 |
 |
 |
| |
Resistance Mutations in HIV-1 Integrase Selected with Raltegravir or Elvitegravir Confer Reduced Susceptibility to a Diverse Panel of Integrase Inhibitors
|
| |
| |
Reported by Jules Levin
XVII HIV Drug Resistance Workshop
June 10-14, 2008
Sitges, Spain
Olivia Goethals, Reginald Clayton, Elisabeth Wagemans, Marcia Van Ginderen, Maxwell Cummings, Peggy Geluykens, Koen Dockx,
Veerle Smits, Geert Meersseman, Dirk Jochmans, Sabine Hallenberger, Kurt Hertogs.
Tibotec BVBA, Mechelen, Belgium
AUTHOR CONCLUSIONS
We suggest N155H, Q148R, E92Q and T66I as the most important RAMS conferring resistance to a diverse panel of integrase inhibitors (INIs), with Q148R for the first time selected in vitro with both RAL and EVG.
As the individual SDMs (site-directed mutants) R20K, V54I, L74M, A128T, E138K, G140A or S230R did not cause significant changes in susceptibility to EVG, RAL or other INIs, these mutations are presumably compensatory mutations. This observation will be further evaluated exploring the additive effect of each of these mutations to one of the RAMs N155H, Q148R, E92Q or T66I on susceptibility towards INIs and replication kinetics.
Abstract
BACKGROUND: The first generation integrase inhibitors, raltegravir (RAL, MK-0518) and elvitegravir (EVG, GS-9137), show great promise for the treatment of HIV infection. However, as with all antiretroviral drugs, resistance to integrase inhibitors (INIs) is expected to emerge. Currently, little is known regarding resistance associated mutations (RAMS) to INIs and the degree of cross resistance conferred by those mutations.
METHODS: We used in vitro selection (IVS) with MT4/IIIB to delineate the emergence of RAMS to raltegravir and elvitegravir, conducting 8 parallel
IVS experiments for each inhibitor. The resulting viruses were genotyped
and phenotyped, and mutants were generated by site-directed mutagenesis
(SDMs) to assess the effect of selected RAMS on the susceptibility to a panel
of INIs including raltegravir, elvitegravir, L-870,810, PYRAZ (a pyrimidoazepine),
PYCA (a pyrido-carboxamide) and L-731,988.
RESULTS:
1) Population genotyping of viruses selected with raltegravir highlighted one pathway towards resistance, i.e. Q148R followed by E138K, G140A and V54I. One selected virus (genotype Q148R E138K G140A V54I) showed greatly reduced susceptibility for both raltegravir and elvitegravir (>600-fold), and significantly reduced susceptibility to other INIs. An SDM carrying the Q148R mutation alone showed reduced susceptibility to raltegravir (22-fold) and elvitegravir (47-fold). Combined mutations Q148R and G140A further reduced susceptibility to raltegravir (>1000-fold) and elvitegravir (260-fold). 2) Resistant viruses selected with elvitegravir contained at least one of the following RAMS: Q148R, E92Q and T66I, in addition to further mutations (L74M, R20K, A128T, E138K and/or S230R). All selected viruses showed significantly reduced susceptibility to elvitegravir and other INIs, with only moderately reduced susceptibility to raltegravir. The SDMs E92Q and T66I were significantly reduced in susceptibility to elvitegravir (57-fold and 35-fold respectively), with small reductions in susceptibility to raltegravir.
CONCLUSIONS: The Q148R mutation is selected by both raltegravir and elvitegravir and conferred resistance to a diverse panel of INIs. Additionally, mutations selected with elvitegravir (E92Q and T66I) conferred significant resistance to many INIs, with a small reduction in susceptibility to raltegravir. We suggest that Q148R, E92Q and T66I be considered as RAMS conferring resistance to a diverse panel of INIs.
Introduction
Integration of retroviral DNA into the host genome is:
· An essential step in HIV life cycle.
· Catalyzed by the viral integrase (IN) enzyme.
· An attractive target for antiretroviral therapy.
Recent progress has resulted in two IN inhibitors (INIs):
· Elvitegravir (GS-9137, EVG) in late stage clinical trials (1).
· Raltegravir (MK-0518, RAL) recently approved for the use in treatment experienced HIV patients (2).
For all retroviral proteins representing clinically validated targets, inhibition by antiretroviral drugs has led to the emergence of resistance in drug-treated HIV infected individuals.
In vitro selection (IVS) of drug-resistant mutants is a useful tool to investigate the:
· Emergence of resistance pathways.
· Changes in drug susceptibility.
· Degree of cross-resistance between structurally related and different INIs.
· Resistance-associated and compensatory mutations.
and provide good indications of clinically relevant resistance pathways in vivo.
Here we report the results of IVS, growing HIV-1 in the presence of INIs EVG and RAL, and the investigation of mutations that arise in the presence of and confer reduced susceptibility to those inhibitors and an extended panel of structurally diverse INIs.
METHODS
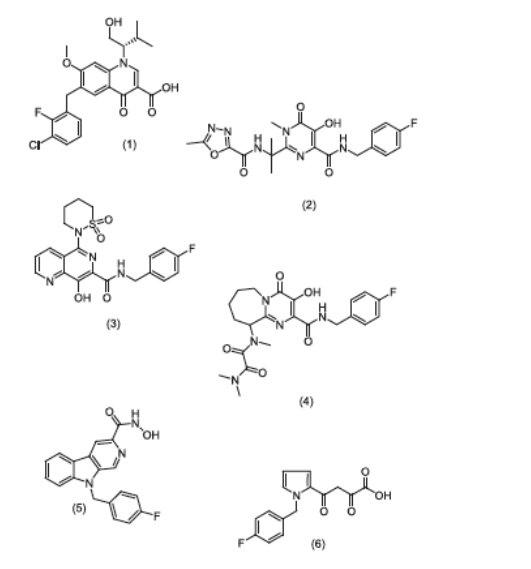
Figure 1. Structure of six structurally different INIs.
(1) EVG; (2) RAL; (3) L-870,810; (4) a pyrimido-azepine (PYRAZ), Merck; (5) a Pyrido-carboxamide (PYCA), Pfizer; (6) L-731,988
Automated in vitro selection experiments in 96-well plates.
IVS was performed in 96-well plates. Each row represents a separate experiment (3).
Genotyping of the viral pools.
Viral RNA was extracted from culture supernatant or virus stock (NucliSens easyMag, bioMérieux). cDNA encoding reverse transcriptase and IN was generated with Expand Reverse Transcriptase (Roche Diagnostics), followed by amplification of the IN region by nested PCR (1456 bp). PCR products were purified (QIAquick kit) and genotyped by automated population-based full sequence analysis (ABI PRISM BigDye Terminator cycle sequencing, Applied Biosystems).
Antiviral assay.
The antiviral activity (EC50) of the different inhibitors was determined in a cell-based HIV-1 replication assay (4).
Generation of the SDM virus stocks.
Site-directed mutants (SDMs) of IN coding sequences were constructed in the pUC19-5'HXB2D vector (XbaISalI fragment of pHXB2D), containing the HIV-1 clone HXB2D IN coding sequence, by using the QuikChange site-directed mutagenesis kit (Stratagene, La Jolla, CA) and HPLC-purified primers (Genset Olivigo, La Jolla, CA).
MT4 cells were pelleted and resuspended in phosphate-buffered saline at a concentration of 3.1x106 cells/ml. A 0.8 ml portion (2.5x106 cells/ml) was used for each transfection. Cells were electroporated with 10 _g of SalI-linearized pUC
19-3'HXB2D (SalI-XbaI fragment of pHXB2D) and 5 _g SalI-digested SDM. Cell cultures were incubated at 37°C and were monitored for the appearance of cytopathic effect (CPE). At virus breakthrough (full CPE), culture supernatant was typically harvested by centrifugation at 8 to 10 days after transfection and was stored at -80°C for subsequent determination of drug susceptibility.
RESULTS
Table 1: Genotypes of eight mutant viruses from the automated IVS experiments with EVG at a final drug concentration of 3 _M.
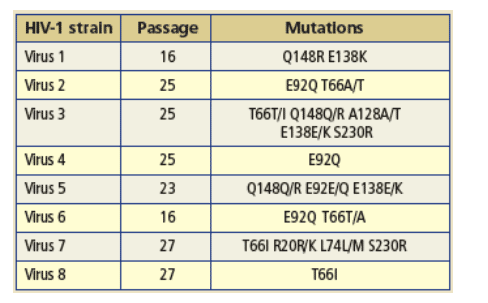
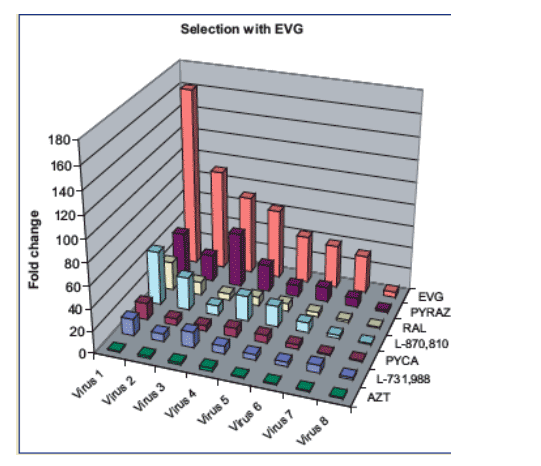
Figure 2: Cross-resistance of EVG-resistant mutants with other INIs.
The viruses from IVS with EVG were tested for their susceptibility to EVG and other structurally different INIs. The fold changes in EC50 of the mutant viruses compared with wild type HIV IIIB virus are displayed.
· Resistant viruses selected in vitro in the presence of EVG contained:
-- At least one of the following RAMS: Q148R, E92Q and T66I.
-- Additional mutations: L74M, R20K, A128T, E138K and/or S230R.
Previously reported to be associated with resistance to EVG:
-- T66I and E92Q: in vitro resistance (5).
-- Q148R, E138K, E92Q and T66I: in vivo resistance after clinical failure to EVG (6).
Table 2: Genotypes of eight viruses from the automated IVS experiments with RAL at a final drug concentration of 6 _M.
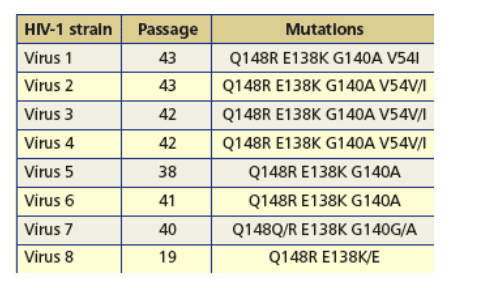
Figure 3: Cross-resistance of RAL-resistant mutants with other INIs.
The viruses from IVS with RAL were tested for their susceptibility to RAL and other structurally different INIs. The fold changes in EC50 of the mutant viruses compared with wild type HIV IIIB virus are displayed.
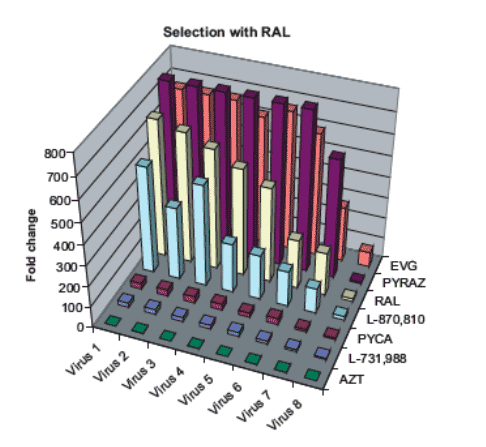
· Resistant viruses selected in vitro in the presence of RAL contained:
-- One RAM: Q148R.
-- Additional mutations: E138K, G140A and V54I.
Previously reported resistance mutations to RAL:
-- Q148R, E138K and G140A: in vivo resistance after clinical failure to RAL (7).
Figure 4: Susceptibility to INIs of HIV-1 IN recombinant viruses (SDMs).
The IN mutations observed from IVS were incorporated as SDMs in the backbone of HIV-1 (HXB2D). The fold changes in EC50 of the mutant viruses, compared with wild type HIV HXB2D virus, are shown.
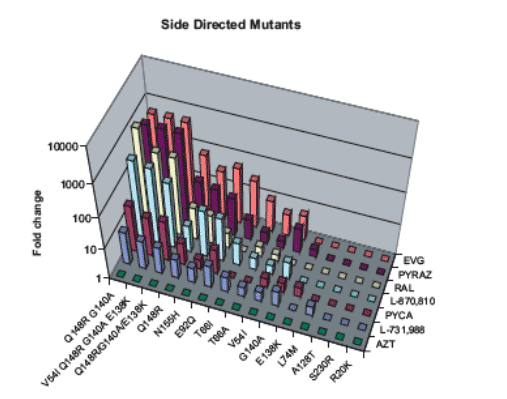
· The mutation Q148R conferred significantly reduced susceptibility to EVG (74-fold), RAL (10-fold) and other structurally diverse INIs.
· While conferring only a small (3-fold) reduction in susceptibility to RAL, E92Q showed large reductions in susceptibility to EVG (54-fold) and the other structurally diverse INIs.
· The mutation T66I caused a 35-fold reduction in susceptibility to EVG but retained near wild-type susceptibility to RAL. This mutation was also able
to reduce susceptibility to the other structurally diverse INIs albeit to a lesser degree than Q148R and E92Q.
· The mutations R20K, V54I, L74M, A128T, E138K, G140A and S230R were observed accompanying one or more of the three most critical RAMs, Q148R, E92Q and T66I. However, as single mutations in the context of SDMs, they resulted in little or no change in susceptibility to EVG, RAL, or any other INI.
· N155H, a clinically relevant mutation (6,7), confers reduced susceptibility to EVG and RAL by 32-fold and 8-fold, respectively.
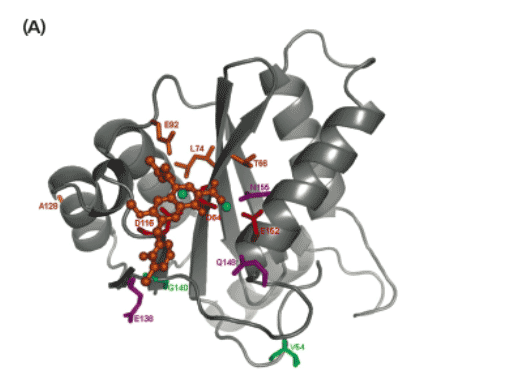
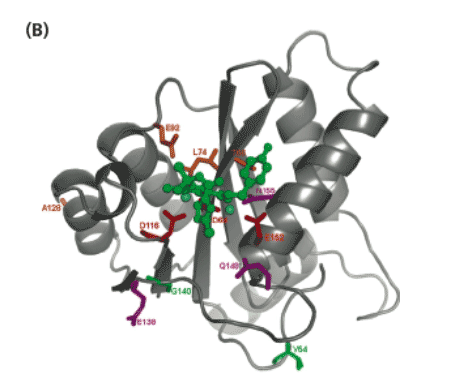
Figure 5. Mapping of the IN mutations within the HIV-1 IN catalytic site.
Three-dimensional docking model of the catalytic core domain (CCD) of HIV-1 IN, based on the model of Savarino (8), with EVG docked in the CCD (orange) (A) or with RAL docked in the CCD (green) (B). The docking of RAL is based on low energy docking (with the program GLIDE) and represents one plausible RAL binding mode. The catalytic triad residues (D64, D116 and E152) are shown in red and the magnesium ions in cyan. Amino acid residues conferring resistance to both EVG and RAL are shown in purple. Amino acid residues conferring resistance to EVG only are shown in orange and amino acid residues conferring resistance to RAL only are shown in green.
REFERENCES
1. Sato, M. et al., 2006; J Med Chem; 49:1506-8.
2. Cahn, P., and O. Sued. 2007; Lancet; 369:1235-6.
3. Jochmans, D. et al., 2007; XVIth IHDRW, Barbados, West Indies. Abstract 23.
4. Jochmans, D., et al., 2006; J Virol 80:12283-92.
5. Shimura, K. et al., 2008; J Virol; 82:764-74.
6. McColl, D. et al., 2007; Antivir Ther; 12: S11. Abstract 9.
7. Hazuda, D. et al., 2007; Antivir Ther; 12:S10. Abstract 8.
8. Savarino, A., 2007; Retrovirology; 4:21.
|
| |
|
 |
 |
|
|