 |
 |
 |
| |
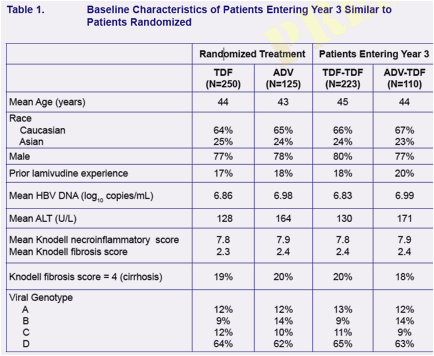
Three Years of Tenofovir Disoproxil Fumarate (TDF) Treatment in HBeAg-Negative Patients with Chronic Hepatitis B (Study 102)
|
| |
| |
Reported by Jules Levin
60th Annual Meeting of the American Association
for the Study of Liver Diseases
October 30 - November 3, 2009
Boston, Massachusetts, USA
P Marcellin1, M Buti2, Z Krastev3, G Germanidis4, K Kaita5, I Kotzev6, P Buggisch7, F Weilert8, H Trinh9, S Gurel10, E J Heathcote11, J Sorbel12, J Anderson12, E Mondou12, F Rousseau12
1Hopital Beaujon, University of Paris, Clichy France; 2Servicio de Medicina Interna Hepatologia, Hospital General Universitari Vall d'Hebron and Ciberehd, Barcelona, Spain; 3University Hospital "St Ivan Rilsky", Sofi a, Bulgaria; 4Papageorgiou Peripheral General Hospital of Thessaloniki, Thessaloniki, Greece; 5John Buhler Research Centre, University of Manitoba, Winnipeg MB Canada; 6University Hospital "Sveta Marina", Varna Bulgaria; 7Universitaetsklinik Eppendorf, Hamburg, Germany; 8Waikato Hospital, Hamilton, New Zealand; 9San Jose Gastroenterology, San Jose, CA; 10University of Uludag, Bursa Turkey; 11University of Toronto, Toronto ON Canada, 12Gilead Sciences, Durham NC

BACKGROUND
Tenofovir DF (TDF) was approved for HIV-1 in 2001 and chronic hepatitis B (CHB) in 2008 : ∼ 2.4 million patient-years of experience
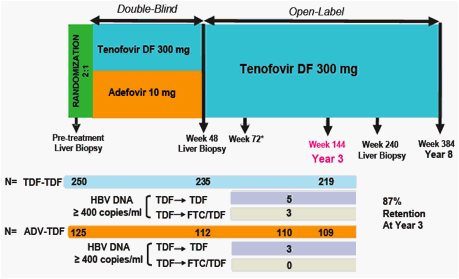
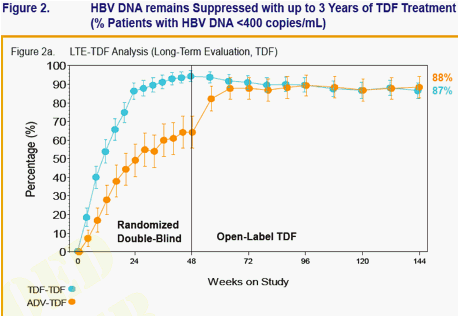
Week 48 Phase 3 data showed TDF superior to adefovir dipivoxil (ADV):
- 93% of HBeAg-negative TDF-treated patients (versus 63% of ADV-treated patients) had HBV DNA < 400 copies/mL
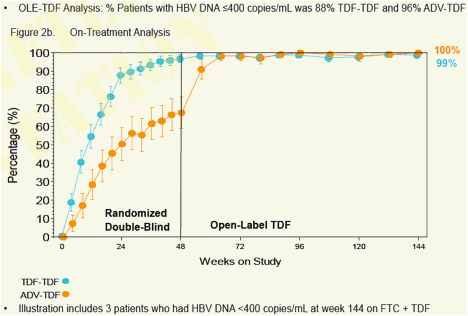
Week 96 Open-Label TDF data showed:
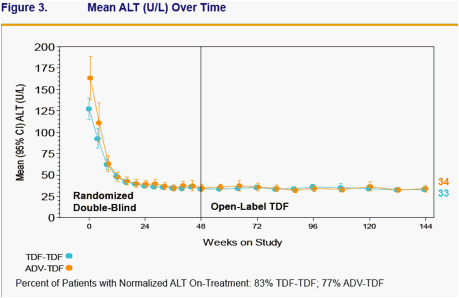
- Both stable and viremic patients on ADV can effectively switch to TDF and achieve or maintain viral suppression (HBV DNA < 400 copies/mL) and normal ALT levels
- Patients treated with TDF for 96 weeks maintained viral suppression and normal ALT levels
OBJECTIVE
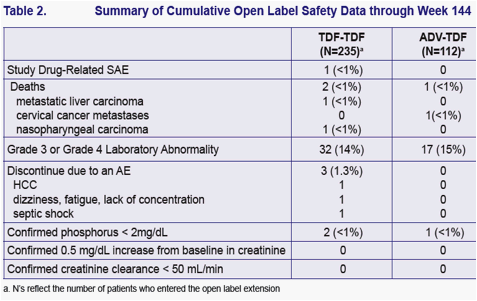
Evaluate the efficacy and safety of up to 3 years of TDF therapy


* Patients had the option to add FTC at the discretion of the investigator if confi rmed HBV DNA ≥400 copies/ml at week 72 or beyond





|
| |
|
 |
 |
|
|