 |
 |
 |
| |
Virological response and safety of BI 201335 protease inhibitor, peginterferon alfa 2a and ribavirin treatment of HCV genotype-1patients with compensated liver cirrhosis and non-response to previous peginterferon/ribavirin
|
| |
| |
Reported by Jules Levin
AASLD, Nov 2 2009, Boston, MA
Stanislas Pol,1 Thomas Berg,2 Maurizio Bonacini,3 Marcus Schuchmann,4 Jacob Lalezari,5 Andreas Erhardt,6 Marc Bourliere,7 Michael Manns,8 Chan-Loi Yong,9 Gerd Steinmann,10 Jerry Stern,9 Joe Scherer,9 Wulf O. Boecher10
1Hopital Cochin, Paris, France; 2Charite, Campus Virchow-Klinikum, Berlin, Germany; 3CPMC Research Institute, San Francisco, CA; 4Internal Medicine 1, University Hospital Mainz, Germany; 5Quest Clinical Research, San Francisco, CA; 6Internal Medicine, University Hospital Duesseldorf, Germany;7Hopital Saint Joseph, Marseilles, France; 8Department of Gastroenterology, Hepatology and Endocrinology, Hannover Medical School, Hannover, Germany; 9Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, CT; 10Boehringer Ingelheim Pharma GmbH, Biberach, Germany

ABSTRACT
Background: BI 201335 is a highly potent and specific HCV NS3/4A protease inhibitor. A phase 1 trial in treatment-experienced HCV GT-1 patients demonstrated a mean viral load (VL) reduction of 5.3 log10 (IU/mL) for BI 201335 given once daily after 28 days in combination with peginterferon alfa (PegIFN) 2a and ribavirin (RBV). We now describe a phase 1b trial which has assessed safety, short-term efficacy, and pharmacokinetics of BI 201335 in GT-1 patients with compensated liver cirrhosis and non-response to previous PegIFN/RBV, a difficult-to-treat HCV population with a high unmet medical need.
Methods: In this open-label, sequential group comparison, HCV GT-1 patients with compensated liver cirrhosis who have never achieved undetectable VL under previous PegIFN/RBV were treated with 240 mg once (QD; n=6) or twice daily (BID; n=7) in combination with PegIFNa2a (180 mcg/week) and RBV (1000/1200mg/d) for 28 days. All patients received a single loading dose of 480mg of BI 201335 as the first dose. Plasma HCV RNA was measured by Roche COBAS TaqMan assay.
Results: Mean age was 54 years, BMI 26 kg/m2. Mean VL at baseline was 6.0 and 6.6 log10 (IU/mL) in both groups. All patients showed a rapid and continuous decline in VL. Mean VL declines on day 28 in the 240mg QD and BID groups were -4.9 and -5.0 log10, respectively. No breakthrough (>0.8 log10 rebound from VL nadir) was observed during treatment.
At day 28, 5/6 and 5/7 patients achieved VL below level of quantification (< 25 IU/ml) in the QD and BID group. Furthermore, 4/6 and 1/7 patients had VL below level of detection (<10 IU/ml) in the 240mg QD and BID groups.
There were no SAE in the 240mg QD group and 2 SAE in the 240mg BID group. Both were cases of mild to moderate hepatic decompensation attributed to PegIFN/RBV by the investigators. Two patients in the BID group discontinued treatment early, one due to nausea, one due to hepatic decompensation (SAE).
Jaundice due to isolated unconjugated hyperbilirubinemia was reported in 2/6 and 1/7 patients at 240mg QD and BID, respectively. Other AE were mainly mild to moderate and typical of PegIFN/RBV. Lab analyses showed decreases of ALT / AST as well as blood cell counts typical of PegIFN/RBV.
Conclusions: BI 201335 once or twice daily combined with PegIFN/RBV exhibited potent antiviral activity in non-responder patients with liver cirrhosis. BI 201335 also exhibited a good safety and tolerability profile in these patients, allowing for their inclusion into the ongoing phase 2 program. These data also confirm that IFN non-responsiveness in previous non-responders can be overcome by rapid and profound inhibition of viral replication by BI 201335.
INTRODUCTION
BI 201335 is a highly potent and specific hepatitis C virus (HCV) NS3/4A protease inhibitor
A phase 1 trial in treatment-experienced (TE) HCV genotype-1 (GT-1) patients demonstrated a mean viral load (VL) reduction of 5.3 log10 (IU/mL) for BI 201335 given 240 mg once daily (QD) after 28 days in combination with peginterferon alfa (PegIFN) 2a and ribavirin (RBV)
We now describe a phase 1b trial which has assessed safety, short-term efficacy, and pharmacokinetics of BI 201335 in GT-1 patients with compensated liver cirrhosis and virologic non-response to previous PegIFN/RBV, a difficult-to-treat HCV population with a high unmet medical need
METHODS
This trial was an open label, sequential group, dose-escalating comparison of 240 mg BI 201335 QD (n=6) or twice daily (BID; n=7), given orally for 28 days in combination with PegIFN (180 µg/week SC) and RBV (weight based: 1,000 or 1,200 mg daily) (Figure 1)- all patients received a single loading dose of 480 mg BI 201335 as the first dose
- the 240 mg BID cohort started enrollment after review of safety and efficacy data from the 240 mg QD cohort, and release by a Data Monitoring Committee (DMC)
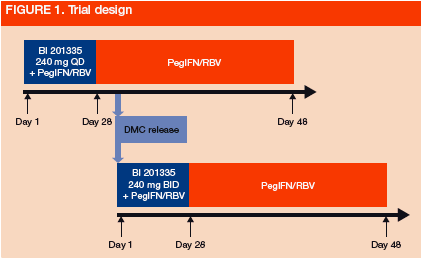
HCV GT-1 patients were entered if they had compensated biopsy-proven liver cirrhosis (Child-Pugh-Turcotte Score A; CPT A) and had never achieved undetectable VL under previous PegIFN/RBV treatment for at least 12 weeks
Only patients with partial response (maximum VL reduction>1 log10 from baseline but never achieved undetectableHCV RNA at any time) or null-response (maximum VL reduction <1 log10 from baseline at any time) were entered; relapserswere excluded
Patients with a history of decompensated liver cirrhosis were excluded
Plasma HCV RNA was measured by Roche COBAS TaqMan assay, with a lower limit of quantification (LLQ) of 25 IU/mL and a lower limit of detection (LLD) of 10 IU/mL
RESULTS
Baseline demographics
Mean age of entered patients was 54 years, body mass index (BMI) 26 kg/m2
Mean VL at baseline was 6.0 and 6.6 log10 (IU/mL) in the240 mg QD and BID groups, respectively- baseline demographics are shown in Table 1
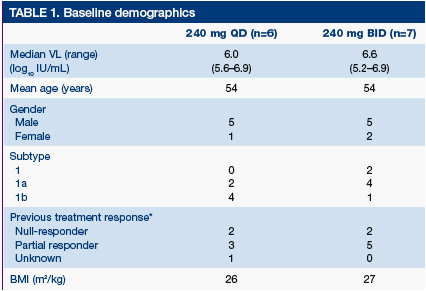
*Partial responder, maximum VL reduction >1 log10 from baseline but never achieved undetectable HCV RNA; null-responder, maximum VL reduction <1 log10 from baseline at any time
EFFICACY
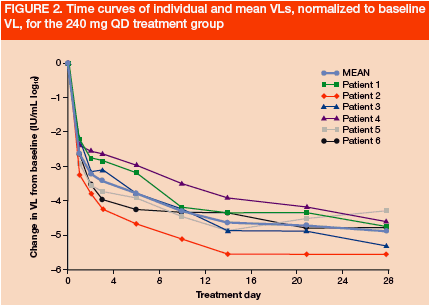
All patients showed a rapid and continuous decline in VL (Figures 2 and 3). Mean VL declines on Day 28 in the 240 mg QD and BID groups were -4.8 and -5.0 log10, respectively
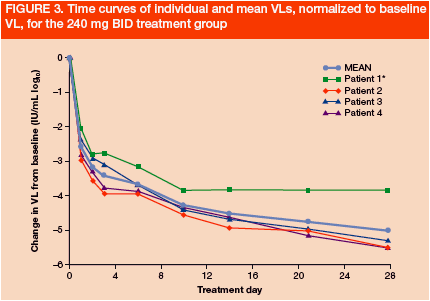
n=4 for VL reduction at 240 mg BID due to lack of baseline VL in 3 patients.
*Patient 1 was <25 IU/mL at Day 10 and <10 IU/mL at Day 29 and later time points
No breakthrough (>0.8 log10 rebound from VL nadir) was observed during treatment
At Day 28, 5/6 and 6/7 patients, respectively, achieved VL below the LLQ in the QD and BID groups- 4/6 and 1/7 patients had VL below the LLD in the 240 mg QD and BID groups, respectively (Table 2)
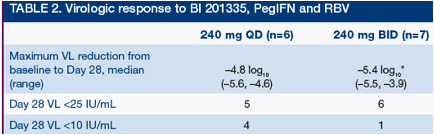
*n=4 for VL reduction at 240 mg BID due to lack of baseline VL in 3 patients
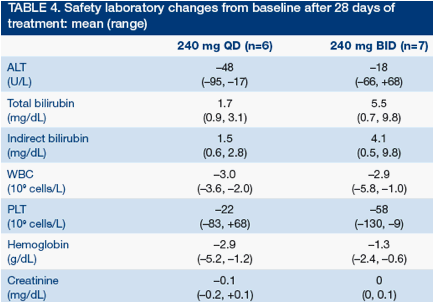
Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) decreased in the majority of patients
Safety and tolerability
Safety and tolerability was good in both dose groups and similar to non-cirrhotic patients.1 Adverse events (AEs) were mostly mild-to-moderate and typical of PegIFN/RBV (Table 3)
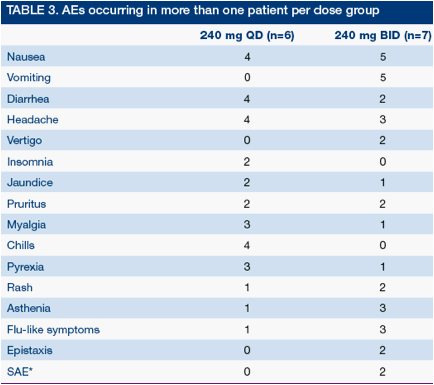
*SAEs due to hospitalization for metabolic decompensation of moderate intensity, recovered and related to PegIFN/RBV by judgment of investigator
Mild-to-moderate jaundice due to isolated unconjugated hyperbilirubinemia was reported in 2/6 and 1/7 patients at240 mg QD and BID, respectively (maximum bilirubin12.7 mg/dL, see below)
There were no serious AEs (SAEs) in the 240 mg QD group and 2 SAEs in the 240 mg BID group
- 1 patient with a low baseline CPT Class B score (high CPT A at screening) was diagnosed with transient jaundice and ascites due to worsening hypoalbuminemia after completion of 28 days of BI 201335, that was attributed to PegIFN/RBV by the investigator. The event was successfully treated allowing the patient to continue PegIFN/RBV thereafter without interruption
- a second patient with a low baseline CPT Class A score experienced an asymptomatic rise of conjugated and unconjugated bilirubin peaking at Day 24 (total bilirubin 12.7 mg/dL, predominantly direct bilirubin, without increase in other liver laboratory tests). Study medications were discontinued as defined in the protocol for bilirubin >10 x ULN and full bilirubin recovery occurred within 8 days
There were 2 discontinuations in the BID group; 1 described above and another in a patient suffering from moderate nausea (Day 6)
Safety laboratory analyses showed that blood cell counts were reduced as is typical for PegIFN/RBV. Isolated unconjugated hyperbilirubinemia was more common and more pronounced at the 240 mg BID dose level (Table 4)
REFERENCE
1. Manns MP, et al. Safety and antiviral activity of BI 201335, a new HCV NS3 protease inhibitor, in combination therapy with peginterferon alfa-2a (P) and ribavirin (R) for 28 days in P+R treatment-experienced patients with chronic hepatitis C genotype 1 infection. The 59th Annual Meeting of the American Association for the Study of Liver Diseases (AASLD), San Francisco, CA, USA; 2008. Abstract 1882.
|
| |
|
 |
 |
|
|