 |
 |
 |
| |
Boceprevir in Genotype 1 Null-Responders with Peg/RBV Lead-In
|
| |
| |
Reported by Jules Levin
AASLD Nov 2 2009 Boston, MA
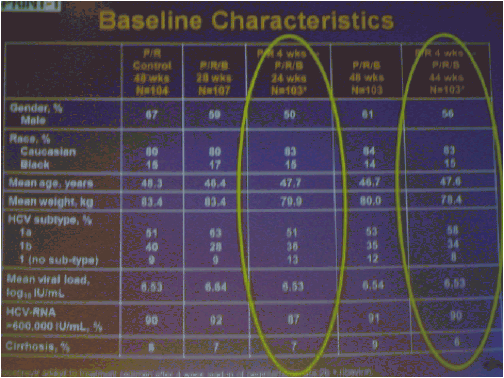
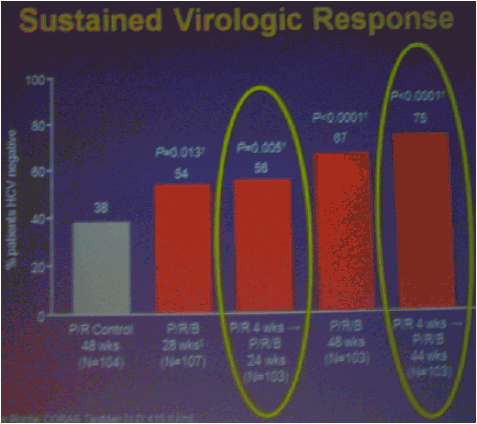
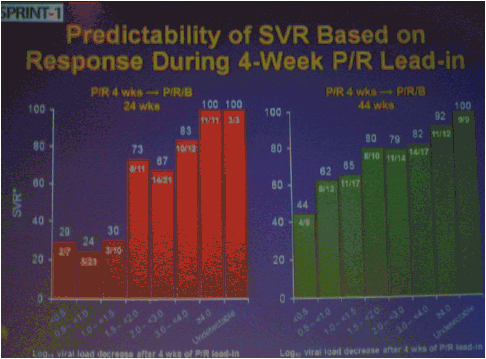
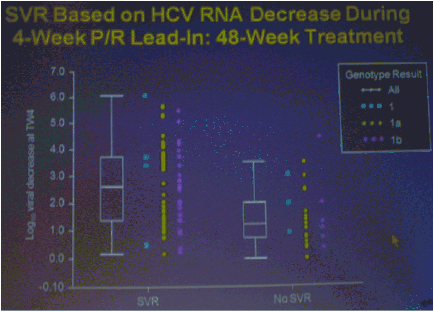
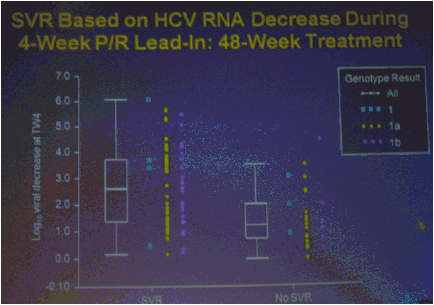
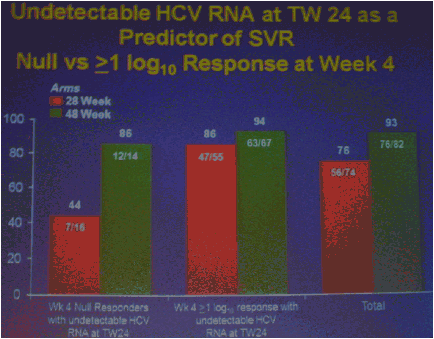
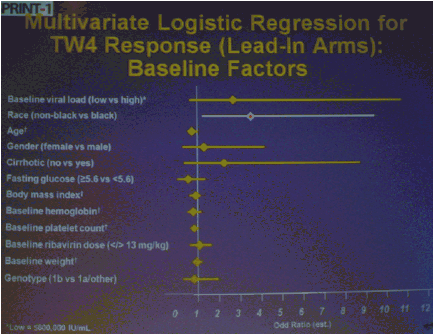
The numbers of patients in this phase 2 study are relatively small but the study results outline an approach to achieving perhaps surprisingly high SVR rates in null responders with a peg/rbv lead-in. The study investigator who presented this oral talk said: "Although the numbers are small, this analysis of the HCV SPRINT-1 study data showed that it was possible to achieve SVR in a proportion of null responders to peginterferon and ribavirin when boceprevir was added to their backbone regimen,".... "However, the risk of developing viral resistance to protease inhibitors in patients who do not achieve SVR must be carefully weighed against the potential benefits of treatment with this new class of direct antiviral agents. With the lead-in strategy, initial peginterferon and ribavirin responsiveness is determined prior to the addition of a protease inhibitor, thus allowing the physician to take into account the potential for the development of resistance." said Paul Kwo, M.D. So patients who cannot wait for next generation therapy where in several years we expect therapy to consist of 2 oral drugs plus peg/RBV where SVR rates are likely to approach 85%, then these patients who cannot wait due to advanced HCV disease using boceprevir or telaprevir plus peg/rbv is an option. But, for patients who can wait for better therapy can consider waiting. In the future therapies will consist of 2 to 3 or 4 oral drugs with or without peg/rbv. The studies must however be first conducted to see if peg/rbv can be eliminated. Of note patients received a 4 week lead-in of peg/RBV before adding the oral protease inhibitor drug boceprevir and patients with <0.5 to <1.5 log viral load reductions during the 4 week lead-in (null-responders as defined by study) achieved 24%-30% SVR rates with a total of 24 weeks therapy with boceprevir+peg/rbv and with 48 weeks therapy achieved 44-65% SVR rates, see graphs below. Null-responders with >1.5-2.0 log response during lead-in achieved 73% SVR with 28 weeks therapy and the bigger the response during lead-in the greater the SVR rate. For patients receiving 48 weeks therapy, those with <0.5 log reduction during lead-in achieved 44% SVR, those with 0.5-<1.0 achieved 62% SVR rate, 1.0-<1.5 log achieved 65% SVR rate, those with >1.5-2.0 80% achieved SVR and with increasing peg/rbv viral responses during lead-in achieved proportionately higher SVR rates. Null responders with 28 weeks therapy undetectable HCV RNA at week 24, 44% (7/16) had SVR, 86% with 48 weeks therapy. For patients with a wk 4 >1.0 log response 86% with 28 weeks & 94% with 48 weeks therapy achieved SVR.






|
| |
|
 |
 |
|
|