 |
 |
 |
| |
Technical Validation of an Enhanced Sensitivity Trofile HIV Co-receptor Tropism Assay for Selecting Patients for Therapy with Entry Inhibitors Targeting CCR5
|
| |
| |
Reported by Jules Levin
XVII HIV Drug Resistance Workshop
June 10-14, 2008
Sitges, Spain
Lan Trinh, Dong Han, Wei Huang, Terri Wrin, Jeffrey Larson, Linda Kiss, Eoin Coakley, Christos Petropoulos, Neil Parkin, Jeannette Whitcomb, and Jacqueline Reeves*
Monogram Biosciences, South San Francisco, CA, USA
AUTHOR SUMMARY & CONCLUSIONS
Validation experiments demonstrate that enhanced Trofile is accurate, precise, reproducible, and has improved sensitivity to detect low levels of CXCR4-using variants in env clone mixtures and patient env populations compared to standard Trofile.
These data support adoption of enhanced Trofile as the assay of choice for co-receptor tropism determination of patient virus populations.
BACKGROUND
Several entry inhibitors which block infection via CCR5 are in clinical development and maraviroc, a CCR5 antagonist, has recently been approved for the treatment of HIV patients with CCR5-tropic (R5) virus.
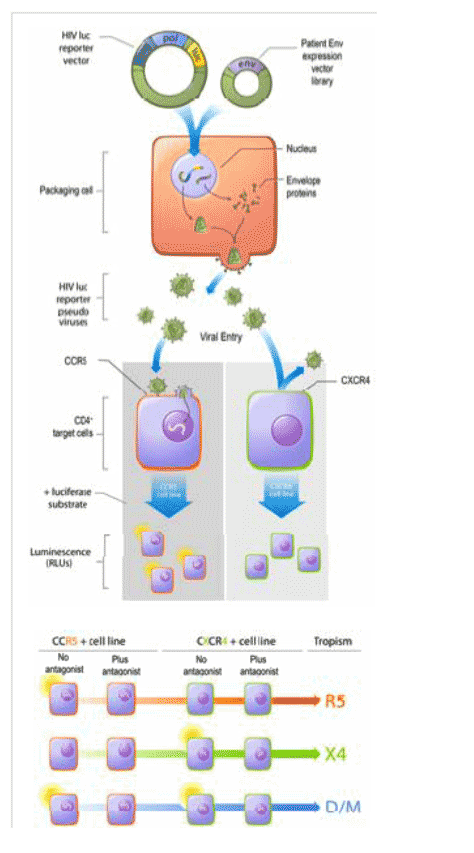
The TrofileTM HIV co-receptor tropism assay (Monogram Biosciences) (Figure 1) determines whether a patient's viral population is CCR5 (R5), CXCR4 (X4) or dual (R5/X4)/mixed (D/M)-tropic, and is useful for selecting patients for appropriate treatment with CCR5 antagonists. The clinical utility of Trofile has been unequivocally demonstrated in trials of CCR5 antagonists including maraviroc and vicriviroc.
In mixed envelope (env) populations, the standard Trofile assay was validated to detect minor R5 and X4 variants at 10% and 5% of the population with 100 and 85% sensitivity, respectively (Figure 2) (levels below 5% were not tested) (1).
Low level CXCR4-using variants below the detection limit of standard Trofile can sometimes be identified by clonal analysis of patient Env populations and may be selected following therapy with CCR5 antagonists.
Recent studies suggest that detection of patients with lower levels of CXCR4-using variants may further optimize patient selection (2,3). We therefore validated
an enhanced sensitivity version of Trofile that allows an average 30-fold improved detection of minor CXCR4-tropic variants in env clone mixtures (100% sensitivity at detecting 0.3% X4 Envs) and earlier detection of minor CXCR4-using subpopulations in longitudinal samples from PR/RT inhibitor experienced patients (4,5,6).
METHODS
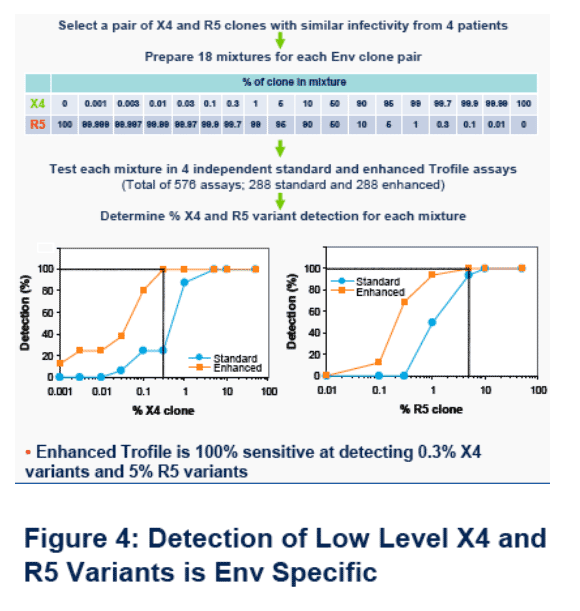
Experiments were performed to validate the performance of enhanced Trofile for patient management applications in compliance with CAP and CLIA regulations. The co-receptor tropism of a panel of well characterized viral isolates and patient-derived HIV-1 envelopes (Envs) were evaluated to assess assay accuracy. Precision and reproducibility were assessed by replicate testing. Minor variant sensitivity was determined using mixtures of paired R5 and X4 env clones derived from 4 patients. R5 and X4 pairs were selected to exhibit similar infectivity of CCR5 and CXCR4 expressing cells, respectively.
RESULTS
Figure 2. Enhanced Trofile accurately determined the coreceptor tropism of a panel of 46 well characterized Envs representing multiple subtypes and clonally analyzed patient Env populations. Intra-assay precision (100%) and interassay reproducibility (99%) were demonstrated from concordant results for 135/135 and 228/230 pair-wise comparisons of R5, X4 and DM Envs clones and from repeat testing of 46 patient Env populations, respectively. Assay validation performance characteristics were equivalent between the standard and enhanced Trofile assays, with the exception that enhanced Trofile is validated to detect 0.3%
X4 Envs with 100% sensitivity, compared to 10% minor variant with 100% sensitivity for standard Trofile.
· Figure 3. Across 288 assays to evaluate minor variant sensitivity, Enhanced Trofile detected X4 clones in 100% of assays when present at 0.3% and in 81% of assays at 0.1%. R5 clone detection was also improved compared to standard Trofile, with 5% R5 clones detected in 100% of assays and 1% in 94% of assays.
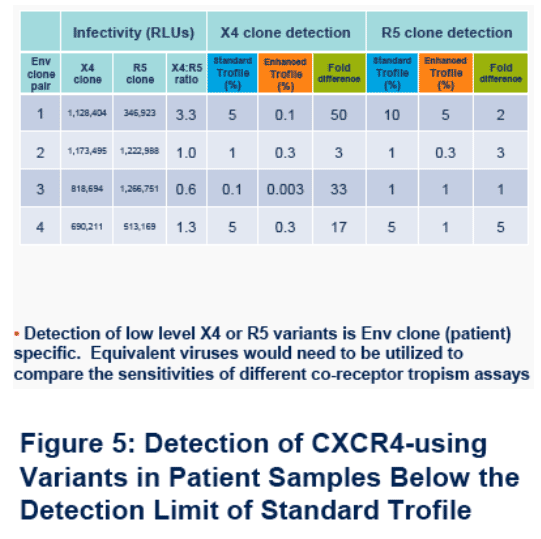
· Figure 4. Detection of X4 and R5 variants was env clone pair (patient) dependent and ranged from 0.003-0.3% for X4 clones and 0.3-5% for R5 clones.
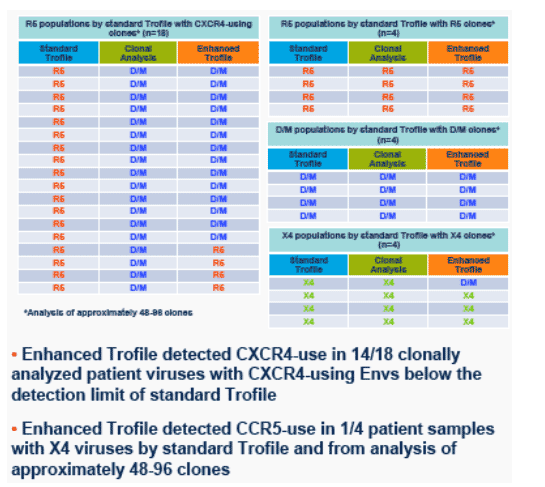
· Figure 5. Enhanced Trofile detected CXCR4-use in 14/18 clonally analyzed patient samples with CXCR4-using variants below the level of detection of the standard Trofile assay and detected CCR5-use in 1/4 clonally analyzed X4 patient samples.


SUMMARY & CONCLUSIONS
Validation experiments demonstrate that enhanced Trofile is accurate, precise, reproducible, and has improved sensitivity to detect low levels of CXCR4-using variants in env clone mixtures and patient env populations compared to standard Trofile.
These data support adoption of enhanced Trofile as the assay of choice for co-receptor tropism determination of patient virus populations.
REFERENCES
(1). Whitcomb et al. AAC Vol. 51, p. 566-575, 2007.
(2). Reeves et al. 15th CROI, Abstract 869, 2008.
(3). Su et al. XVII International HIV Drug Resistance Workshop, Abstract 88, 2008.
(4). Reeves et al. 47th ICAAC, H-1026/283, 2007.
(5). Reeves et al. 11th European AIDS Conference, P1.2/03, 2007.
(6). Reeves et al. 3rd International Workshop on Targeting HIV Entry, 11, 2007.
|
| |
|
 |
 |
|
|