 |
 |
 |
| |
Loss of HBsAg in Nucleoside-Naïve HBeAg(+) Chronic Hepatitis B Patients Following Treatment with Entecavir or Lamivudine: Evaluation of HBV Genotypes
|
| |
| |
Reported by Jules Levin
AASLD, The Liver Meeting in Boston, MA, October 30 - November 3, 2009
Robert Gish1, Ting-Tsung Chang2, Ching-Lung Lai3, Robert de Man4, Fred Poordad5, Dong Xu6, Helena Brett-Smith6, Melissa Harris6, Uchenna Iloeje6, Hong Tang6
1Division of Hepatology and Complex GI, Physicians Foundation California Pacific Medical Center, San Francisco, California, USA; 2National Cheng Kung University Medical College, Tainan, Taiwan; 3Department of Medicine, University of Hong Kong, Hong Kong SAR, China; 4Erasmus Medical Center, University Hospital Rotterdam, The Netherlands; 5Department of Hepatology and Liver Transplantation, Cedars-Sinai Medical Center, Los Angeles, California, USA; 6Bristol-Myers Squibb Company, Research & Development, USA

Background
Spontaneous HBsAg seroclearance has been estimated to occur at a rate of 0.5% to 1.7% per year1-4
Rate of HBsAg seroclearance varies among different patient populations1-5
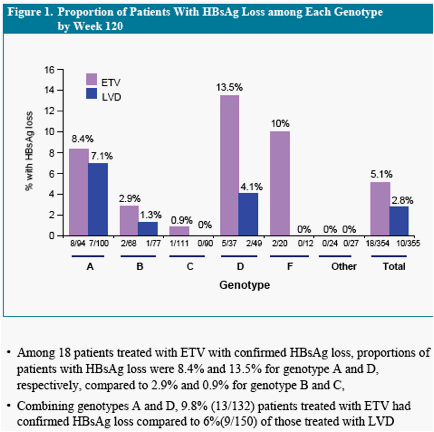
In the phase III study ETV-022, 5.1% of nucleoside-naïve HBeAg(+) chronic
hepatitis B (CHB) patients treated with entecavir (ETV) had confirmed HBsAg
loss by week 1206
In this analysis we evaluated the distribution of HBsAg loss according to the HBV
genotypes in patients treated with ETV or lamivudine (LVD) in Study ETV-022
Objective
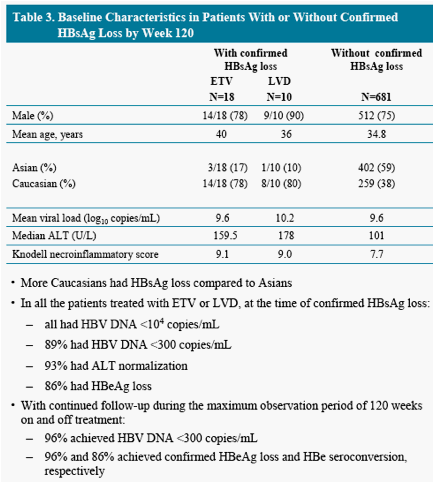
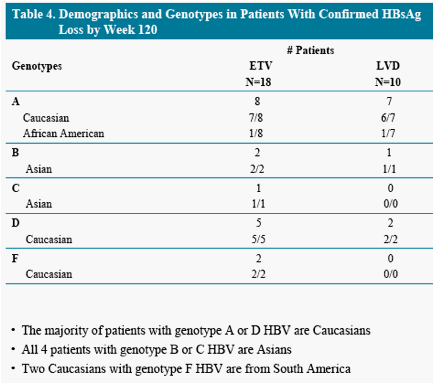
To describe characteristics of patients who achieved a confirmed HBsAg loss
treated with entecavir (ETV) or lamivudine (LVD) in study ETV-022


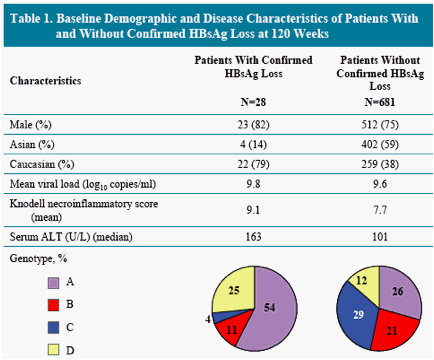
* Others include genotype E, mixed genotypes, indeterminate and subjects with missing genotype information



|
| |
|
 |
 |
|
|