 |
 |
 |
| |
Efficacy and safety of albinterferon alfa-2b in combination with ribavirin in treatment-naïve patients with chronic hepatitis C genotype 1
|
| |
| |
Reported by Jules Levn
AASLD Nov 2009, Boston, MA
M. Sulkowski, S. Zeuzem, E. Lawitz, M. Grigorescu, A. Tice,
V. Rustgi, M. Rodriguez-Torres, Y. Lurie, J. Cianciara, B.R. Bacon, V. Bain, W. Kryczka, E. Pulkstenis, M. Subramanian, J. McHutchison
Sponsored by Human Genome Sciences, Inc., Rockville, Maryland, US, and Novartis Pharma AG, Basel, Switzerland



aAdjusted for baseline stratification factors HCV RNA, BMI, and race per US Food and Drug Administration request; CI, confidence interval.

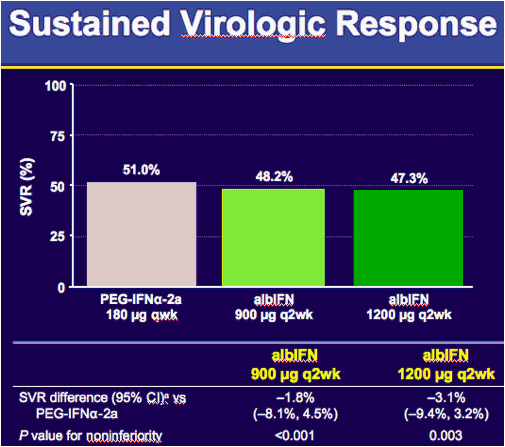
Secondary analyses of the primary efficacy endpoint included noninferiority testing in the ITT population but unadjusted for baseline stratification factors, as well as in the per protocol population (adjusted and unadjusted). The per protocol analysis included subjects who received the treatment assigned via randomization, met major entry criteria, and did not have missing SVR data (Section 5.9.8.1.5) or administration of protocol-prohibited alternative anti-hepatitis C therapy prior to assessment of SVR, or prior to meeting criteria declaring them non-responders (lack of EVR12 or HCV RNA ≥ 100 IU/mL at Week 24 or beyond). As expected, SVR rates were slightly higher in the per protocol population than in the ITT population in all groups (Table 7-3). Both doses of albIFN achieved noninferiority vs PEG-IFNα2a in each of the secondary analyses.
Other analyses of the primary efficacy endpoint were requested by FDA following the
pre-BLA meeting (22 June 09). FDA per-protocol analysis of SVR (adjusted and unadjusted based on baseline factors) included all subjects who completed the study without major deviations from the protocol entry criteria (see Section 5.9.8.1.5). Subjects who discontinued treatment (for all reasons except LOE), had inadequate adherence to drug therapy (ie, received < 80% of IFN or RBV), who received additional medication intented to treat HCV, or who did not not return for their final follow-up visit were excluded from the analysis. Both doses of albIFN achieved noninferiority vs PEG-IFNα2a in each of the FDA per-protocol analyses.
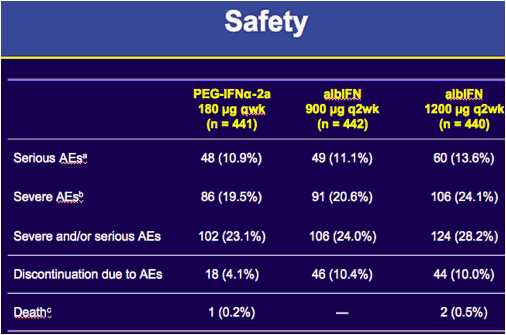
aAdverse events (AEs) that were fatal, life-threatening, resulted in hospitalisation, or were otherwise medically important; bgrade 3/4 severity; c1 suicide with PEG-IFNα-2a 180 µg qwk, 1 interstitial lung disease (ILD) with albIFN 1200 µg q2wk, and 1 bacterial pneumonia with albIFN 1200 µg q2wk.
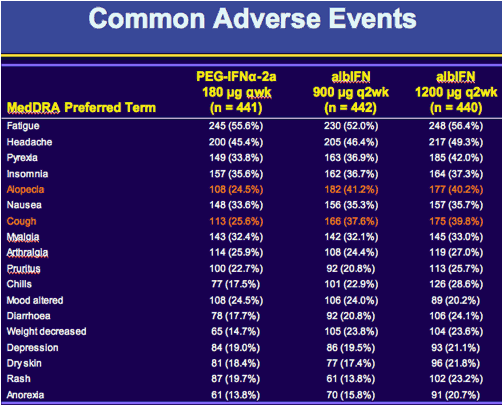

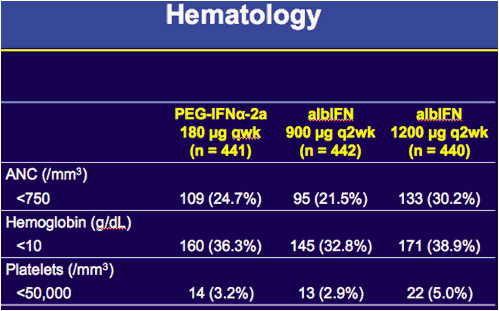
|
| |
|
 |
 |
|
|