 |
 |
 |
| |
Telaprevir (TVR, T) q8h or q12h Combined With Either Peginterferon (PEG-IFN, P) Alfa-2a or Alfa-2b and Ribavirin (RBV, R) in Treatment-Naïve Genotype 1 Hepatitis C: Week 12 Interim Results of the Randomized, Open-Label, Multicenter Phase 2 Study C208
|
| |
| |
Reported by Jules Levin
13th ISVHLD Wash DC March 20-24 2009
Xavier Forns,1 Patrick Marcellin,2 Tobias Goser,3 Peter Ferenci,4 Frederik Nevens,5 Giampiero Carosi,6 Joost P, Drenth,7 Katrien De Backer,8 Rolf van Heeswijk,8 Els De Paepe,8 Gaston Picchio,9 Maria Beumont-Mauviel8
1 Liver Unit, University of Barcelona, Barcelona, Spain; 2 Hopital Beaujon, Clichy, France; 3 Klinikum der Universitat zu Koln, Koln, Germany; 4 Department of Internal Medicine, Medical University of Vienna, Vienna, Austria; 5 Department of Hepatology, University Hospital Gasthuisberg, Leuven, Belgium; 6 Clinic of Infectious and Tropical Diseases, University of Brescia, Brescia, Italy; 7 Radboud University Nijmegen Medical Center, Nijmegen, Netherlands; 8 Tibotec BVBA, Mechelen, Belgium; 9 Tibotec Inc., Yardley, PA, USA.


Figure 1. C208 Study Design.
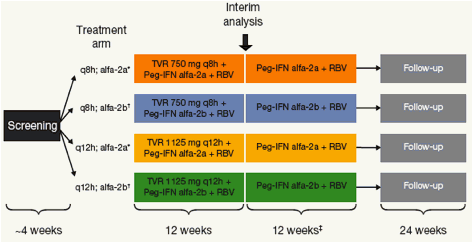
*TVR + Peg-IFN alfa-2a (180 µg/wk) + RBV (1000-1200 mg/d); TVR + Peg-IFN alfa-2b (1.5 µg/kg/wk) + RBV (800-1200 mg/d);If undetectable HCV RNA from Week 4 through Week 20. All patients not meeting this criterion receive 36 additional weeks of Peg-IFN + RBVq8h



Table 1. Demographics and Baseline Characteristics.
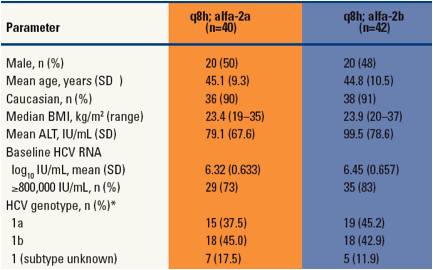
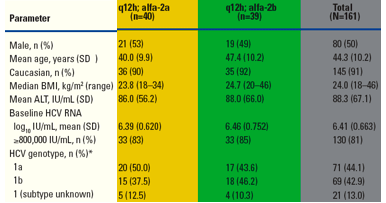
*Determined by TRUGENE HCV 5'NC genotyping assay (Siemens Medical Solutions Diagnostics)
SD = standard deviation; BMI = body mass index; ALT = alanine aminotransferase
Figure 3. Virologic Response Rate at Week 4 (ITT, NC=F).
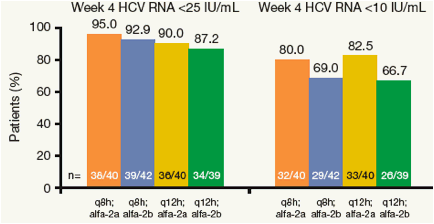
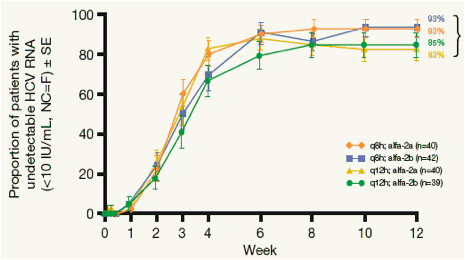
No treatment group comparisons were statistically significant

Figure 4. Virologic Response Rate Through Week 12 (ITT, NC=F).

shown are number (%) of patients with undetectable HCV RNA (<10 IU/mL)







|
| |
|
 |
 |
|
|