 |
 |
 |
| |
Resistance Surveillance for up to 144 Weeks in HBeAg+ and HBeAg- Hepatitis B Patients Treated with Tenofovir DF Showed No Relationship Between Virologic Breakthrough and Emergence of Genotypic Changes in HBV Polymerase
|
| |
| |
Reported by Jules Levin
60th Annual Meeting of the American Association
for the Study of Liver Diseases
October 30 - November 3, 2009
Boston, Massachusetts, USA
A Snow-Lampart,1 B Chappell,1 M Curtis,1 Y Zhu,1 J Heathcote,2 P Marcellin,3 and K Borroto-Esoda1
1Gilead Sciences, NC, USA; 2Toronto General Hospital, Ontario, Canada; 3Hospital Beaujon, Clichy, France

INTRODUCTION
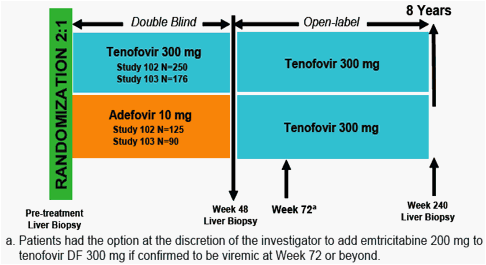
Tenofovir disoproxil fumarate (tenofovir DF, TDF) is a nucleotide analog with potent antiviral activity in patients mono-infected with HBV and co-infected with HIV-1 and HBV
HBV pol/RT resistance mutations have been identifi ed following administration of other oral anti-HBV agents (lamivudine, adefovir dipivoxil, entecavir, and telbivudine)
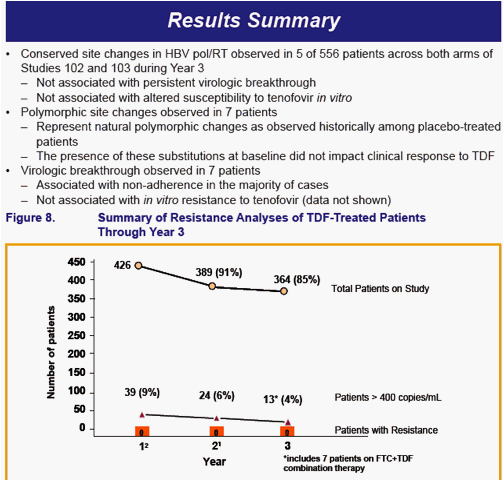
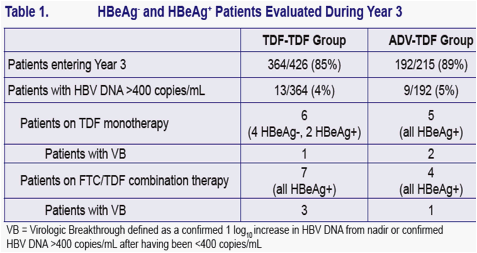
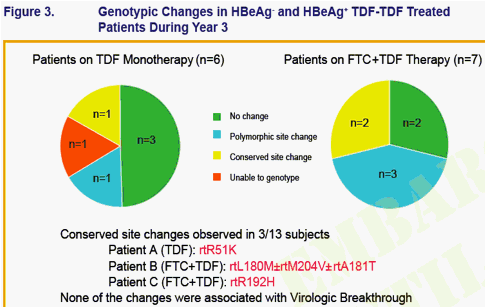
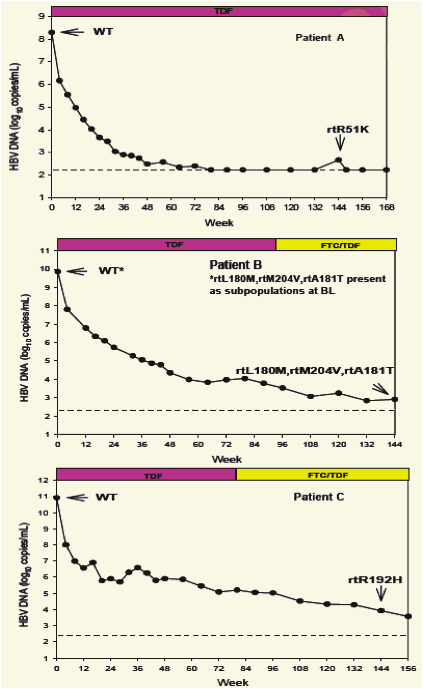
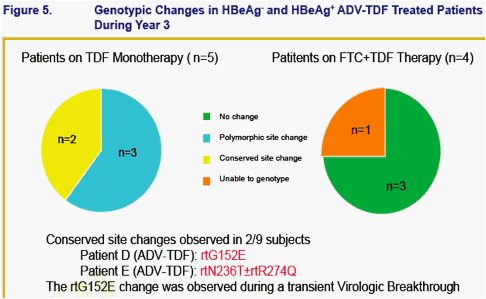
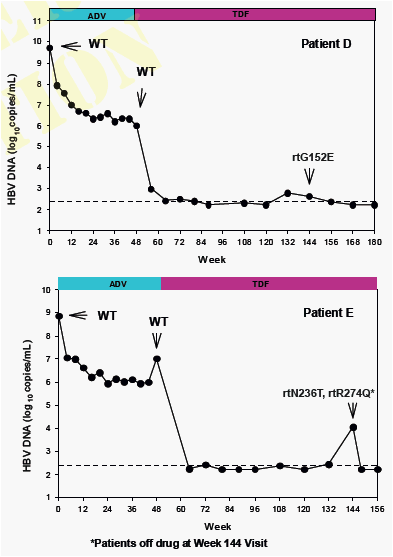
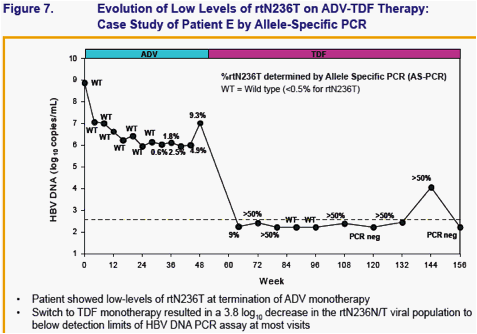
No amino acid substitutions associated with resistance to tenofovir DF were detected in the HBV pol/RT during the fi rst 96 weeks of TDF treatment of HBeAg- and HBeAg+ patients in Studies 102 and 1031
OBJECTIVES
To identify amino acid substitutions in the HBV pol/RT following up to 144 weeks of therapy with TDF 300 mg once daily
To evaluate the effects of these substitutions on the clinical response to TDF monotherapy in chronic hepatitis B
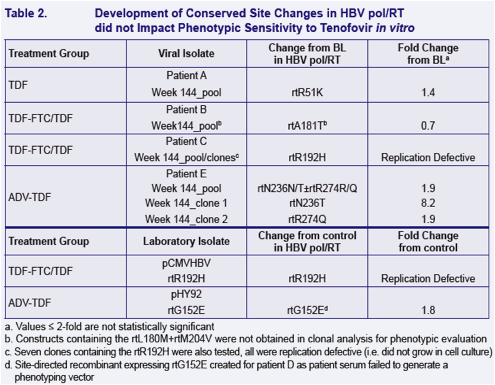
To determine whether these substitutions alter susceptibility to tenofovir using in vitro HBV replication assays and to evaluate the cross-resistance profi le of these substitutions







REFERENCES
1. Snow-Lampart et. al. AASLD 2008, Poster #977
2. Snow-Lampart et. al. EASL Monothematic conference on Hepatitis B and C Virus Resistance to Antiviral Therapy 2008, Poster #4 1. Snow-Lampart et. al. AASLD 2008, Poster #977
2. Snow-Lampart et. al. EASL Monothematic conference on Hepatitis B and C Virus Resistance to Antiviral Therapy 2008, Poster #4
|
| |
|
 |
 |
|
|