 |
 |
 |
| |
A Phase 1b Dose-Ranging Study of 4 Weeks of PEG-Interferon (IFN) Lambda (PEG-rIL-29) in Combination with Ribavirin (RBV) in Patients with Chronic Genotype 1 Hepatitis C Virus (HCV) Infection
|
| |
| |
Reported by Jules Levin
AASLD Nov 2 2009, Boston, MA
Andrew J. Muir1, Mitchell L. Shiffman2, Atif Zaman3, Boris Yoffe4, Juan Carlos Lopez-Talavera5, Sherri Souza6, Diana F. Hausman6, Naomi N Hunder6, David Fontana6,Eric Lawitz7
1Duke University, Durham, NC, USA; 2VCU Medical Center, Richmond, VA, USA; 3Oregon Health Sciences University, Portland, OR, USA; 4Baylor College of Medicine, Houston, TX, USA; 5Bristol-Myers Squibb, Research and Development, Princeton, NJ, USA; 6ZymoGenetics Inc, Seattle, WA, USA; 7Alamo Medical Research, San Antonio, TX, USA.
AUTHOR CONCLUSIONS
PEG-IFN-λ was associated with robust antiviral activity at all dose levels tested in both IFN-α relapse and treatment-naïve HCV patients
Majority of patients achieved >2 log reduction in HCV RNA during the 4-week treatment period; anti-viral activity appears to be dose-dependent
Treatment was generally safe and well-tolerated
Minimal constitutional symptoms or myelosuppressive hematologic effects were observed
Dose-limiting toxicities of elevations in ALT or AST with or without increased bilirubin were reversible upon treatment cessation
PK data suggest that PEG-IFN-λ exposure is dose dependent and support once-weekly dosing and exploration of fixed dosing
These results support the initiation of dose-ranging Phase 2 studies in treatment-naïve HCV patients
ABSTRACT
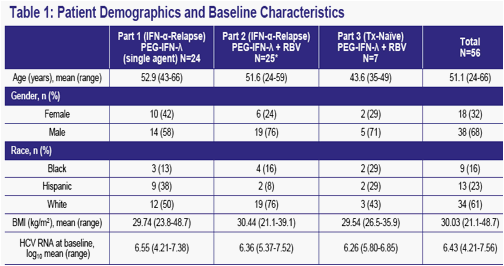
Background: PEG-IFN-lambda (PEG-IFN-λ) is a unique interferon that has fewer flu-like symptoms and hematologic adverse effects than are typically observed with alpha IFNs, likely due to more focused expression of the IFN-lambda receptor. A Phase 1b study of PEG-IFN-λ in HCV genotype 1 patients who relapsed after treatment with IFN-alpha + RBV or are treatment-naïve
is ongoing. Initial results previously presented demonstrated that weekly administration of PEG-IFN-λ as a single agent was well-tolerated and associated with greater HCV RNA reduction than every other week administration.
Methods: This open-label dose-ranging study is evaluating 4 weeks of PEG-IFN-λ administered QW subcutaneously in combination with daily RBV (1000 or 1200 mg/day) in patients with HCV genotype 1 and prior relapse. A treatment-naïve cohort is currently being enrolled. Assessments include adverse events (AEs), laboratory values, and changes in HCV RNA.
Results: A total of 16 patients with chronic HCV and prior relapse have completed 4 weeks of treatment with PEG-IFN-λ QW (0.5 [n=4], 0.75 [n=3], 1.5 [n=6], and 2.25 [n=3] µg/kg) and daily RBV.
Treatment has been well-tolerated with minimal flu-like symptoms and no significant hematologic changes other than RBV-associated decreases in hemoglobin. The most common AEs, regardless of dose level, are fatigue (4/16; 25%), nausea (4/16; 25%), and insomnia (3/16; 19%), all Grade 1 or 2.
As previously reported, one patient treated at PEG-IFN-λ 1.5 µg/kg QW + RBV experienced reversible Grade 3/4 increases in ALT, AST, and bilirubin. No other patient receiving PEG-IFN-λ + RBV has experienced clinically-significant elevations in these parameters.
Dose-dependent antiviral activity in patients completing 4 weeks of treatment has been observed at all dose levels, with 12/16 (75%) relapse patients achieving >2-log decrease (range 0.1-5.6) in HCV RNA, and all 3 subjects treated at 2.25 µg/kg achieving HCV RNA levels <1000 IU/mL at Day 29.
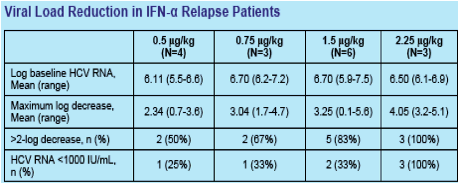
Conclusions: PEG-IFN-λ QW subcutaneously + daily RBV administered for 4 weeks has been well-tolerated with minimal flu-like symptoms or hematologic effects in patients with chronic HCV and prior relapse. This treatment is also associated with robust antiviral activity across a broad range of doses.
INTRODUCTION
Treatment of chronic hepatitis C (HCV) with PEG-Interferon-α
(PEG-IFN-α) and ribavirin (RBV) is frequently associated with significant
toxicities, dose reductions and avoidance of treatment1
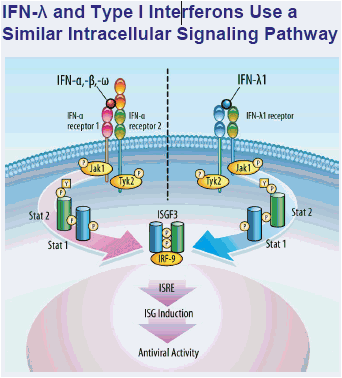
PEG-IFN Lambda 1a (PEG-IFN-λ) is in development as a new treatment for chronic HCV
- Member of the Type III/λ IFN family2,3 which binds to a unique
receptor with more restricted distribution than IFN-α/Type-I IFN receptors
- Potential for more favorable tolerability and side-effect profile
(expected to have less hematological toxicity) than IFN-α or other Type-1 IFNs
Here we report the four-week antiviral activity, safety and pharmacokinetic (PK)
results from the Phase 1b dose ranging study in IFN-α relapse and treatment naïve genotype-1 HCV patients

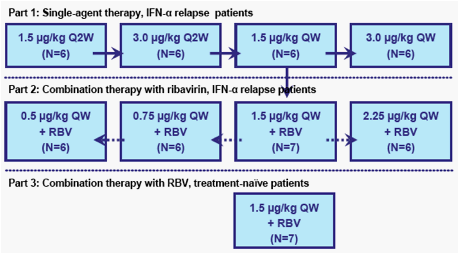

*One patient discontinued due to an unrelated adverse drug reaction to meperidine
Antiviral Activity
Anti-viral activity was observed at all dose levels. Higher PEG-IFN-λ doses were associated with greater declines in HCV RNA (Figure 2)
2 of 7 (29%) patients in treatment-naïve cohort achieved RVR (undetectable HCV RNA by week 4)
Figure 2: Mean (SE) Change in HCV RNA from Baseline following Four-Weeks of PEG-IFN-λ + RBV in IFN-α Relapse and Treatment-Naïve Patients
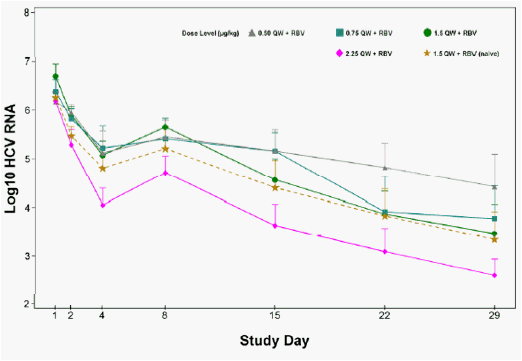
A summary of virological response for each treatment arm is shown in Table 2
Table 2: PEG-IFN-λ Virological Response
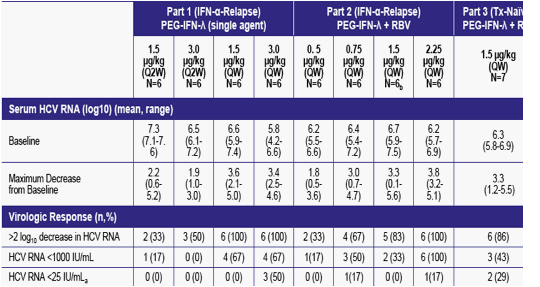
a Lower limit of detection= HCV RNA <25 IU/mL, b Excludes one patient who discontinued due to an unrelated adverse drug reaction to meperidine
Safety and Tolerability
Table 3: PEG-IFN-λ Adverse Event (AE) Summary
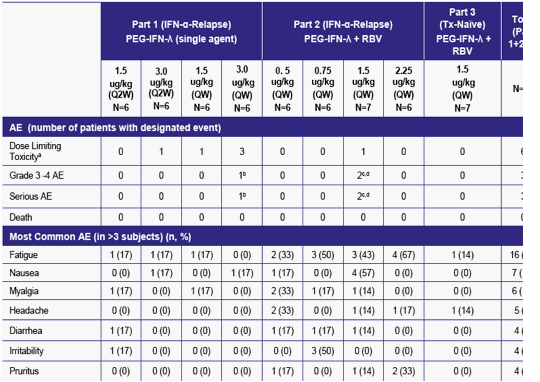
a 5 patients met protocol defined DLT criteria due to elevated hepatic lab values, one patient had DLT of possible grade 3 idiopathic
thrombocytopenic purpura (ITP)
b Possible grade 3 idiopathic thrombocytopenic purpura
c Grade 3 hepatotoxicity (SAE), pruritus, and increased serum lipase/amylase without signs of pancreatitis, probably related to study drug
d Adverse drug reaction to meperidine (acute respiratory distress) in a subject with a history of migraine headaches and COPD, unrelated to study drug
PEG-IFN-λ was generally well-tolerated with the majority of AEs mild (Grade 1) or moderate (Grade 2) in severity
Most common AEs in all patients were fatigue (29%) and nausea (13%)
Adverse events were more commonly observed in relapsed patients treated with PEG-IFN-λ + RBV (Part 2) than in naïve subjects treated with PEG-IFN-λ + RBV (Part 3) or when used as a single agent in relapsed subjects (Part 1)
6/56 (11%) experienced reversible increases in ALT/AST with or without increases in bilirubin that met thee protocol-defined criteria for DLT or withholding a PEG-IFN-λ dose (majority at the 3.0 µg/kg single agent dose)
Figure 3: Neutrophil and Platelet Counts following Four Weeks of PEG-IFN-λ + RBV in IFN-α Relapse and Treatment-Naïve Patients
No clinically-meaningful changes in neutrophil or platelet counts were observed during PEG-IFN-λ treatment
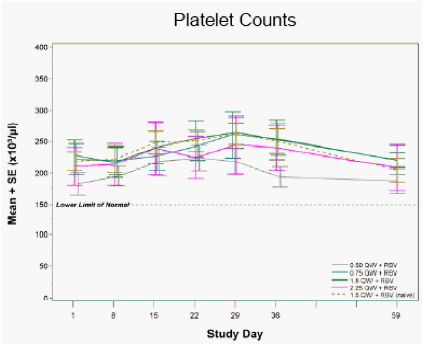
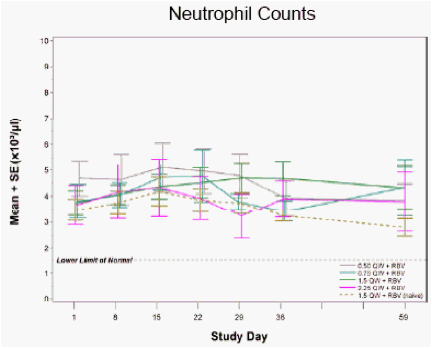
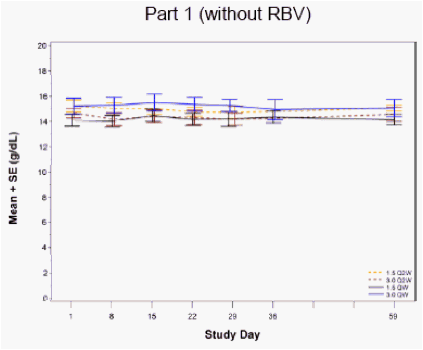
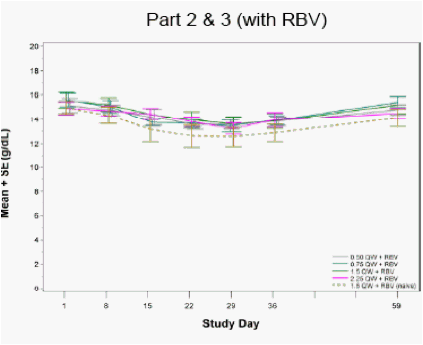
Figure 4: Hemoglobin Levels following Four Weeks of PEG-IFN-λ Treatment
No clinically-meaningful changes in hemoglobin levels were observed (apart from RBV associated reductions)
Pharmacokinetics
The exposure of PEG-IFN-λ appears to be dose dependent. Dose-normalized AUC was similar across the range of dose
No strong influence of body weight on exposure was observed
Modest accumulation during treatment was expected and observed, supporting the weekly dosing schedule proposed in Phase 2
Table 4: Single Dose Pharmacokinetic Parameters
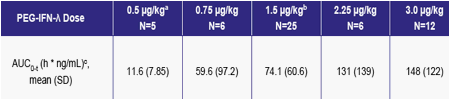
a One patient excluded due to AUC0-t and Cmax values inconsistent with the nominal dose administered
b One patient excluded due to lack of pharmacokinetic samples as a result of premature discontinuation of the study
c AUC0-t values for some patients were not estimated due to a lack of quantifiable data, and were imputed to be 1/2 of the lowest reported AUC0-t (6.78 h*ng/mL / 2 = 3.39 h*ng/mL).
REFERENCES
1. Ghany et al. Hepatology. 2009; 49(9): 1335-1374
2. Kotenko et al. Nature Immunology. 2003; 4(1): 69-77
3. Sheppard et al. Nature Immunology. 2003; 4(1):63-68
|
| |
|
 |
 |
|
|