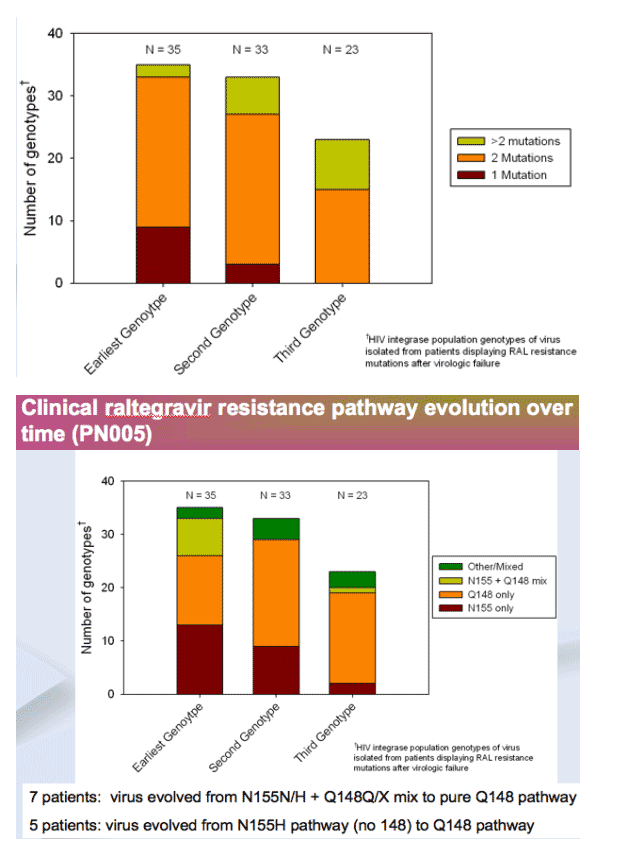
Initial resistance analysis (cross-sectional): Integrase signature mutations were observed in 35 of 38 patients failing RAL, usually with other mutation(s)
Summary of phenotypic data (see poster)
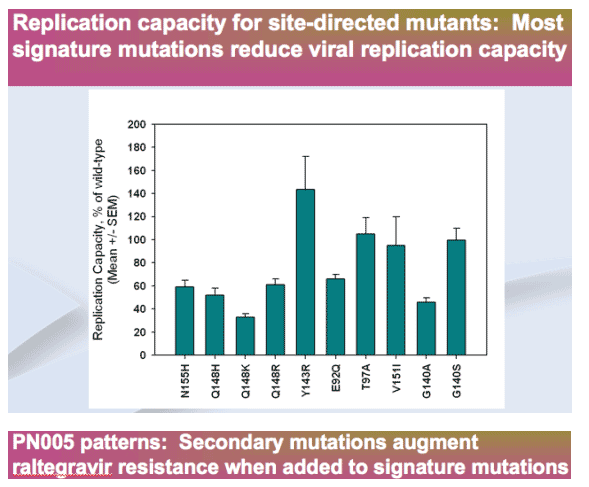
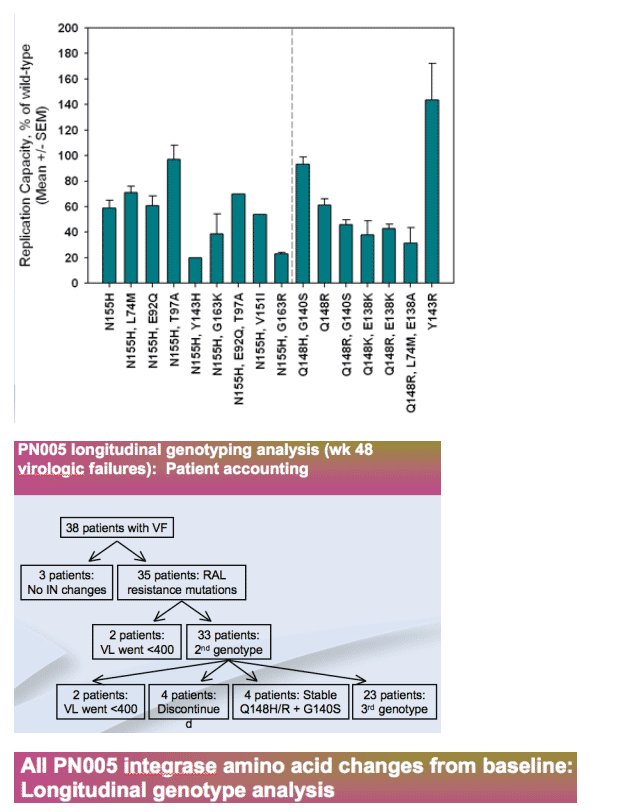
-- Signature mutations (N155H, Q148H/K/R) confer >10-fold RAL resistance and reduce replication capacity
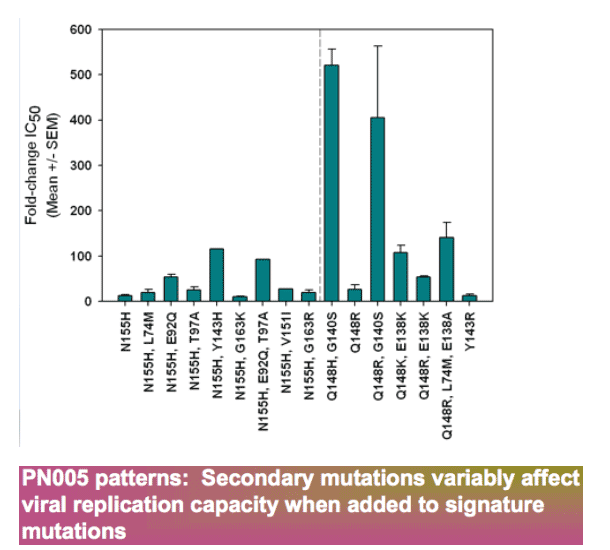
-- Secondary mutations consistently augment augment resistance, but have inconsistent effects on replication capacity
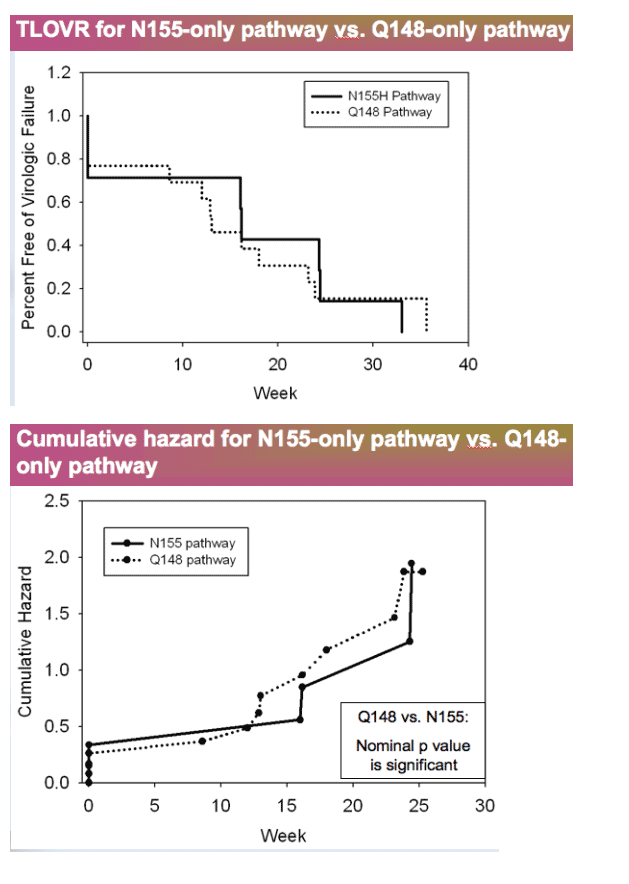
-- Q148 pathway has higher resistance than N155 pathway
|
| |
|
 |
 |
|
|